VMAT2 Inhibitors Market Overview
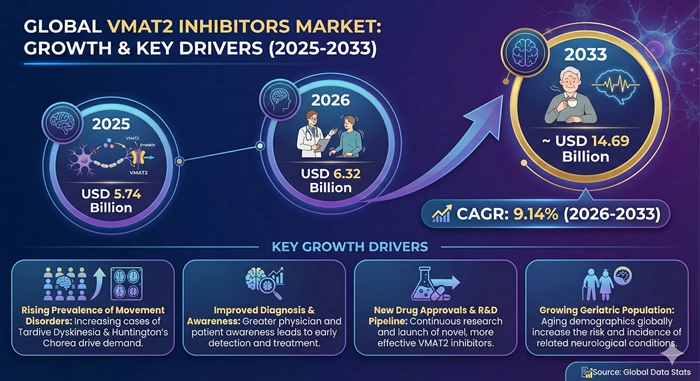
The global VMAT2 inhibitors market size is valued at USD 5.74 billion in 2025 and is predicted to increase from USD 6.32 billion in 2026 to approximately USD 14.69 billion by 2033, growing at a CAGR of 9.14% from 2026 to 2033.
The increasing prevalence of movement disorders including tardive dyskinesia and Huntington's disease worldwide has created substantial demand for effective vesicular monoamine transporter 2 inhibitors that provide symptomatic relief by modulating dopamine release in neural pathways. These pharmaceutical agents work by reversibly blocking the VMAT2 protein responsible for loading monoamines like dopamine into synaptic vesicles, thereby reducing excessive dopamine activity that causes involuntary movements and motor dysfunction. Healthcare providers are increasingly prescribing VMAT2 inhibitors as first-line treatments for patients experiencing debilitating hyperkinetic movements associated with long-term antipsychotic medication use or inherited neurodegenerative conditions. The market benefits from growing awareness about these movement disorders among neurologists and psychiatrists, expanding regulatory approvals for broader therapeutic indications, improved safety profiles of newer drug formulations, rising healthcare expenditure on specialty pharmaceuticals, and continuous innovation by leading pharmaceutical companies developing next-generation molecules with enhanced efficacy and tolerability.
AI Impact on the VMAT2 Inhibitors Industry
Artificial Intelligence Transforms Drug Discovery Processes and Enhances Clinical Trial Efficiency in Neurological Therapeutics Development
Artificial intelligence is fundamentally reshaping the VMAT2 inhibitors market by accelerating drug discovery timelines and optimizing clinical trial designs through advanced computational modeling and predictive analytics. AI-powered platforms analyze vast molecular databases to identify promising VMAT2-targeting compounds with improved binding affinity, reduced off-target effects, and enhanced pharmacokinetic properties, dramatically shortening the research phase that traditionally requires years of laboratory experimentation. Machine learning algorithms predict patient responses to specific VMAT2 inhibitors based on genetic markers, disease severity, concurrent medications, and demographic factors, enabling pharmaceutical companies to stratify clinical trial participants more effectively and increase the likelihood of demonstrating statistically significant efficacy outcomes. These intelligent systems process neurological imaging data from thousands of patients to identify subtle treatment response patterns invisible to human observers, providing deeper insights into optimal dosing regimens and treatment durations. AI-driven safety monitoring tools continuously analyze adverse event reports across global patient populations, detecting rare side effects earlier than conventional pharmacovigilance methods and enabling proactive risk mitigation strategies.
Furthermore, AI is revolutionizing patient identification and recruitment for VMAT2 inhibitor clinical trials by mining electronic health records to locate individuals meeting specific enrollment criteria, addressing one of the most persistent challenges in rare neurological disease research. Natural language processing algorithms extract relevant clinical information from unstructured medical notes, identifying patients with tardive dyskinesia or Huntington's disease who might benefit from VMAT2 inhibitor therapy but remain undiagnosed or undertreated. Predictive modeling helps pharmaceutical companies forecast market demand across different geographic regions by analyzing epidemiological trends, prescription patterns, demographic shifts, and healthcare access indicators, informing strategic decisions about manufacturing capacity and distribution networks. AI-powered digital therapeutics integrated with VMAT2 inhibitor treatments provide real-time symptom tracking and medication adherence monitoring, generating valuable real-world evidence that strengthens regulatory submissions and supports favorable reimbursement decisions. As AI capabilities continue advancing, the VMAT2 inhibitors market will benefit from faster innovation cycles, more personalized treatment approaches, improved patient outcomes, and enhanced commercial success for novel therapies.
