UTI Drugs Market Overview
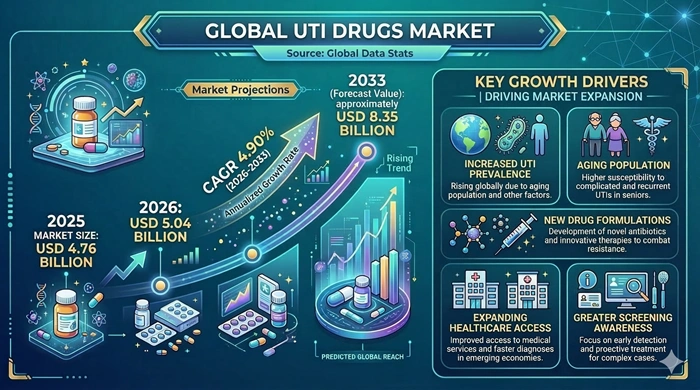
The global UTI Drugs market size is valued at USD 4.76 billion in 2025 and is predicted to increase from USD 5.04 billion in 2026 to approximately USD 8.35 billion by 2033, growing at a CAGR of 4.90% from 2026 to 2033.
Urinary tract infections are among the most prevalent bacterial infections globally, affecting women, older adults, and immunocompromised patients at exceptionally high rates and generating persistent, large-scale demand for antibiotic and alternative urinary anti-infective treatments. The UTI drugs market is expanding on the back of rising infection incidence, a rapidly aging global population, growing antibiotic resistance creating clinical need for newer drug agents, and steadily improving healthcare access in emerging economies. As pharmaceutical pipelines advance novel treatments and as awareness of recurrent UTI management deepens, this market continues to deliver consistent and broad-based growth.
AI Impact on the UTI Drugs Industry
Artificial Intelligence Is Transforming UTI Drug Discovery, Accelerating Antimicrobial Resistance Surveillance, and Enabling Precision Antibiotic Prescribing That Is Improving Patient Outcomes Across Clinical Settings Globally
Artificial intelligence is beginning to make a meaningful difference in the UTI Drugs market, most immediately in the area of diagnostic precision and targeted antibiotic selection. AI-powered clinical decision-support platforms can now analyze urine culture data, patient medical history, and local resistance patterns simultaneously to recommend the most appropriate UTI treatment for individual patients — significantly reducing the dependence on broad-spectrum empirical antibiotic prescribing that has historically driven resistance development. These platforms are being increasingly adopted by hospital systems and outpatient clinical networks in North America and Europe, where antibiotic stewardship programs are incorporating AI tools to improve prescribing appropriateness and reduce adverse resistance outcomes.
On the drug discovery side, machine learning models are dramatically compressing the preclinical research timeline for identifying novel antibacterial compounds with activity against resistant UTI pathogens. AI platforms can screen millions of molecular candidates in silico within hours — a process that previously required years of laboratory testing — and predict antimicrobial resistance emergence trajectories with growing accuracy. This predictive capability is helping pharmaceutical companies and public health bodies stay ahead of rapidly evolving uropathogen resistance profiles and prioritize development of new drug classes that address the most critical clinical gaps. The combined impact of AI on clinical practice and drug development is positioning the UTI pharmaceutical landscape for a period of meaningfully accelerated innovation.
Growth Factors
Rising UTI Burden in Aging and Vulnerable Populations, Escalating Antibiotic Resistance Driving Demand for New Drug Classes, and Expanding Healthcare Access in Emerging Markets Are the Three Core Drivers of UTI Drugs Market Growth
The most fundamental and enduring driver of the UTI Drugs market is the persistent and growing global burden of urinary tract infections. UTIs are the second most common bacterial infection presenting in clinical practice, with women disproportionately affected due to anatomical factors and older adults facing escalating infection risk due to reduced immune function, increased catheter use, and comorbidities such as diabetes and chronic kidney disease. As the global population ages and as the proportion of high-risk patient groups expands, the absolute volume of UTI episodes — and the corresponding demand for drug treatment — grows consistently year over year. This demographic momentum is the bedrock of the market's stable and sustained growth trajectory.
