TUDS Test Market Overview
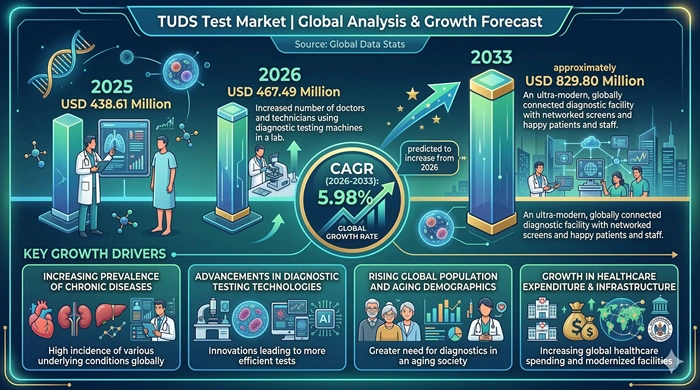
The global TUDS Test market size is valued at USD 438.61 million in 2025 and is predicted to increase from USD 467.49 million in 2026 to approximately USD 829.80 million by 2033, growing at a CAGR of 5.98% from 2026 to 2033.
The TUDS test — the tuberculin unit dosage skin test, widely administered as the intradermal Mantoux test — remains one of the most broadly used and clinically essential tools for tuberculosis screening and diagnosis globally. Rising global TB burden, expanding national TB control programs, and growing adoption of occupational health screening protocols are collectively sustaining consistent demand for tuberculin testing products. As healthcare infrastructure strengthens across emerging economies and as governments invest more aggressively in TB elimination programs, the TUDS test market is positioned for stable and sustained growth through 2033.
AI Impact on the TUDS Test Industry
Artificial Intelligence Is Beginning to Transform Tuberculin Skin Test Interpretation, TB Surveillance Data Analysis, and Screening Program Optimization in Ways That Are Expanding the Clinical Value and Reach of TUDS Testing Globally
Artificial intelligence is creating meaningful improvements in how tuberculin skin test results are interpreted and integrated into broader TB surveillance systems. AI-powered image analysis tools are now being developed and validated to assess the induration size from Mantoux test reactions using smartphone cameras, enabling more objective, reproducible measurements compared to the traditional manual caliper-based reading — a method that is known to carry significant inter-reader variability. These digital reading platforms are being piloted in high-burden TB countries across Asia and Africa, where they have the potential to significantly improve the reliability and scalability of mass tuberculin screening programs.
On the public health side, AI-driven epidemiological modeling tools are helping national TB programs analyze tuberculin test data at population scale — identifying geographic hotspots, high-risk demographic clusters, and latent TB infection prevalence trends with far greater speed and granularity than traditional surveillance methods allow. These tools are enabling more targeted and cost-effective deployment of TUDS test screening resources, ensuring that limited public health budgets are directed toward the populations and settings where tuberculin testing will deliver the greatest epidemiological impact. The convergence of AI with tuberculin diagnostics is gradually elevating the TUDS test from a simple clinical procedure to a data-rich component of intelligent, adaptive TB control systems.
Growth Factors
High Global TB Burden, Expanding Government TB Screening Programs, and Growing Occupational Health Testing Requirements Are the Core Forces Driving Consistent TUDS Test Market Growth
The primary growth driver of the TUDS Test market is the persistent and devastating global burden of tuberculosis, which the WHO continues to rank among the world's top infectious disease killers. With an estimated 10 million new TB cases occurring annually worldwide, the clinical and public health demand for reliable, accessible, and low-cost tuberculin screening tools remains extremely strong. The Mantoux tuberculin skin test — the principal TUDS test procedure — continues to be a cornerstone of TB screening in resource-limited settings due to its well-established methodology, low per-test cost, and the absence of the infrastructure requirements that more complex molecular diagnostic tools demand. This combination of clinical necessity and economic accessibility ensures robust baseline demand across all major TB-burden geographies.