Growth Factors
Rising Antipsychotic Usage and Growing Awareness of Movement Disorder Treatments Drive Market Expansion
The VMAT2 inhibitors market is experiencing robust growth fueled by the dramatic increase in antipsychotic medication prescriptions worldwide, which inadvertently creates a larger patient population at risk of developing tardive dyskinesia as an iatrogenic side effect of long-term dopamine receptor antagonist therapy. Mental health conditions including schizophrenia, bipolar disorder, major depressive disorder with psychotic features, and various behavioral disturbances affect hundreds of millions of individuals globally, with many requiring prolonged or lifelong antipsychotic treatment to maintain psychiatric stability and prevent relapse. Epidemiological studies indicate that approximately 20 to 30 percent of patients taking first-generation antipsychotics and 10 to 15 percent of those on second-generation agents develop tardive dyskinesia, characterized by involuntary repetitive movements of the face, tongue, limbs, and trunk that significantly impair quality of life and social functioning. The cumulative risk increases with treatment duration, creating a steadily expanding pool of patients who become candidates for VMAT2 inhibitor therapy when movements become problematic despite antipsychotic dose adjustments or medication switches. Psychiatrists and neurologists have historically struggled with limited treatment options for established tardive dyskinesia, but the approval of selective VMAT2 inhibitors has transformed clinical practice by offering effective symptom control without requiring discontinuation of necessary psychiatric medications.
Growing awareness among healthcare professionals and patients about the existence of FDA-approved treatments for tardive dyskinesia and Huntington's disease chorea represents another critical driver accelerating the VMAT2 inhibitors market expansion. Medical education initiatives sponsored by pharmaceutical companies, professional medical associations, and patient advocacy organizations have successfully elevated the visibility of these conditions and highlighted the availability of targeted therapies that can meaningfully improve patient outcomes. Continuing medical education programs train psychiatrists to recognize early signs of tardive dyskinesia and initiate appropriate screening protocols, leading to earlier diagnosis and treatment intervention before movements become severe and potentially irreversible. Patient support groups and online communities share information about VMAT2 inhibitors, empowering individuals to advocate for themselves during medical consultations and request evaluations when experiencing movement symptoms. Direct-to-consumer advertising in markets where permitted has raised public awareness that tardive dyskinesia is not an inevitable consequence that must be endured but rather a treatable condition with available pharmacological interventions. Healthcare systems are implementing systematic screening programs to identify tardive dyskinesia cases among psychiatric patient populations, creating structured pathways for VMAT2 inhibitor initiation and expanding treatment penetration beyond specialty movement disorder clinics to general psychiatry practices.

Market Outlook
Pipeline Expansion and Label Extensions Position Market for Sustained Long-Term Growth
The VMAT2 inhibitors market outlook remains exceptionally positive as pharmaceutical companies advance robust clinical development pipelines exploring additional therapeutic applications beyond the currently approved indications of tardive dyskinesia and Huntington's disease chorea. Neurocrine Biosciences initiated Phase 2 clinical studies evaluating NBI-1065890, a potentially longer-acting VMAT2 inhibitor designed to improve convenience and adherence through extended dosing intervals, while also launching Phase 1 trials for NBI-1140675, a novel second-generation molecule with refined pharmacological properties aimed at enhancing efficacy while minimizing side effects. Ongoing research investigates VMAT2 inhibitor efficacy for treating tics associated with Tourette syndrome, choreiform movements in other neurological conditions, and even psychiatric symptoms where dopamine modulation might provide therapeutic benefits. Label expansions for existing approved agents represent significant commercial opportunities, as demonstrating efficacy in additional patient populations can substantially broaden prescribing indications and expand addressable markets without requiring entirely new drug development programs. Combination therapy approaches pairing VMAT2 inhibitors with complementary neurological or psychiatric medications offer potential synergistic benefits that could improve outcomes for patients with complex symptom profiles.
The maturation of generic competition for tetrabenazine and potential future patent expiries for newer agents will create complex market dynamics balancing price erosion against volume expansion as lower-cost alternatives improve treatment accessibility for price-sensitive patients and healthcare systems. Generic manufacturers including Lupin, Aurobindo Pharma, and other companies are positioned to enter markets as exclusivity periods expire, potentially democratizing access to VMAT2 inhibitor therapy for patients in emerging economies where branded pricing remains prohibitively expensive. Pharmaceutical companies holding branded product franchises are responding by emphasizing differentiated clinical benefits including superior safety profiles, more convenient dosing regimens, comprehensive patient support programs, and robust real-world evidence demonstrating long-term efficacy and tolerability. Biosimilar development remains less relevant for small molecule VMAT2 inhibitors compared to biologics, but pharmaceutical innovation continues through next-generation molecules with improved pharmacokinetic properties, reduced drug-drug interaction potential, and mechanisms targeting specific VMAT2 isoforms. The expanding integration of telemedicine into neurological and psychiatric care creates opportunities for remote patient monitoring and medication management that could increase VMAT2 inhibitor utilization by reducing barriers related to specialty clinic access.
Expert Speaks
-
Kyle W. Gano, Ph.D., CEO of Neurocrine Biosciences, stated in February 2025 that he takes pride in the significant advancements achieved last year, particularly the sustained growth of INGREZZA for individuals suffering from tardive dyskinesia and chorea linked to Huntington's disease, emphasizing that with a rapidly evolving pipeline and solid financial standing, the company is strategically positioned to establish itself as a leader in the neuroscience field.