The rapidly intensifying global challenge of antibiotic resistance in UTI-causing pathogens is creating powerful additional demand for new-generation drug development. The growing prevalence of multidrug-resistant uropathogens — including ESBL-producing Enterobacteriaceae, fluoroquinolone-resistant E. coli, and carbapenem-resistant organisms in hospital settings — is rendering standard first-line UTI antibiotics ineffective in an increasing proportion of cases. This resistance-driven clinical need is motivating substantial pharmaceutical R&D investment in novel antibiotic agents, anti-virulence approaches, and combination therapies. As new drugs targeting resistant UTI pathogens progress through clinical pipelines and receive regulatory approval, they are expected to add meaningful premium-priced revenue to the overall market during the 2026–2033 forecast period.

Market Outlook
A Deepening Antibiotic Resistance Crisis, Maturing Novel Drug Pipelines, and Geographic Market Expansion in Asia Pacific and Latin America Collectively Ensure a Consistently Positive Long-Term Outlook for the UTI Drugs Market
The long-term outlook for the UTI Drugs market is steadily positive. The forecast period from 2026 to 2033 is expected to see the commercial launch of several important new UTI drug agents — including novel oral penem antibiotics, next-generation cephalosporins with ESBL-inhibitor combinations, and early non-antibiotic therapeutics — that will expand the treatment toolkit and introduce new premium-priced product revenues to the market. The FDA's approval of Orlynvah in 2025 as the first oral penem antibiotic for UTIs in over 25 years signals the beginning of a new drug approval cycle in the UTI space that is expected to accelerate through the forecast period as additional pipeline candidates mature.
Geographic market expansion in Asia Pacific, Latin America, and the Middle East is also a critical element of the market's forward trajectory. Improving healthcare access, rising antibiotic prescription rates, expanding private health insurance coverage, and growing pharmaceutical distribution infrastructure in these high-population regions are progressively enlarging the addressable patient population for UTI drug products. Increasing clinical awareness of UTI diagnosis and treatment in markets where historically underdiagnosis was common will further accelerate demand growth. Together, these dynamics ensure that the UTI Drugs market maintains a positive and multi-supported growth trajectory through 2033 and beyond.
Expert Speaks
-
Albert Bourla, CEO, Pfizer — "Antibiotic resistance in urinary tract infections is a growing public health challenge that demands ongoing pharmaceutical innovation. Pfizer remains committed to developing new antibacterial agents targeting resistant UTI pathogens, and we are investing in the science and manufacturing capabilities needed to bring meaningful new treatment options to patients worldwide."
-
Emma Walmsley, CEO, GSK — "The recent UK regulatory approval of gepotidacin — our first new mechanism antibiotic for UTIs — represents a significant milestone in addressing drug-resistant bacterial infections. We are proud to be at the forefront of developing new antibiotics that offer clinicians and patients effective alternatives when standard treatments fail, and we see a strong long-term commercial opportunity in this space."
-
Pascal Soriot, CEO, AstraZeneca — "Antimicrobial resistance is one of the defining healthcare challenges of our time, and UTIs sit at the heart of this crisis given their extraordinary global incidence. We view investment in new UTI therapeutics not just as a commercial opportunity but as a genuine public health obligation, and our pipeline reflects this strategic commitment."
Key Report Takeaways
-
North America dominates the UTI Drugs market, holding approximately 33.60% of global revenue share in 2025, supported by high UTI diagnosis and treatment rates, well-developed hospital and retail pharmacy distribution infrastructure, robust pharmaceutical R&D investment, and the presence of leading companies including Pfizer, Merck & Co., and AbbVie with established UTI drug portfolios.
-
Asia Pacific is the fastest-growing regional market for UTI drugs, driven by a massive and rapidly aging population with high UTI susceptibility, rapidly expanding healthcare coverage and infrastructure, rising clinical awareness of UTI diagnosis, and strong growth in domestic pharmaceutical manufacturing and distribution across China, India, and Southeast Asia.
-
Fluoroquinolones are the leading drug class, accounting for approximately 22.40% of total global market share in 2025, owing to their broad-spectrum antibacterial activity, excellent oral bioavailability, established clinical track record, and widespread physician familiarity — though growing resistance and prescribing restrictions are beginning to moderate their dominance in favor of alternative agents.
-
Carbapenems are the fastest-growing drug class, projected to expand at a CAGR of 6.20% through the forecast period, driven by their critical role as last-line agents against multidrug-resistant Gram-negative UTI pathogens in hospital settings — a demand that is growing as resistance to earlier antibiotic classes becomes more prevalent globally.