Government-led national TB control and elimination programs are simultaneously providing an important structural demand driver for TUDS testing products. Governments across high-burden TB countries in Asia Pacific, Africa, and Latin America — supported by international health organizations including the WHO, STOP-TB Partnership, and the Global Fund — are scaling up latent TB infection screening programs to meet WHO End TB Strategy targets. These programs generate large-volume, government-funded procurement of tuberculin testing products, creating a stable and growing institutional demand channel that complements the more variable private clinical sector. Additionally, growing mandatory occupational health screening requirements for TB in healthcare workers, correctional facility staff, and teachers in multiple countries are expanding the addressable market for TUDS test products beyond traditional clinical diagnosis settings.

Market Outlook
With TB Elimination Goals Driving Unprecedented Government Investment in Latent TB Screening and Emerging Markets Rapidly Scaling Healthcare Infrastructure, the TUDS Test Market Is Set for Consistent Long-Term Expansion
The long-term outlook for the TUDS Test market is positive and well-supported by multiple structural demand drivers. The forecast period from 2026 to 2033 is expected to be shaped by the progressive scaling of national latent TB infection screening programs in high-burden countries — a key pillar of the WHO End TB Strategy — which will generate substantial and growing procurement volumes for tuberculin skin test products. As governments in Asia Pacific, Latin America, and sub-Saharan Africa increasingly prioritize TB control as a public health and economic development imperative, the institutional demand base for TUDS test products will expand meaningfully across regions that currently have the highest unmet TB diagnostic need.
The market will also benefit from the ongoing expansion of occupational health screening in both developed and developing economies, where TB testing requirements for healthcare workers, food service employees, and education sector staff are becoming more systematically enforced. While newer blood-based TB diagnostics such as interferon-gamma release assays (IGRAs) present a degree of competitive substitution pressure in well-resourced clinical settings, the TUDS test will continue to hold its position as the preferred and most accessible first-line TB screening tool in the vast majority of global healthcare settings through 2033, supported by its lower cost, established clinical infrastructure, and strong familiarity among healthcare providers worldwide.
Expert Speaks
-
Tedros Adhanom Ghebreyesus, Director-General, WHO (public sector) — "Tuberculosis remains one of the most urgent infectious disease challenges of our time, and scaling up testing — including accessible skin testing methods — is central to our End TB Strategy. Every investment in expanding TB screening infrastructure brings us closer to the goal of eliminating this disease as a public health threat."
-
Albert Bourla, CEO, Pfizer — "Infectious disease diagnostics, including tuberculosis skin testing, remain a critical part of the global health ecosystem. Ensuring that reliable, affordable TB screening tools remain available at scale in high-burden countries is not just a humanitarian priority — it is a fundamental component of global infectious disease preparedness."
-
Emma Walmsley, CEO, GSK — "Tuberculosis disproportionately affects the world's most vulnerable populations, and accessible, reliable screening tools are indispensable to controlling its spread. The pharmaceutical and diagnostics industries have a responsibility to maintain and improve the supply of high-quality TB testing products for the populations that need them most."
Key Report Takeaways
-
North America dominates the TUDS Test market, holding the largest regional revenue share in 2025, driven by well-established TB screening mandates for healthcare workers, correctional facility staff, and school employees, a highly developed hospital and diagnostic laboratory infrastructure, and the presence of leading tuberculin product manufacturers and regulatory-compliant supply chains in the U.S. and Canada.
-
Asia Pacific is the fastest-growing regional market for TUDS testing, driven by the region's extremely high TB prevalence — which accounts for the majority of global TB cases — combined with rapidly expanding national TB control programs, improving healthcare infrastructure, strong government and international funding support for TB screening, and large unmet demand for latent TB diagnosis tools across China, India, Southeast Asia, and the Western Pacific.
-
Hospitals are the dominant end-user segment, accounting for the largest market share in 2025, as hospitals represent the primary settings for TB diagnosis, treatment initiation, and healthcare worker TB screening — generating the highest consistent volumes of tuberculin skin test administration across both inpatient and outpatient clinical environments.
-
The intradermal tuberculin skin test (Mantoux test) is the leading application segment, contributing the largest revenue share in 2025 by a wide margin, reflecting its status as the globally standardized and most widely used method for tuberculin skin testing in routine clinical diagnosis, contact investigation, and TB screening programs.