-
Richard Francis, CEO of Teva Pharmaceutical Industries, emphasized during 2024 that Teva remains committed to its strategic priorities of delivering innovative medicines addressing complex neurological conditions, leveraging the company's expertise in specialty pharmaceuticals and generic drug development to ensure patients worldwide have access to essential treatments that improve quality of life.
-
Thomas Apker, CEO of Bausch Health Companies, has consistently highlighted throughout 2024 and 2025 that the company's neurology and psychiatry portfolio represents a critical component of their diversified pharmaceutical business, with continued investment in research, development, and commercialization of therapies addressing significant unmet medical needs in movement disorders and other neurological conditions affecting millions of patients globally.
Key Report Takeaways
-
North America dominates the VMAT2 inhibitors market with approximately 39.5% market share in 2025, driven by high diagnosis rates of tardive dyskinesia and Huntington's disease, advanced neurological care infrastructure, favorable reimbursement policies supporting specialty pharmaceuticals, strong presence of key pharmaceutical companies, early adoption of innovative therapies, and extensive awareness campaigns targeting healthcare professionals and patients about available treatment options.
-
Asia Pacific represents the fastest-growing regional market with the highest projected CAGR during the forecast period from 2026 to 2033, fueled by rapidly improving healthcare infrastructure, expanding networks of specialty neurology clinics, rising awareness of movement disorders among medical professionals, increasing diagnosis rates as screening programs proliferate, growing geriatric populations susceptible to neurodegenerative conditions, and expanding access to specialty medications through improved insurance coverage and patient assistance programs.
-
Valbenazine segment leads the market with a commanding 54.2% market share in 2025 due to its favorable once-daily oral dosing regimen improving patient adherence, superior long-term safety profile demonstrated in extensive clinical trials, broad physician acceptance as first-line therapy for tardive dyskinesia, strong commercial infrastructure supporting widespread distribution, comprehensive patient support programs, established brand recognition under the trade name Ingrezza, and continuous post-marketing research reinforcing clinical confidence.
-
Tardive dyskinesia application accounts for the largest share at approximately 74.9% of the VMAT2 inhibitors market in 2025, reflecting the substantial patient population experiencing this iatrogenic movement disorder as a consequence of long-term antipsychotic therapy, rising prescription rates of psychiatric medications creating larger at-risk populations, increasing recognition among psychiatrists about the importance of screening and treatment, strong clinical evidence demonstrating VMAT2 inhibitor efficacy in reducing abnormal involuntary movements, and growing awareness among patients empowering them to seek treatment.
-
Oral administration route dominates with 94.5% market share in 2025, attributed to patient preference for convenient home-based medication administration avoiding clinic visits, well-established oral formulations for all approved VMAT2 inhibitors, ease of dose titration enabling personalized treatment optimization, superior adherence rates compared to injectable alternatives, and suitability for long-term chronic disease management requiring sustained daily therapy throughout extended treatment periods.
-
Deutetrabenazine segment projected to achieve fastest growth throughout the forecast period, expanding rapidly as clinical experience demonstrates its flexible dosing schedule accommodating individual patient needs, reduced peak-to-trough concentration variability improving tolerability, proven efficacy across multiple movement disorders including both tardive dyskinesia and Huntington's disease chorea, increasing physician familiarity through targeted medical education initiatives, and expanding market penetration as healthcare providers seek alternatives to first-generation VMAT2 inhibitors.
Market Scope
| Report Coverage Details | |
|---|---|
| Market Size by 2033 | USD 14.69 Billion |
| Market Size by 2025 | USD 5.74 Billion |
| Market Size by 2026 | USD 6.32 Billion |
| Market Growth Rate from 2026 to 2033 | CAGR of 9.14% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2033 |
| Segments Covered | Drug Type, Application, Route of Administration, Distribution Channel, Region |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
Market Dynamics
Drivers Impact Analysis
Increasing Prevalence of Movement Disorders and Regulatory Support Accelerate Market Growth
| Impact Factor | Details |
|---|---|
| ≈ Impact on CAGR Forecast | +2.0% to +2.5% |
| Geographic Relevance | Global with strongest impact in developed markets |
| Impact Timeline | Immediate and sustained through 2033 |
The escalating prevalence of movement disorders worldwide represents a fundamental driver propelling the VMAT2 inhibitors market forward as both iatrogenic conditions like tardive dyskinesia and genetic neurodegenerative diseases like Huntington's disease affect increasingly larger patient populations requiring effective pharmacological management. Tardive dyskinesia emerges as a delayed consequence of dopamine receptor antagonist exposure, with cumulative incidence rates reaching 30 percent or higher among patients receiving traditional antipsychotic medications for chronic psychiatric conditions over extended treatment durations. The aging global population exhibits higher rates of psychiatric disorders requiring antipsychotic treatment, creating demographic trends that inherently expand the pool of individuals at risk for developing tardive dyskinesia during their lifetimes. Huntington's disease, while rarer than tardive dyskinesia, affects approximately 6 to 9 per 100,000 individuals in Western populations and causes progressive motor, cognitive, and psychiatric deterioration requiring comprehensive symptomatic management including VMAT2 inhibitors to control choreiform movements. Genetic testing advances enable earlier Huntington's disease diagnosis before symptom onset, creating opportunities for proactive therapeutic planning and earlier treatment initiation when movements become functionally significant.