-
Uncomplicated UTI is the dominant indication segment, contributing approximately 68.60% of total market revenue in 2025, reflecting the far higher absolute patient volume of uncomplicated infections compared to complicated presentations, despite lower per-episode drug expenditure due to the widespread use of inexpensive generic oral antibiotics.
-
Online pharmacy is the fastest-growing distribution channel, projected to expand at a CAGR of 7.90% from 2026 to 2033, driven by the increasing prevalence of telehealth-integrated e-pharmacy platforms that allow UTI patients to receive prescriptions and medication home delivery seamlessly — particularly valued by the large population of women managing recurrent UTI episodes.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 8.35 Billion |
| Market Size by 2025 | USD 4.76 Billion |
| Market Size by 2026 | USD 5.04 Billion |
| Market Growth Rate (2026–2033) | CAGR of 4.90% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Segments Covered | Drug Class, Route of Administration, Indication Type, Distribution Channel |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Growing Global UTI Incidence in High-Risk Demographics, Mounting Antibiotic Resistance, and Expanding Pharmaceutical Access in Emerging Markets Are the Three Pillars Powering UTI Drugs Market Expansion
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising UTI incidence in aging and immunocompromised populations | ~34% | Global — especially North America, Europe, Asia Pacific | Short to Long-term |
| Escalating antibiotic resistance driving new drug class development | ~30% | Global — especially North America, Europe | Medium to Long-term |
| Expanding healthcare access and antibiotic use in emerging markets | ~24% | Asia Pacific, Latin America, MEA | Short to Medium-term |
| Growing recurrent UTI burden and demand for preventive therapies | ~12% | North America, Europe | Medium to Long-term |
The persistent and growing global burden of urinary tract infections continues to function as the most durable structural demand driver for the UTI Drugs market. As populations in North America, Europe, and Asia Pacific age, the proportion of elderly individuals — particularly those residing in long-term care facilities with frequent catheter use — who are at high risk for both primary and recurrent UTIs is expanding consistently. Rising rates of diabetes, obesity, and immunosuppressive therapy use are simultaneously expanding the high-risk patient demographic in all major geographies, creating a steadily growing base demand for UTI antibiotic and prophylactic drug products that is largely independent of cyclical market dynamics.
The rapidly escalating global challenge of antibiotic resistance is functioning simultaneously as a market headwind — by moderating consumption of older generic agents — and as a powerful growth driver, by generating urgent clinical and commercial demand for new drug development. As fluoroquinolones, trimethoprim-sulfamethoxazole, and first-generation cephalosporins become progressively less effective against prevalent UTI pathogens, clinicians are increasingly required to prescribe more expensive, second- and third-line antibiotic agents. This prescribing shift toward premium-priced novel agents is raising the average revenue per treated UTI episode and driving total market value growth at a pace that exceeds the underlying growth in raw prescription volumes.

Restraints Impact Analysis
Heavy Generic Competition Compressing UTI Drug Revenues, Antibiotic Stewardship Programs Moderating Prescription Volumes, and Safety Concerns Around Key Drug Classes Create Meaningful Market Headwinds
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Dominant generic drug penetration in standard UTI treatment segments | ~35% | Global — especially North America, Europe | Short to Medium-term |
| Antibiotic stewardship programs reducing unnecessary prescribing | ~28% | North America, Europe | Medium to Long-term |
| Regulatory restrictions and safety concerns limiting fluoroquinolone use | ~25% | North America, Europe | Short to Medium-term |
| Patient self-medication and antibiotic misuse worsening resistance | ~12% | Emerging markets in Asia Pacific, Latin America | Short-term |
The overwhelming dominance of generic antibiotics in treating uncomplicated UTI — the indication responsible for approximately 68.60% of total market volume — is the most significant structural revenue restraint in the UTI Drugs market. The vast majority of uncomplicated cystitis cases are treated with inexpensive generic nitrofurantoin, trimethoprim-sulfamethoxazole, or fosfomycin, keeping average per-episode drug expenditure low despite the enormous patient volume. This limits the total market revenue potential relative to the scale of UTI incidence and constrains the growth rate of the overall market even as the absolute number of treated UTI episodes continues to expand. Branded pharmaceutical companies must therefore focus their UTI investment on the more complex, higher-value complicated and recurrent indication segments where generic competition is less dominant.