-
The 5 TU (tuberculin unit) strength is the dominant dosage segment, holding the largest market share in 2025 as it represents the globally standardized, guideline-recommended dose for routine Mantoux testing in the vast majority of national TB screening protocols and clinical settings worldwide.
-
Epidemiological screening programs are the fastest-growing application segment, projected to expand at the highest CAGR through 2033 as governments and international health organizations dramatically scale up population-level latent TB infection screening programs targeting high-risk groups — including contacts of active TB cases, migrants, prisoners, and healthcare workers — in alignment with WHO End TB Strategy milestones.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 829.80 Million |
| Market Size by 2025 | USD 438.61 Million |
| Market Size by 2026 | USD 467.49 Million |
| Market Growth Rate (2026–2033) | CAGR of 5.98% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Segments Covered | Strength/Dosage, Application, End User, Distribution Channel |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
High Global TB Incidence, Expanding Latent TB Screening Mandates, and Growing Occupational Health Testing Requirements Are Collectively Sustaining Strong and Broad-Based TUDS Test Market Demand
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High global TB burden and WHO End TB Strategy screening targets | ~35% | Global — especially Asia Pacific, Africa, Latin America | Short to Long-term |
| Government-funded national TB control and screening programs | ~30% | Asia Pacific, Latin America, MEA | Short to Medium-term |
| Expanding occupational health TB screening mandates | ~22% | North America, Europe | Short to Medium-term |
| Growing latent TB infection diagnosis in immunocompromised populations | ~13% | Global — especially North America, Europe | Medium to Long-term |
The persistent global burden of tuberculosis — with approximately 10 million new cases annually and an estimated 1.7 billion people carrying latent TB infection worldwide — creates an enormous and structurally stable demand foundation for the TUDS test market. Public health systems in high-burden countries must maintain large-scale tuberculin skin testing programs to identify and treat latent TB infections before they progress to active disease, generating consistent and high-volume institutional procurement of tuberculin testing products. This demand is particularly strong in Asia Pacific and sub-Saharan Africa, where TB prevalence is highest and where national TB control programs are under increasing pressure from international health bodies to demonstrate progress toward WHO End TB Strategy targets.
Government investment in TB control is simultaneously functioning as a direct procurement driver that is both stable and growing. National TB programs funded through government health budgets and supplemented by international financing mechanisms — including the Global Fund to Fight AIDS, Tuberculosis and Malaria and USAID — regularly procure tuberculin testing products in very large volumes for population-level screening campaigns. These institutional procurement channels provide tuberculin product manufacturers with reliable, forward-contracted demand that is less subject to the variability of private clinical sector purchasing. The expansion of these government-funded programs in middle-income Asian and African countries during the 2026–2033 forecast period is expected to be one of the most significant demand growth drivers for the TUDS test market.

Restraints Impact Analysis
Competition From IGRA-Based Blood Tests, Cold-Chain Logistics Challenges in Remote Markets, and Two-Visit Protocol Requirements Limit the Speed of TUDS Test Market Penetration
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Competitive substitution pressure from IGRA-based TB diagnostics | ~35% | North America, Europe, developed Asia Pacific | Short to Medium-term |
| Cold-chain storage and transport requirements for tuberculin products | ~28% | Latin America, MEA, parts of Asia Pacific | Short to Medium-term |
| Two-visit protocol limiting patient compliance and program efficiency | ~25% | Global — especially in mobile or transient populations | Short-term |
| False-positive results in BCG-vaccinated populations reducing utility | ~12% | Asia Pacific, Latin America, MEA | Medium-term |
The most significant competitive restraint facing the TUDS Test market is the growing clinical adoption of interferon-gamma release assay (IGRA) blood tests — including QuantiFERON-TB Gold and T-SPOT.TB — as preferred alternatives to tuberculin skin testing in high-income markets. IGRAs offer several clinical advantages over the Mantoux TUDS test, including the elimination of BCG vaccine-related false positives, a single-visit blood draw protocol, and better specificity in certain patient populations. In North America and Western Europe, updated national TB screening guidelines are increasingly recommending or preferring IGRAs over TST in healthcare worker screening and immunocompromised patient testing contexts, which is moderating TUDS test volume growth in these high-value markets.