Supportive regulatory environments and favorable government policies represent another powerful driver accelerating the VMAT2 inhibitors market expansion by facilitating drug development, expediting approvals, and improving patient access to these specialty neurological therapies. Regulatory agencies including the FDA have granted orphan drug designations and priority review status to VMAT2 inhibitors targeting rare neurological conditions, providing development incentives including extended market exclusivity periods, reduced regulatory fees, and streamlined approval pathways that lower barriers to market entry. Government-funded research initiatives invest substantial resources in neurological disease research, advancing scientific understanding of movement disorder pathophysiology and identifying new therapeutic targets that guide pharmaceutical innovation. Patient assistance programs sponsored by pharmaceutical companies in collaboration with government agencies and nonprofit organizations provide financial support enabling access to VMAT2 inhibitors for uninsured and underinsured patients who would otherwise face prohibitive out-of-pocket costs. Expanded insurance coverage mandates require payers to include specialty neurological medications on formularies, reducing administrative barriers like prior authorizations and step therapy requirements that historically delayed treatment initiation. International harmonization of regulatory standards facilitates simultaneous multinational drug development programs and accelerates global market launches following initial approvals.

Restraints Impact Analysis
High Treatment Costs and Potential Side Effects Present Growth Challenges
| Impact Factor | Details |
|---|---|
| ≈ Impact on CAGR Forecast | -0.8% to -1.1% |
| Geographic Relevance | Particularly acute in price-sensitive emerging markets |
| Impact Timeline | Ongoing challenge throughout forecast period |
The substantial cost associated with branded VMAT2 inhibitor therapies represents a significant restraint limiting market penetration, particularly in healthcare systems with constrained pharmaceutical budgets and among patients lacking comprehensive insurance coverage for specialty medications. Annual treatment costs for branded VMAT2 inhibitors frequently exceed USD 100,000 per patient when prescribed at therapeutic doses, creating financial barriers that prevent many individuals from accessing these potentially life-changing therapies despite clear medical indications. Private insurance companies and government payers implement utilization management strategies including prior authorization requirements, step therapy protocols mandating trials of less expensive alternatives before approving VMAT2 inhibitors, and quantity limits restricting prescriptions to specific dosing regimens regardless of individual patient needs. High patient cost-sharing obligations through deductibles, copayments, and coinsurance create affordability challenges even for insured individuals, with some facing thousands of dollars in annual out-of-pocket expenses that force difficult decisions about medication adherence. Healthcare systems in emerging economies often exclude expensive specialty neurological drugs from public formularies entirely, making VMAT2 inhibitors effectively unavailable to the majority of patients in countries representing large portions of global disease burden.
Concerns about potential adverse effects and tolerability issues associated with VMAT2 inhibitor therapy present another restraint affecting market expansion as both physicians and patients weigh potential benefits against documented side effect risks when making treatment decisions. Common adverse events including sedation, somnolence, fatigue, and extrapyramidal symptoms can significantly impact patients' daily functioning and quality of life, sometimes necessitating dose reductions or treatment discontinuation despite achieving therapeutic benefits on motor symptoms. Depression and suicidality represent particularly concerning risks that require careful monitoring, especially in psychiatric patient populations already vulnerable to mood disturbances due to underlying mental illness. Drug-drug interactions between VMAT2 inhibitors and other medications commonly prescribed to neurological and psychiatric patients create prescribing complexities and potential safety concerns requiring careful medication reconciliation and ongoing pharmacological surveillance. QT interval prolongation documented with some VMAT2 inhibitors necessitates baseline and periodic electrocardiographic monitoring, creating additional healthcare utilization and costs while potentially limiting use in patients with cardiac comorbidities. Prescribing physicians must balance the demonstrated efficacy of VMAT2 inhibitors in reducing abnormal involuntary movements against these tolerability considerations, with some concluding that risks outweigh benefits for patients with mild symptoms or those particularly vulnerable to specific adverse effects.