Antibiotic stewardship programs — now mandated in hospital settings across most developed markets — are deliberately working to reduce empirical, broad-spectrum antibiotic prescribing, including for UTIs where more targeted or non-antibiotic management may be appropriate. While these programs are critically important for public health, they introduce a deliberate moderating force on antibiotic prescription volume growth in the highest-value markets. Regulatory restrictions on fluoroquinolone prescribing for uncomplicated UTI — implemented by the FDA and EMA in recent years due to safety concerns about serious adverse effects — are further reshaping prescribing patterns and moderating revenue from what was historically one of the market's highest-volume drug classes.
Opportunities Impact Analysis
Novel Non-Antibiotic UTI Therapeutics, UTI Vaccine Development, Telehealth-Driven Prescribing Expansion, and Rapid Pharmaceutical Growth in Emerging Markets Offer Substantial Untapped Growth Opportunities
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Development and commercialization of non-antibiotic UTI treatments | ~36% | North America, Europe | Medium to Long-term |
| UTI vaccine programs advancing through clinical development | ~28% | North America, Europe, Asia Pacific | Long-term |
| Telehealth and online pharmacy-driven UTI treatment access expansion | ~24% | North America, Asia Pacific, Europe | Short to Medium-term |
| Pharmaceutical infrastructure and market growth in Asia Pacific | ~12% | China, India, Southeast Asia | Short-term |
The development of non-antibiotic approaches to UTI treatment and prevention represents the most commercially significant long-term opportunity in the UTI Drugs market. Bacteriophage therapies, anti-virulence agents targeting uropathogen adhesion and toxin production, D-mannose and cranberry-based standardized clinical products, and immunostimulating compounds are all gaining scientific and clinical momentum as alternatives or adjuncts to conventional antibiotic therapy. If late-stage clinical trials validate their safety and efficacy, these approaches would enter a large and established prescription market while commanding significant pricing premiums over generic antibiotics — creating a substantial new revenue segment that could meaningfully accelerate overall market growth during the later portion of the forecast period.
The rapid expansion of telehealth services and online pharmacy platforms is simultaneously creating a significant structural opportunity to expand UTI drug market reach and per-episode revenue. Telehealth-integrated e-pharmacy models — where patients can consult a clinician, receive a UTI diagnosis and prescription, and have medication delivered within hours without an in-person visit — are proving particularly popular among the large population of women managing recurrent UTI episodes. This convenience-driven model is increasing treatment-seeking rates and prescription adherence, expanding the commercially addressable UTI patient population and driving the online pharmacy distribution channel to become one of the market's fastest-growing segments during the 2026–2033 forecast period.

Segment Analysis
By Drug Class
Fluoroquinolones Lead the UTI Drugs Market by Revenue Share While Carbapenems Are Rapidly Emerging as the Highest-Growth Drug Class Driven by Multidrug-Resistant Pathogen Prevalence
Fluoroquinolones hold the dominant position within the drug class segment, accounting for approximately 22.40% of total UTI Drugs market revenue in 2025. Their market leadership reflects decades of widespread clinical use as broad-spectrum, orally bioavailable agents with excellent urinary tissue penetration and established efficacy against the most common UTI pathogens, including E. coli and Klebsiella pneumoniae. However, growing evidence of resistance among uropathogenic E. coli isolates and regulatory restrictions on fluoroquinolone prescribing for uncomplicated UTI in the U.S. and EU are progressively shifting clinical practice away from fluoroquinolones toward alternative first-line agents, moderating their share growth trajectory. North America holds the largest fluoroquinolone UTI drug revenue, though guideline-directed prescribing transitions are most advanced here, and major generic manufacturers — including Teva Pharmaceutical Industries and Mylan — dominate commercial volume in this high-generic-penetration drug class. The segment remains the revenue leader due to the accumulated base of complicated UTI and pyelonephritis prescriptions where fluoroquinolones remain clinically appropriate in susceptible isolate cases.
Carbapenems represent the fastest-growing drug class in the UTI Drugs market, projected to expand at a CAGR of 6.20% from 2026 to 2033 as they serve as the essential last-line defense against multidrug-resistant Gram-negative uropathogens — including carbapenem-sensitive ESBL-producing organisms and KPC-producing Klebsiella — in hospital and intensive care settings. Their growth is directly correlated with the rising prevalence of MDR infections in hospital-acquired UTIs and catheter-associated UTIs, which are increasing as antibiotic resistance progresses globally. Europe is a key growth region for carbapenem UTI drug usage, where hospital antibiotic stewardship programs that restrict carbapenem use to confirmed resistant infections are paradoxically driving more intensive and high-volume use in exactly the cases where they are genuinely needed. Key companies with carbapenem UTI portfolios include Merck & Co. (imipenem-cilastatin-relebactam), Pfizer, and AstraZeneca, all of whom are investing in next-generation carbapenem combinations designed to extend efficacy against increasingly resistant organisms.