The requirement for patients to return for a second clinical visit 48–72 hours after tuberculin injection to have the test read represents a practical barrier to program efficiency and patient compliance, particularly in transient, mobile, or socioeconomically disadvantaged populations that are often at the highest TB risk. Missed readings result in wasted reagent, inconclusive screening outcomes, and additional healthcare resource consumption. In resource-limited settings, cold-chain requirements for tuberculin product storage and transport also present logistical challenges that can compromise product quality and increase the per-test operational cost, constraining market penetration in the lowest-income geographies where TB burden is often highest.
Opportunities Impact Analysis
Expanding Latent TB Infection Screening in High-Risk Populations, WHO End TB Strategy Milestones, and Growing Healthcare Infrastructure in High-Burden Emerging Markets Present Significant Untapped Growth Opportunities
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Scaling latent TB infection screening in high-risk groups | ~36% | Global — especially Asia Pacific, Latin America, MEA | Short to Medium-term |
| WHO End TB Strategy 2030 targets driving mass screening investment | ~28% | Asia Pacific, Africa, Latin America | Short to Medium-term |
| Healthcare infrastructure expansion in high-burden emerging markets | ~24% | India, China, Southeast Asia, Sub-Saharan Africa | Medium-term |
| Digital tuberculin test reading platforms improving program scalability | ~12% | Global — piloting in Asia Pacific, Africa | Medium to Long-term |
The WHO End TB Strategy's ambitious targets — including a 90% reduction in TB incidence and a 95% reduction in TB deaths by 2030 compared to 2015 baselines — require dramatic scale-up of latent TB infection screening and treatment globally. Achieving these targets necessitates extensive tuberculin skin testing programs across high-burden countries, creating a powerful policy-driven demand mandate for TUDS test products that is backed by international funding commitments. Countries including India, Indonesia, the Philippines, and South Africa — which collectively account for a substantial share of global TB burden — are scaling up national latent TB infection programs specifically, generating significant new procurement volumes for tuberculin testing products during the 2026–2033 forecast period.
The development and validation of digital tuberculin test reading platforms — which use smartphone-based image analysis to objectively measure Mantoux test induration without the traditional in-person second visit — represents a potentially transformative opportunity to overcome one of the TUDS test's most significant practical limitations. If these platforms achieve regulatory validation and widespread deployment, they could substantially increase the efficiency, scalability, and patient compliance of tuberculin skin testing programs, expanding the TUDS test market's reach into community-based and mobile screening contexts that are currently underserved by the traditional two-visit clinical model.

Segment Analysis
By Strength/Dosage
The 5 TU Dosage Segment Commands Global Market Leadership as the Universally Standardized Mantoux Testing Dose, While the 10 TU Segment Emerges as the Fastest-Growing Alternative for Targeted Clinical Applications
The 5 TU (tuberculin unit) dosage segment holds dominant leadership within the strength/dosage segmentation of the TUDS Test market, accounting for the largest share of global revenue in 2025. The 5 TU dose is the globally standardized, guideline-recommended administration for routine intradermal Mantoux tuberculin skin testing, endorsed by the WHO, the CDC, and national TB control programs in the vast majority of countries. Its dominance is self-reinforcing — decades of clinical protocol development, healthcare professional training, and supply chain infrastructure have all been built around the 5 TU dose as the default standard for TB screening. North America holds the largest revenue contribution in this dosage segment, where CDC-aligned clinical guidelines consistently specify 5 TU/0.1 mL PPD as the standard dose for healthcare worker screening, contact investigations, and routine TB diagnostic testing. Major suppliers of 5 TU tuberculin products include Sanofi (through its Tubersol product), Aplisol by Par Pharmaceutical, and several international producers supplying WHO-prequalified tuberculin for use in national programs across Asia and Africa.