Opportunities Impact Analysis
Pipeline Expansion and Emerging Market Penetration Create Growth Avenues
| Impact Factor | Details |
|---|---|
| ≈ Impact on CAGR Forecast | +1.6% to +2.0% |
| Geographic Relevance | Global with emphasis on Asia Pacific and Latin America |
| Impact Timeline | Medium to long term (2027-2033) |
The development of next-generation VMAT2 inhibitors with improved pharmacological characteristics represents a substantial opportunity for the market as pharmaceutical innovation addresses limitations of currently available therapies and expands treatment options for clinicians and patients. Novel molecules in clinical development pipelines target enhanced selectivity for VMAT2 over other vesicular monoamine transporters, potentially reducing off-target effects while maintaining or improving therapeutic efficacy on movement symptoms. Extended-release formulations enable less frequent dosing schedules compared to immediate-release products requiring multiple daily administrations, improving convenience and adherence particularly among patients managing complex medication regimens for multiple comorbid conditions. Researchers are exploring prodrug strategies and alternative chemical modifications that optimize absorption, distribution, metabolism, and excretion properties to achieve more consistent plasma concentrations with reduced peak-to-trough variability that contributes to tolerability issues. Formulation innovations including orally disintegrating tablets benefit patients with dysphagia or swallowing difficulties common in neurological disease populations, expanding the accessible patient population. Combination products integrating VMAT2 inhibitors with complementary therapeutic agents offer potential for synergistic benefits and simplified treatment regimens, though regulatory approval pathways for fixed-dose combinations present development challenges.
Substantial untapped market potential exists in developing regions including Asia Pacific, Latin America, and Middle East where rising healthcare standards, expanding specialist networks, and improving diagnostic capabilities create opportunities for VMAT2 inhibitors market penetration currently limited by awareness gaps and infrastructure constraints. Economic development across emerging markets generates growing middle classes with increasing healthcare purchasing power and expectations for access to advanced therapies comparable to those available in Western countries. Government initiatives modernizing healthcare infrastructure establish specialty neurology centers and movement disorder clinics capable of diagnosing and managing conditions like tardive dyskinesia and Huntington's disease that previously went unrecognized or untreated. Physician education programs conducted by pharmaceutical companies, professional medical societies, and academic institutions raise awareness about movement disorders and familiarize neurologists and psychiatrists with VMAT2 inhibitor therapies, creating prescriber networks capable of supporting market development. Patient advocacy organizations emerging in developing regions educate communities about neurological conditions, reduce stigma surrounding psychiatric illnesses and their treatments, and advocate for policy changes improving access to specialty medications. International pharmaceutical companies are implementing tiered pricing strategies and establishing patient assistance programs specifically designed for emerging markets, making VMAT2 inhibitors financially accessible to broader patient populations while maintaining commercial viability.

Segment Analysis
By Drug Type
Valbenazine Maintains Market Leadership Through Clinical Differentiation and Strong Commercial Infrastructure
The valbenazine segment dominates the VMAT2 inhibitors market with an impressive 54.2% market share in 2025, driven by its status as the first VMAT2 inhibitor specifically approved for tardive dyskinesia treatment in 2017, providing critical first-mover advantages including early establishment of clinical evidence, brand recognition, and prescriber relationships that create substantial competitive moats. Marketed under the trade name Ingrezza by Neurocrine Biosciences, valbenazine offers a convenient once-daily oral dosing regimen that simplifies medication administration compared to alternatives requiring multiple daily doses or complex titration schedules, directly improving patient adherence and persistence on therapy. The molecule's favorable pharmacokinetic profile produces consistent plasma concentrations throughout the 24-hour dosing interval, minimizing peak-to-trough fluctuations that can trigger breakthrough symptoms or tolerability issues common with shorter-acting agents. Extensive clinical trial data spanning multiple Phase 3 studies demonstrate robust efficacy in reducing abnormal involuntary movement scores with response rates exceeding 50 percent of treated patients, providing the evidence base that gives physicians confidence when prescribing valbenazine as first-line therapy. Long-term safety data accumulated over years of post-marketing experience and extension studies document acceptable tolerability profiles with well-characterized adverse event patterns, further reinforcing clinical adoption.
North America represents the strongest market for valbenazine within the global VMAT2 inhibitors market, with United States prescriptions accounting for the majority of global sales driven by strong commercial infrastructure, comprehensive insurance coverage, direct-to-consumer marketing campaigns, and established relationships with key opinion leaders in neurology and psychiatry. Neurocrine Biosciences maintains dedicated sales forces targeting high-prescribing psychiatrists and neurologists, ensuring frequent contact and educational engagement that keeps valbenazine top-of-mind when treatment decisions are made. Comprehensive patient support programs provide resources including financial assistance, adherence tools, and educational materials that improve patient experiences and reduce barriers to treatment initiation and continuation. The company invests heavily in real-world evidence generation through observational studies and patient registries, continuously demonstrating valbenazine effectiveness in routine clinical practice settings that complement controlled trial data. Europe and Asia Pacific markets are experiencing rapid valbenazine adoption as regulatory approvals expand geographic availability and reimbursement access improves, with the segment projected to maintain strong growth throughout the forecast period supported by pipeline expansion into additional indications and ongoing commercialization investments.