By Distribution Channel
Hospital Pharmacy Leads UTI Drug Distribution by Revenue While Online Pharmacy Is the Undisputed Fastest-Growing Channel Driven by Telehealth Integration and Patient Convenience Demand
Hospital pharmacy is the dominant distribution channel in the UTI Drugs market, accounting for approximately 39.60% of total market revenue share in 2025. Hospital pharmacies command the largest revenue share because they dispense the highest-value drug regimens — including intravenous carbapenems, novel beta-lactam combinations, and expensive branded agents — used to treat complicated UTIs, catheter-associated UTIs, and pyelonephritis cases requiring inpatient care. These high-acuity cases generate substantially greater per-episode pharmaceutical expenditure than community-treated uncomplicated UTIs, making hospital pharmacy the highest-value distribution channel despite not necessarily handling the highest absolute volume of prescriptions. North America is the dominant region for hospital pharmacy UTI drug revenue, where large academic medical centers and specialty infectious disease programs manage the most complex and expensive UTI cases. Companies such as Merck & Co., Pfizer, and AstraZeneca rely heavily on hospital pharmacy channels to distribute their carbapenem and novel antibiotic products through group purchasing organizations and hospital formulary inclusion programs.
Online pharmacy is the fastest-growing distribution channel in the UTI Drugs market, projected to expand at a CAGR of 7.90% from 2026 to 2033 — the highest of any distribution channel. This extraordinary growth is driven by the accelerating adoption of telehealth-integrated e-pharmacy platforms that allow UTI patients — particularly women managing frequent recurrent infections — to receive clinical consultations, UTI diagnoses, and antibiotic prescriptions with same-day home medication delivery, eliminating the friction and inconvenience of in-person pharmacy visits. North America is the leading region for online pharmacy UTI drug growth, where major digital health platforms including Teladoc, Amazon Pharmacy, and numerous dedicated women's health telehealth services are building large and loyal patient bases for convenient UTI management. Asia Pacific is also emerging as a rapidly growing online pharmacy UTI drug market, driven by the exceptional mobile internet penetration, growing comfort with digital health services, and rapid expansion of licensed e-pharmacy platforms in China, India, and Southeast Asia.

Regional Insights
North America
North America Leads the UTI Drugs Market With the Largest Revenue Share, Strongest Pharmaceutical R&D Base, and Most Advanced Clinical Infrastructure for Antibiotic Resistance Management
North America holds approximately 33.60% of global UTI Drugs market revenue in 2025 and is projected to maintain its regional leadership at a CAGR of approximately 5.63% from 2026 to 2033. The United States is the primary revenue driver within the region, where the highest per-capita healthcare spending, a large and aging UTI-prone patient population, well-developed hospital and community pharmacy infrastructure, and the concentration of major pharmaceutical companies with UTI drug portfolios and active R&D programs collectively support the region's market dominance. The U.S. alone accounts for approximately USD 1.28 billion of the North American market in 2025, driven by high UTI prescription volumes across both inpatient and outpatient settings. Key companies with strong North American UTI drug operations include Pfizer, Merck & Co., AbbVie, GlaxoSmithKline, and Iterum Therapeutics — the latter having received FDA approval for Orlynvah, the first new branded uncomplicated UTI drug treatment in over 25 years, in August 2025.
Canada contributes meaningfully to the North American market, with consistent government healthcare system procurement of both branded and generic UTI antibiotics through provincial formularies and hospital purchasing programs. The strong presence of federal and provincial antibiotic stewardship initiatives mirrors U.S. trends, reshaping UTI prescribing patterns toward more targeted, guideline-aligned antibiotic selection. The region's advanced tele health and online pharmacy infrastructure is simultaneously accelerating prescription convenience and expanding the addressable UTI patient population in both the U.S. and Canada, reinforcing North America's position as the most commercially mature and highest-value regional market for UTI drugs through 2033.