The 10 TU dosage segment is expected to record the fastest growth rate within the strength/dosage category of the TUDS test market during the 2026–2033 forecast period, driven by its emerging clinical application in specific scenarios where higher immunogenicity sensitivity is required. The 10 TU dose is increasingly being evaluated in clinical research and public health programs for use in immunocompromised patients — including those with HIV infection, malnutrition, or immunosuppressive therapy — where the standard 5 TU dose may fail to elicit a measurable skin reaction despite genuine latent TB infection. Asia Pacific is a key region for the growth of 10 TU tuberculin testing, particularly in countries with high HIV-TB co-infection rates such as India, South Africa (Middle East & Africa), and Southeast Asian nations where the dual burden of HIV and TB creates significant demand for enhanced-sensitivity tuberculin testing protocols.
By Application
The Intradermal Mantoux Test Remains the Anchor Application in the TUDS Test Market, While Epidemiological Screening Programs Drive the Highest Growth as Governments Scale Up Latent TB Interventions
The intradermal tuberculin skin test — universally known as the Mantoux test — is the dominant application segment in the TUDS Test market, contributing the largest revenue share in 2025 by a substantial margin. The Mantoux test's clinical dominance reflects its status as the globally established standard procedure for tuberculin skin testing, with a methodology that has been refined and validated over more than a century of clinical and public health application. It remains the first-line TB screening tool in the vast majority of the world's healthcare systems, from highly resourced academic medical centers in North America to primary health posts in remote rural settings across Africa and Asia. North America is the largest regional contributor to Mantoux test revenue, where stringent TB screening requirements for healthcare workers, correctional facility employees, and school staff generate high and consistent annual testing volumes. Companies such as Sanofi (Tubersol) and Par Pharmaceutical (Aplisol) are the dominant suppliers of Mantoux tuberculin testing products in the North American market, benefiting from long-standing regulatory approvals and strong institutional procurement relationships.
Epidemiological screening programs represent the fastest-growing application segment in the TUDS test market, driven by the unprecedented scale of government and international health organization investment in population-level latent TB infection detection as a core strategy for achieving TB elimination goals. These programs systematically test large, defined population groups — including contacts of active TB cases, immigrants from high-burden countries, residents and staff of congregate settings such as prisons and homeless shelters, and healthcare workers — generating very large, government-funded tuberculin testing volumes. Asia Pacific is the fastest-growing region for epidemiological TB screening program activity, where governments including India, China, and the Philippines are implementing national latent TB infection screening strategies aligned with WHO End TB targets. International organizations supplying tuberculin products for large-scale epidemiological programs include Serum Institute of India, as well as several European manufacturers whose WHO-prequalified tuberculin products are procured through international health program tenders.

Regional Insights
North America
North America Leads the TUDS Test Market With the Strongest Institutional Screening Infrastructure, Robust Occupational Health TB Testing Mandates, and the Largest Concentration of Regulatory-Approved Tuberculin Product Suppliers
North America holds the dominant position in the global TUDS Test market in 2025, maintaining its regional leadership throughout the forecast period at a CAGR of approximately 5.40% from 2026 to 2033. The United States is the primary revenue driver within the region, where TB screening requirements are systematically enforced across healthcare workers, correctional facility employees, school staff, and new immigrants — generating high, consistent, and institutionally driven annual tuberculin testing volumes that underpin the region's market leadership. The U.S. market is served primarily by two dominant tuberculin product manufacturers: Sanofi, whose Tubersol brand holds a leading market position, and Par Pharmaceutical, which produces Aplisol — together accounting for the vast majority of the U.S. tuberculin supply. The region's advanced hospital infrastructure, well-developed occupational health program networks, and high standards of product regulatory compliance provide a stable and high-value commercial environment for TUDS test product suppliers.
Canada contributes meaningfully to North American market revenue through its provincial health systems' consistent procurement of tuberculin products for healthcare worker TB screening programs and newcomer health assessments — a significant and growing demand driver given Canada's high annual immigration intake from TB-endemic countries. The region benefits from well-coordinated national TB surveillance infrastructure and strong CDC and Public Health Agency of Canada guidance that reinforces the role of the Mantoux TUDS test as a core component of TB control programs. North America's market position is expected to remain secure through 2033 despite competitive pressure from IGRA diagnostics, as the established cost advantage and widespread clinical familiarity of tuberculin skin testing ensures its continued use in high-volume screening contexts.