By Application
Tardive Dyskinesia Application Commands Market Dominance Through Large Patient Population and Strong Clinical Evidence
The tardive dyskinesia application segment leads the VMAT2 inhibitors market with a commanding 74.9% market share in 2025, reflecting the substantial global burden of this iatrogenic movement disorder affecting millions of patients worldwide who have received long-term dopamine receptor antagonist therapy for psychiatric conditions. Tardive dyskinesia develops insidiously over months to years of antipsychotic exposure, causing involuntary stereotypical movements of the orofacial region, trunk, and limbs that significantly impair social functioning, occupational performance, and psychological well-being while creating profound distress for affected individuals and their families. The condition represents a major clinical concern for psychiatrists who must balance the psychiatric benefits of continued antipsychotic therapy against the neurological risks of movement disorder development, creating ethical dilemmas particularly when alternative psychiatric treatments prove less effective. Rising awareness of tardive dyskinesia among both healthcare providers and patients has increased diagnosis rates, with systematic screening programs identifying previously unrecognized cases and expanding the treated patient population. VMAT2 inhibitors represent the only FDA-approved pharmacological treatments specifically indicated for tardive dyskinesia, establishing their position as standard-of-care options when movements become functionally significant or distressing despite conservative management strategies.
Clinical evidence demonstrating VMAT2 inhibitor efficacy in reducing tardive dyskinesia severity through multiple randomized controlled trials has transformed treatment paradigms from nihilistic acceptance of irreversible movements to proactive symptom management with achievable therapeutic outcomes. Studies document clinically meaningful reductions in Abnormal Involuntary Movement Scale scores among 40 to 60 percent of treated patients, with some individuals experiencing complete or near-complete symptom resolution enabling return to normal social and occupational activities. Treatment responses typically emerge within 2 to 8 weeks of initiating therapy and continue improving with sustained treatment over months, providing tangible benefits that motivate medication adherence. The chronic nature of tardive dyskinesia necessitates indefinite VMAT2 inhibitor therapy for most patients, creating recurring revenue streams that support market sustainability and growth. Leading pharmaceutical companies including Neurocrine Biosciences, Teva Pharmaceutical Industries, and Bausch Health Companies compete vigorously for tardive dyskinesia market share through differentiated clinical positioning, robust medical affairs activities, and comprehensive commercial strategies. The segment benefits from favorable reimbursement policies as payers recognize tardive dyskinesia as a serious condition warranting access to specialized treatments, though prior authorization requirements and step therapy protocols remain common utilization management tactics.

Regional Insights
North America
Advanced Healthcare Infrastructure and Early Treatment Adoption Sustain Regional Market Leadership
North America maintains commanding leadership in the VMAT2 inhibitors market with approximately 39.5% of global market share in 2025, supported by sophisticated neurological and psychiatric care delivery systems featuring extensive networks of movement disorder specialists, comprehensive diagnostic capabilities, and established referral pathways connecting primary care providers with subspecialty expertise. The United States dominates regional performance with an estimated market value of USD 1.79 billion in 2025, driven by high prevalence of psychiatric conditions requiring long-term antipsychotic therapy that creates substantial at-risk populations for tardive dyskinesia development. American healthcare providers demonstrate strong awareness of VMAT2 inhibitor therapies through continuing medical education programs, medical literature, pharmaceutical representative detailing, and professional conference presentations that maintain high visibility for these products. Favorable reimbursement environments ensure most patients with commercial insurance or government coverage through Medicare and Medicaid can access VMAT2 inhibitors following prior authorization approval, though cost-sharing requirements create affordability challenges for some individuals. Strong pharmaceutical industry presence with companies like Neurocrine Biosciences headquartered in the United States facilitates rapid commercialization, extensive clinical research activities, and robust post-marketing surveillance generating real-world evidence.
Canada and Mexico contribute meaningfully to North American market performance, with Canadian provincial drug plans generally providing coverage for VMAT2 inhibitors when medically indicated, though approval timelines and formulary restrictions vary across provinces creating access disparities. Major pharmaceutical companies including Neurocrine Biosciences, Teva Pharmaceutical Industries, and others maintain comprehensive commercial operations across North America with dedicated sales forces, medical science liaisons, and market access teams supporting product adoption. Patient advocacy organizations including Tardive Dyskinesia Awareness Network and Huntington's Disease Society of America conduct educational campaigns raising public awareness about movement disorders and available treatments, empowering patients to advocate for themselves during medical consultations. The region benefits from extensive clinical trial infrastructure enabling participation in pivotal studies and access to investigational therapies, positioning North American sites as leaders in VMAT2 inhibitor research and development. North America's market is projected to grow at approximately 9.86% CAGR from 2026 to 2033, sustained by aging populations developing higher psychiatric disorder rates, continued innovation in treatment options, expanding awareness among healthcare providers and patients, and evolving reimbursement policies balancing access with cost containment objectives.