Asia Pacific
Asia Pacific Is the Fastest-Growing UTI Drugs Market Globally, Powered by a Massive Aging Population, Rapidly Expanding Healthcare Coverage, and Surging Domestic Pharmaceutical Manufacturing Capacity
Asia Pacific is the fastest-growing regional segment in the UTI Drugs market, projected to expand at a CAGR of approximately 6.80% from 2026 to 2033 — the highest of any region globally. China and India are the two largest and most rapidly growing national markets within the region, each with enormous and rapidly aging populations carrying high absolute UTI burdens, expanding government-led universal health coverage programs that are bringing antibiotic treatment within financial reach of larger patient populations, and thriving domestic pharmaceutical industries producing affordable generic UTI drug products for both domestic consumption and global export. Key multinational companies with active Asia Pacific UTI drug operations include Pfizer, Bayer AG, Abbott Laboratories, and GlaxoSmithKline, alongside major domestic players such as Sun Pharmaceutical Industries (India), Cipla Limited (India), and CSPC Pharmaceutical Group (China) — the latter group increasingly capturing market share from multinationals through competitive generic pricing and strong regional distribution networks.
Japan and South Korea represent the most mature and highest-value UTI drug sub-markets within Asia Pacific, where aging populations, well-developed healthcare systems, and high antibiotic prescription standards drive strong per-capita UTI drug revenue. Japan's aging demographic — one of the oldest nationally in the world — ensures consistently growing UTI incidence in elderly and long-term care patient segments, sustaining predictable demand for both standard and newer antibiotic agents. The combination of enormous and growing patient volume in developing Asian economies and strong per-episode drug revenue in mature Asian markets makes Asia Pacific the most strategically significant long-term growth region for UTI Drugs market participants investing in geographic expansion through 2033 and beyond.
Top Key Players
-
Pfizer Inc. (United States)
-
Merck & Co., Inc. (United States)
-
Bayer AG (Germany)
-
GlaxoSmithKline plc (United Kingdom)
-
AstraZeneca plc (United Kingdom)
-
AbbVie Inc. (United States)
-
Johnson & Johnson (United States)
-
Sun Pharmaceutical Industries Ltd. (India)
-
Cipla Limited (India)
-
Teva Pharmaceutical Industries Ltd. (Israel)
-
CSPC Pharmaceutical Group Limited (China)
-
Lupin Limited (India)
-
Iterum Therapeutics plc (Ireland)
Recent Developments
-
Iterum Therapeutics (2025) — Received FDA approval in August 2025 for Orlynvah (sulopenem etzadroxil and probenecid), marking a landmark regulatory milestone as the first new oral antibiotic approved specifically for uncomplicated UTIs in over 25 years. This approval represents a major commercial breakthrough in the UTI Drugs space, offering a critically needed new treatment option for uncomplicated UTIs caused by designated susceptible microorganisms in adult women.
-
GlaxoSmithKline (2025) — Secured regulatory approval in the United Kingdom for gepotidacin (Blujepa), a first-in-class triazaacenaphthylene antibiotic with a novel dual-targeting mechanism of action, approved for the treatment of uncomplicated UTIs caused by susceptible organisms. This represents GSK's first new class antibiotic launch in decades and a significant competitive entry into the growing market for novel UTI drug agents addressing antibiotic resistance.
-
Pfizer Inc. (2024) — Announced a strategic licensing and co-development agreement with a European biotech company advancing a novel non-antibiotic anti-virulence agent for recurrent UTI prevention, expanding Pfizer's UTI pipeline beyond conventional antibiotics and positioning the company for long-term participation in the growing market for resistance-bypassing UTI therapeutic approaches.
-
Merck & Co. (2025) — Advanced its novel beta-lactam/beta-lactamase inhibitor combination candidate targeting ESBL-producing and carbapenem-resistant uropathogens into a pivotal Phase III clinical trial, with FDA Qualified Infectious Disease Product designation confirmed. A successful trial outcome could result in one of the most clinically significant new UTI drug approvals of the decade for complicated UTI treatment.
-
Sun Pharmaceutical Industries (2024) — Expanded its UTI antibiotic portfolio across key Asia Pacific and African markets through the launch of new branded generic formulations of fosfomycin and pivmecillinam — two agents increasingly recommended by international guidelines as preferred alternatives to fluoroquinolones for uncomplicated UTI — capitalizing on the global prescribing trend shift away from fluoroquinolones.