Asia Pacific
Asia Pacific Is the Fastest-Growing TUDS Test Market, Driven by the World's Highest TB Burden, Rapidly Scaling National TB Programs, and Unprecedented Government Investment in Latent TB Infection Screening
Asia Pacific is the fastest-growing regional segment in the TUDS Test market, projected to expand at a CAGR of approximately 7.10% from 2026 to 2033 — the highest of any region. The region accounts for the largest proportion of global TB cases, with India, China, Indonesia, the Philippines, and Pakistan collectively representing the majority of the world's annual new TB infection burden. Governments across the region are scaling up national TB control and elimination programs at an unprecedented pace, driven by WHO End TB Strategy targets and by growing recognition of the enormous economic cost of high TB prevalence on national productivity and healthcare systems. India alone — the country with the highest global TB case load — has launched an ambitious National TB Elimination Programme targeting TB elimination by 2025 (revised to 2030), which includes large-scale latent TB infection screening using tuberculin skin tests as a key component. Major tuberculin product suppliers in Asia Pacific include Serum Institute of India — the world's largest manufacturer of WHO-prequalified tuberculin products — as well as regional suppliers in China and several international manufacturers who supply tuberculin through WHO and government tender procurement channels.
Japan, South Korea, and Australia represent the more mature sub-markets within Asia Pacific, where TB incidence is significantly lower but where structured occupational health TB screening programs for healthcare workers and systematic screening of migrants from high-burden countries generate consistent tuberculin testing demand. The TUDS test market in these sub-markets is also beginning to experience a degree of substitution by IGRA diagnostics in clinical settings, mirroring trends seen in North America and Europe. However, the sheer scale of TB burden and government screening program activity in South and Southeast Asia far outweighs this substitution effect at the regional level, ensuring that Asia Pacific maintains its position as the market's most dynamic and fastest-growing geographic segment throughout the 2026–2033 forecast period.
Top Key Players
-
Sanofi S.A. (France)
-
Par Pharmaceutical, Inc. — Endo International (United States)
-
Serum Institute of India Pvt. Ltd. (India)
-
Becton, Dickinson and Company (United States)
-
Thermo Fisher Scientific Inc. (United States)
-
Bio-Rad Laboratories, Inc. (United States)
-
Statens Serum Institut (Denmark)
-
Span Diagnostics Ltd. (India)
-
Arkray, Inc. (Japan)
-
Chembio Diagnostics, Inc. (United States)
-
Creative Diagnostics (United States)
-
Coris BioConcept (Belgium)
Recent Developments
-
Sanofi S.A. (2025) — Reinforced its leadership in the North American tuberculin market by securing renewed long-term supply contracts for Tubersol with major U.S. hospital networks and occupational health program providers, and expanded its tuberculin product distribution agreement to cover additional Latin American public health procurement channels, strengthening its global market position.
-
Serum Institute of India (2024) — Expanded its WHO-prequalified tuberculin product manufacturing capacity significantly to meet growing international demand from national TB programs across Asia Pacific and sub-Saharan Africa, positioning itself as the world's largest volume supplier of tuberculin testing products for government-funded TB control and elimination programs globally.
-
Becton, Dickinson and Company (2025) — Launched an updated tuberculin skin test administration kit featuring an improved intradermal injection device with enhanced precision and reduced operator variability for the Mantoux test, targeting hospitals, public health clinics, and occupational health programs in North America and Europe seeking to improve the consistency of TUDS test administration.
-
Statens Serum Institut (2024) — Received WHO prequalification renewal for its tuberculin PPD RT23 product — the world's most widely used reference tuberculin — ensuring continued eligibility for procurement by UN agencies and international health programs and reinforcing Denmark's role as a critical supplier of standardized tuberculin for global TB screening programs.
-
Thermo Fisher Scientific (2025) — Advanced the development of a digital tuberculin skin test reading platform integrating smartphone-based induration measurement with automated result interpretation, initiating field validation studies in partnership with public health authorities in India and the Philippines to assess the platform's accuracy and scalability as a tool for improving TUDS test program efficiency in high-burden settings.