Asia Pacific
Rapid Healthcare Modernization and Expanding Specialist Networks Position Region as Fastest-Growing Market
Asia Pacific represents the fastest-growing regional market for VMAT2 inhibitors, projected to achieve the highest CAGR during the forecast period from 2026 to 2033, driven by transformative healthcare infrastructure investments across populous nations and expanding recognition of neurological disorders requiring specialized pharmacological management. Countries including China, India, Japan, South Korea, and Southeast Asian nations are substantially increasing healthcare expenditures to meet the needs of their large populations, with governments prioritizing mental health and neurological care as public health imperatives deserving dedicated resources and policy attention. Economic development generates rising middle classes with increasing disposable incomes enabling out-of-pocket healthcare spending supplementing public insurance systems, creating demand for advanced pharmaceutical therapies including specialty medications like VMAT2 inhibitors. Urbanization concentrates populations in cities where major medical centers and specialty clinics establish movement disorder programs staffed by Western-trained neurologists familiar with contemporary diagnostic criteria and evidence-based treatment approaches. Medical tourism flourishes across Asia Pacific with countries like Thailand, India, and Singapore attracting international patients seeking high-quality neurological care at lower costs compared to Western markets, stimulating domestic market development.
Japan maintains a sophisticated pharmaceutical market with early adoption of innovative neurological therapies, stringent regulatory standards, and comprehensive national health insurance providing access to approved treatments including VMAT2 inhibitors for both tardive dyskinesia and Huntington's disease. India demonstrates rapid growth potential given its massive population exceeding 1.4 billion people, expanding private healthcare sector, growing awareness of movement disorders among the medical community, and domestic pharmaceutical industry capable of producing generic medications once patent protections expire. China's healthcare reforms aim to improve access to quality medical care across urban and rural regions, with government initiatives supporting specialty drug availability and expanding insurance coverage for previously unaffordable treatments. Pharmaceutical companies including global leaders and regional manufacturers are establishing commercial operations across Asia Pacific through partnerships with local distributors, direct sales infrastructure, and medical affairs teams conducting physician education. The combination of large patient populations, improving diagnostic capabilities, rising healthcare investments, expanding specialist networks, and growing awareness about treatment options positions Asia Pacific as the most dynamic growth region for VMAT2 inhibitors throughout the coming decade, with market penetration rates still low compared to developed markets indicating substantial untapped potential.
Top Key Players
-
Neurocrine Biosciences, Inc. (United States)
-
Teva Pharmaceutical Industries Ltd. (Israel)
-
Bausch Health Companies Inc. (Canada)
-
Lundbeck A/S (Denmark)
-
Sun Pharmaceutical Industries Ltd. (India)
-
Lupin Limited (India)
-
Hetero Drugs Limited (India)
-
Apotex Inc. (Canada)
-
Dr. Reddy's Laboratories Ltd. (India)
-
Amneal Pharmaceuticals LLC (United States)
-
Mylan N.V. (Viatris Inc.) (United States)
-
Aurobindo Pharma Limited (India)
-
Pfizer Inc. (United States)
-
Novartis AG (Switzerland)
-
AbbVie Inc. (United States)
Recent Developments
-
In January 2026, Neurocrine Biosciences announced the initiation of a Phase 2 clinical study for NBI-1065890 in adults with tardive dyskinesia, representing a randomized, double-blind, placebo-controlled trial enrolling approximately 100 participants to assess the efficacy, safety, and tolerability of this potentially longer-acting VMAT2 inhibitor over an eight-week treatment period, advancing the company's pipeline of next-generation therapies with improved pharmacological characteristics.
-
In March 2025, Neurocrine Biosciences launched a Phase 1 clinical study for NBI-1140675, a novel oral selective second-generation VMAT2 inhibitor being evaluated for safety, tolerability, pharmacokinetics, and pharmacodynamics in healthy adult participants, demonstrating the company's continued commitment to innovation and development of improved treatment options for patients with movement disorders requiring VMAT2 inhibition.
-
In February 2025, Neurocrine Biosciences reported fourth quarter and fiscal 2024 financial results highlighting INGREZZA achieving USD 615 million in Q4 sales and USD 2.3 billion for full-year 2024, representing year-over-year growth of 26 percent, while announcing 2025 sales forecast for INGREZZA estimated between USD 2.5 and 2.6 billion, demonstrating sustained market momentum and strong commercial execution.
-
In 2024, Teva Pharmaceutical Industries continued expanding its VMAT2 inhibitor portfolio through strategic investments in research and development, manufacturing capacity enhancements, and geographic market expansion initiatives aimed at increasing global availability of deutetrabenazine for patients with tardive dyskinesia and Huntington's disease chorea across diverse healthcare systems and reimbursement environments.
-
Throughout 2024, Bausch Health Companies maintained its focus on neurological and psychiatric portfolio development, advancing clinical programs, strengthening commercial infrastructure, and implementing patient access initiatives to ensure individuals requiring VMAT2 inhibitor therapy can obtain necessary treatments despite financial, administrative, and geographic barriers that historically limited specialty medication availability.