Market Trends
The Approval of Novel Mechanism UTI Antibiotics and the Growing Clinical Adoption of Non-Antibiotic UTI Management Are Defining a New Era of Treatment Innovation in the UTI Drugs Market
The most consequential recent development reshaping the UTI Drugs market is the emergence and early commercialization of new-mechanism antibiotic agents for UTI treatment after decades of limited innovation in first-line treatment options. The FDA approval of Orlynvah in 2025 and the UK approval of gepotidacin mark the beginning of a new drug approval cycle in UTI therapeutics that is expected to accelerate significantly during the 2026–2033 forecast period as additional late-stage candidates advance to regulatory submission. These new agents offer clinicians genuinely effective treatment options for uncomplicated UTIs caused by organisms that are increasingly resistant to fluoroquinolones and trimethoprim-sulfamethoxazole, addressing a critical gap in the UTI treatment toolkit and commanding substantial pricing premiums over generic standard-of-care antibiotics. The commercial success of these early new-mechanism approvals is incentivizing further pharmaceutical investment in UTI drug development, creating a positive cycle of innovation and market growth.
The second defining trend is the accelerating clinical and commercial momentum behind non-antibiotic approaches to UTI prevention and treatment, particularly for the large and underserved recurrent UTI patient population. Standardized D-mannose formulations, clinically validated cranberry proanthocyanidin extracts, intravesical hyaluronic acid and chondroitin sulfate preparations, and immunostimulating polybacterial lysate products are gaining growing evidence bases and increasing physician endorsement as adjuncts or alternatives to antibiotic suppressive therapy in recurrent UTI management. These non-antibiotic modalities are attracting significant pharmaceutical and nutraceutical investment, and their progressive integration into clinical guidelines for recurrent UTI management is expected to create a meaningful new commercial sub-segment within the broader UTI Drugs market throughout the forecast period.
Segments Covered in the Report
By Drug Class:
-
Fluoroquinolones
-
Cephalosporins
-
Nitrofurantoin
-
Trimethoprim-Sulfamethoxazole
-
Fosfomycin
-
Penicillins
-
Carbapenems
-
Aminoglycosides
By Route of Administration:
-
Oral
-
Intravenous
-
Intramuscular
By Indication Type:
-
Uncomplicated UTI
-
Complicated UTI
-
Pyelonephritis
-
Recurrent UTI Prophylaxis
By Distribution Channel:
-
Hospital Pharmacy
-
Retail Pharmacy
-
Online Pharmacy
-
Clinic Dispensing
By Region:
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the UTI Drugs market and what is its forecast value by 2033?
Answer: The UTI Drugs market was valued at USD 4.76 billion in 2025 and is projected to reach USD 8.35 billion by 2033. It is expected to grow at a CAGR of 4.90% from 2026 to 2033, driven by rising UTI incidence, antibiotic resistance, and novel drug approvals.
Question 2: What are the primary growth drivers of the UTI Drugs market?
Answer: Rising UTI burden in aging and immunocompromised populations and escalating antibiotic resistance creating demand for new-generation drug classes are the two most powerful drivers. Expanding healthcare access in Asia Pacific and Latin America is also meaningfully broadening the addressable patient base for UTI drug products globally.
Question 3: Which region leads the UTI Drugs market and which is the fastest growing?
Answer: North America holds the dominant position in the UTI Drugs market with approximately 33.60% of global revenue in 2025. Asia Pacific is the fastest-growing region, expanding at approximately 6.80% CAGR from 2026 to 2033, driven by a massive aging population and rapidly improving healthcare and pharmacy infrastructure.
Question 4: Which drug class and distribution channel are leading in the UTI Drugs market?
Answer: Fluoroquinolones are the leading drug class, contributing approximately 22.40% of total UTI Drugs market revenue in 2025, while carbapenems are the fastest-growing class. Hospital pharmacy leads the distribution channel segment, while online pharmacy is the fastest-growing channel at a CAGR of 7.90% through 2033.
Question 5: What recent approvals and innovations are shaping the future of the UTI Drugs market?
Answer: The 2025 FDA approval of Orlynvah and the UK approval of gepotidacin mark a significant new drug innovation cycle in the UTI Drugs market after decades of limited first-line treatment options. Growing clinical adoption of non-antibiotic UTI prevention approaches — including anti-virulence agents, bacteriophage therapies, and standardized D-mannose formulations — is simultaneously expanding the market's treatment paradigm beyond conventional antibiotics.