Market Trends
Digital Tuberculin Test Reading Technology and Expanding Government-Funded Latent TB Screening Programs Are the Two Most Consequential Trends Reshaping the TUDS Test Market During the Forecast Period
The most transformative technological trend emerging in the TUDS test market is the development and progressive validation of digital tuberculin skin test reading platforms that use smartphone-based imaging and AI-powered measurement algorithms to objectively quantify Mantoux test reactions. These platforms are designed to eliminate the need for the traditional second in-person clinical visit by allowing trained community health workers — or even patients themselves in supervised home-based testing protocols — to capture standardized photographs of the test site for automated induration measurement and interpretation. Early field validation studies in South Asian and African settings are showing promising accuracy results, and if these platforms achieve regulatory clearance in major markets during the forecast period, they could significantly improve the scalability, compliance, and cost-effectiveness of tuberculin skin testing programs in high-burden settings — potentially accelerating market volume growth by extending TUDS testing into community and mobile screening contexts that have historically been difficult to serve.
The second dominant trend is the unprecedented scale-up of government-funded latent TB infection screening programs globally, driven by the WHO End TB Strategy's 2025 and 2030 milestone targets and by growing scientific consensus that treating latent TB infection is essential to achieving TB elimination. Countries with high latent TB prevalence — including India, China, Brazil, and South Africa — are implementing or expanding national systematic testing and treatment programs that target specific high-risk population groups, generating very large incremental procurement volumes for tuberculin testing products. The combination of direct government funding, international donor support through mechanisms such as the Global Fund, and growing political commitment to TB elimination across high-burden countries creates a powerful and durable institutional demand tailwind for the TUDS test market that is expected to sustain above-baseline market growth throughout the 2026–2033 forecast period.
Segments Covered in the Report
By Strength/Dosage:
-
5 TU (Tuberculin Units)
-
10 TU
-
1 TU
-
Other Dosage Strengths
By Application:
-
Intradermal Tuberculin Skin Test (Mantoux Test)
-
Pre-Employment and Occupational Health Screening
-
Epidemiological Screening Programs
-
Clinical Diagnosis of Latent TB Infection
-
Post-Vaccination Assessment
By End User:
-
Hospitals
-
Diagnostic Laboratories
-
Public Health Clinics
-
Research Institutes and Universities
-
TB Control Programs
By Distribution Channel:
-
Direct Hospital Procurement
-
Pharmaceutical Distributors
-
Government Public Health Procurement
-
Online Medical Supply Platforms
By Region:
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the TUDS Test market and what is its projected value by 2033?
Answer: The TUDS Test market was valued at USD 438.61 million in 2025 and is projected to reach USD 829.80 million by 2033. It is expected to grow at a CAGR of 5.98% from 2026 to 2033, driven by high global TB burden, expanding national screening programs, and growing occupational health testing requirements.
Question 2: What are the primary growth drivers of the TUDS Test market?
Answer: The persistent global burden of tuberculosis and government-funded national TB control and elimination programs are the two most powerful drivers of the TUDS Test market. Growing mandatory occupational health TB screening requirements in healthcare, education, and correctional settings are also meaningfully expanding the addressable market for tuberculin testing products globally.
Question 3: Which region leads the TUDS Test market and which is the fastest growing?
Answer: North America leads the TUDS Test market in 2025, supported by robust institutional TB screening mandates and a strong regulatory-compliant tuberculin product supply chain. Asia Pacific is the fastest-growing region, expanding at approximately 7.10% CAGR through 2033 due to the world's highest TB burden and rapidly scaling national TB screening programs.
Question 4: Which dosage segment and application dominate the TUDS Test market?
Answer: The 5 TU dosage segment dominates the TUDS Test market as the globally standardized Mantoux test dose recommended by the WHO and CDC for routine TB screening. The intradermal tuberculin skin test (Mantoux test) is the leading application segment, while epidemiological screening programs represent the fastest-growing application driven by government-funded latent TB infection detection initiatives.
Question 5: What key innovations are shaping the future of the TUDS Test market?
Answer: Digital tuberculin skin test reading platforms using AI-powered smartphone-based induration measurement are the most transformative emerging innovation in the TUDS Test market. These tools have the potential to eliminate the traditional two-visit protocol barrier and extend tuberculin testing into community-based and mobile screening settings — significantly improving program scalability and patient compliance in high-burden geographies.