Market Trends
Shift Toward Personalized Dosing Strategies and Digital Health Integration Transform Treatment Paradigms
The VMAT2 inhibitors market is experiencing a pronounced trend toward personalized medicine approaches that optimize individual patient dosing based on treatment response monitoring, tolerability assessment, and pharmacogenomic profiling rather than relying solely on standardized dose escalation protocols used in clinical trials. Neurologists and psychiatrists increasingly recognize substantial inter-patient variability in VMAT2 inhibitor responses, with some individuals achieving satisfactory symptom control on lower doses while others require maximum approved dosages to obtain meaningful therapeutic benefits. Clinical practice emphasizes titration flexibility allowing providers to balance efficacy and tolerability for each patient's unique circumstances, starting at conservative initial doses and gradually increasing until optimal outcomes emerge or side effects become limiting. Genetic testing to identify cytochrome P450 enzyme polymorphisms affecting drug metabolism is beginning to inform VMAT2 inhibitor prescribing decisions, with poor metabolizers requiring dose reductions to avoid excessive drug accumulation while ultra-rapid metabolizers may need higher doses achieving therapeutic plasma concentrations. This trend toward precision medicine reflects broader healthcare evolution recognizing that standardized treatment protocols inadequately address individual biological diversity requiring personalized therapeutic strategies.
Digital health technologies including wearable sensors, smartphone applications, and telemedicine platforms are becoming increasingly integrated into VMAT2 inhibitor therapy management, fundamentally transforming how patients and providers monitor treatment responses and manage medication regimens. Movement disorder tracking applications enable patients to record symptom severity, medication timing, and side effect experiences, generating longitudinal data that informs treatment optimization discussions during clinic visits or telehealth consultations. Wearable accelerometers and gyroscopes objectively quantify involuntary movements providing data-driven assessments complementing subjective clinical rating scales, potentially enabling earlier detection of treatment responses or symptom progression. Telemedicine platforms expand access to movement disorder specialists for patients in rural or underserved regions lacking local expertise, facilitating VMAT2 inhibitor initiation and ongoing management without requiring extensive travel. Digital medication adherence tools including smart pill bottles and reminder applications address the persistent challenge of treatment non-adherence that undermines clinical outcomes, with studies demonstrating that approximately 40 to 50 percent of patients with chronic conditions fail to take medications as prescribed. These digital innovations create opportunities for pharmaceutical companies to differentiate their products through integrated technology platforms and support services enhancing patient experiences beyond the molecules themselves.
Segments Covered in the Report
By Drug Type
-
Valbenazine (Ingrezza)
-
Deutetrabenazine (Austedo)
-
Tetrabenazine (Xenazine)
-
Others
By Application
-
Tardive Dyskinesia
-
Huntington's Disease (Chorea)
-
Tourette Syndrome
-
Others
By Route of Administration
-
Oral (Tablets/Capsules)
-
Injectable Solutions
By Distribution Channel
-
Hospital Pharmacies
-
Retail Pharmacies
-
Online/E-Pharmacies
By Region
-
North America
-
United States
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
United Kingdom
-
France
-
Italy
-
Spain
-
Rest of Europe
-
-
Asia Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
Southeast Asia
-
Rest of Asia Pacific
-
-
Latin America
-
Brazil
-
Argentina
-
Rest of Latin America
-
-
Middle East and Africa
-
GCC Countries
-
South Africa
-
Rest of Middle East and Africa
-
Frequently Asked Questions
Question 1: What is the current VMAT2 inhibitors market size and projected growth rate?
Answer: The global VMAT2 inhibitors market is valued at USD 5.74 billion in 2025 and is expected to reach USD 14.69 billion by 2033, expanding at a CAGR of 9.14% from 2026 to 2033. This growth is driven by increasing prevalence of movement disorders and rising awareness of available treatments.
Question 2: Which region dominates the VMAT2 inhibitors market globally?
Answer: North America leads the VMAT2 inhibitors market with approximately 39.5% market share in 2025, attributed to advanced healthcare infrastructure and high diagnosis rates. Asia Pacific represents the fastest-growing region with the highest projected CAGR driven by expanding specialist networks and improving healthcare access.
Question 3: What drug type holds the largest share in the VMAT2 inhibitors market?
Answer: Valbenazine dominates the VMAT2 inhibitors market with 54.2% share in 2025 due to convenient once-daily dosing and first-mover advantage. Deutetrabenazine is projected to grow at the fastest rate driven by flexible dosing schedules and expanding clinical adoption across multiple movement disorders.
Question 4: Which application accounts for the largest VMAT2 inhibitors market share?
Answer: Tardive dyskinesia accounts for approximately 74.9% of the VMAT2 inhibitors market in 2025 due to substantial patient populations and strong clinical evidence. Huntington's disease segment is expected to grow significantly as awareness increases and access to specialty treatments improves globally.
Question 5: Who are the major companies operating in the VMAT2 inhibitors market?
Answer: Leading players in the VMAT2 inhibitors market include Neurocrine Biosciences Inc., Teva Pharmaceutical Industries Ltd., and Bausch Health Companies Inc. Other key companies include Lundbeck A/S, Sun Pharmaceutical Industries Ltd., Lupin Limited, Dr. Reddy's Laboratories, Amneal Pharmaceuticals, and Aurobindo Pharma Limited.
