Tuberculosis Diagnostics Market Overview
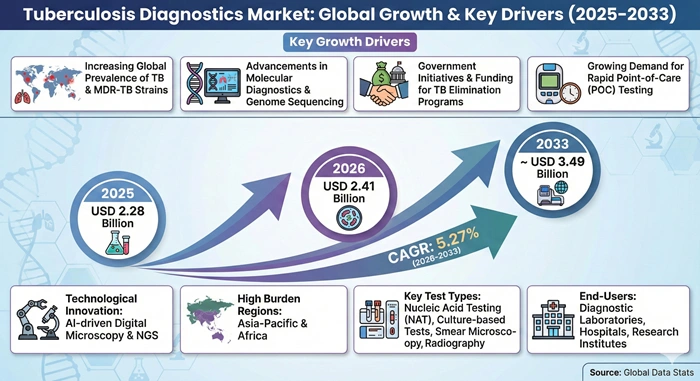
The global tuberculosis diagnostics market size is valued at USD 2.28 billion in 2025 and is predicted to increase from USD 2.41 billion in 2026 to approximately USD 3.49 billion by 2033, growing at a CAGR of 5.27% from 2026 to 2033.
The rising global burden of tuberculosis, particularly in developing nations, has created urgent demand for accurate and rapid diagnostic solutions that enable early disease detection and appropriate treatment initiation. TB remains one of the deadliest infectious diseases worldwide, causing millions of new infections annually and presenting significant public health challenges across both developed and emerging economies. Healthcare systems are increasingly prioritizing investment in advanced diagnostic technologies including molecular tests, rapid detection systems, and point-of-care solutions that deliver faster results compared to traditional culture methods requiring weeks for confirmation. The market benefits from strong government initiatives supporting TB elimination programs, growing awareness about the importance of early diagnosis, technological breakthroughs in nucleic acid amplification tests, expanding healthcare infrastructure in high-burden countries, and continuous innovation by leading diagnostic companies developing next-generation testing platforms with improved sensitivity and specificity for detecting both drug-susceptible and drug-resistant TB strains.
AI Impact on the Tuberculosis Diagnostics Industry
Artificial Intelligence Revolutionizes Diagnostic Accuracy and Accelerates Disease Detection in Global TB Control Efforts
Artificial intelligence is fundamentally transforming the tuberculosis diagnostics market by enhancing the accuracy and speed of TB detection through advanced image analysis algorithms that interpret chest X-rays and CT scans with precision matching or exceeding human radiologists. AI-powered diagnostic platforms analyze radiological images to identify characteristic patterns associated with pulmonary tuberculosis, including cavitary lesions, infiltrates, nodules, and pleural effusions that indicate active disease. Machine learning models trained on millions of chest radiographs from diverse patient populations can detect subtle abnormalities invisible to the human eye, reducing false-negative rates that historically allowed infected individuals to go undiagnosed and continue transmitting disease within communities. These intelligent systems provide rapid preliminary screening results in resource-limited settings lacking specialized radiologists, enabling healthcare workers to quickly triage patients requiring further confirmatory testing. AI integration into portable X-ray devices creates point-of-care screening capabilities suitable for remote locations, mobile health clinics, and community outreach programs targeting high-risk populations including homeless individuals, prisoners, and migrant workers with limited healthcare access.
Furthermore, AI is revolutionizing laboratory workflows by automating the interpretation of microscopy images, molecular test results, and bacterial culture analyses, dramatically reducing turnaround times and human error in TB diagnosis. Computer vision algorithms examine sputum smear microscopy slides to identify acid-fast bacilli with consistency superior to manual reading, particularly important in high-volume laboratories processing thousands of samples daily. Predictive analytics help identify individuals at highest risk for developing active TB disease from latent infections by analyzing demographic factors, medical histories, chest imaging findings, and immunological markers, enabling targeted preventive interventions. Natural language processing tools extract relevant clinical information from unstructured electronic health records to flag patients with symptoms suggesting possible TB, prompting clinicians to order appropriate diagnostic tests. AI-driven supply chain optimization ensures diagnostic facilities maintain adequate stocks of testing reagents, reducing delays caused by inventory shortages. As AI capabilities continue advancing, the tuberculosis diagnostics market will benefit from improved diagnostic accuracy, faster disease detection, expanded screening coverage, and more efficient resource utilization supporting global TB elimination goals.
Growth Factors
Rising TB Prevalence and Government Initiatives Drive Robust Market Expansion
The tuberculosis diagnostics market is experiencing substantial growth driven by the persistently high global burden of TB infections, with the World Health Organization reporting millions of new cases annually concentrated in high-burden countries across Asia, Africa, and Eastern Europe. Drug-resistant tuberculosis including multidrug-resistant TB and extensively drug-resistant TB presents escalating public health threats requiring sophisticated molecular diagnostic tests capable of rapidly identifying resistance mutations to guide appropriate treatment selection. TB co-infection with HIV creates particularly vulnerable patient populations experiencing higher mortality rates and requiring specialized diagnostic approaches addressing the challenges of diagnosing TB in immunocompromised individuals who may present with atypical disease manifestations. Population factors including urbanization, overcrowding in cities, malnutrition, poverty, and inadequate living conditions facilitate TB transmission and sustain endemic disease in many regions. The COVID-19 pandemic disrupted TB control programs and reduced diagnosis rates during lockdown periods, creating accumulated undiagnosed cases that are now emerging as active disease requiring diagnostic services.
Strong government and multilateral organization support for TB control programs represents a critical growth driver accelerating the tuberculosis diagnostics market through funding initiatives, policy frameworks, and technical assistance strengthening national laboratory networks. The Global Fund to Fight AIDS, Tuberculosis and Malaria provides billions in financial resources supporting TB diagnosis and treatment in low and middle-income countries, enabling procurement of modern diagnostic equipment and testing supplies. National tuberculosis elimination programs implement systematic screening strategies targeting high-risk populations and contacts of confirmed cases, generating steady diagnostic testing volumes. WHO policy recommendations endorsing specific molecular tests as preferred initial diagnostic approaches over traditional microscopy drive technology adoption in public health laboratories. Regulatory approvals and emergency use authorizations for novel diagnostic platforms accelerate market entry and clinical implementation. Public-private partnerships bring together governments, nonprofit organizations, and commercial diagnostic companies to expand testing access, reduce prices for resource-limited settings, and develop innovative solutions addressing unmet diagnostic needs in TB control.

Market Outlook
Point-of-Care Technologies and Digital Solutions Transform TB Diagnosis Landscape
The tuberculosis diagnostics market outlook remains highly positive as point-of-care testing technologies advance toward true bedside diagnosis capabilities enabling same-day treatment initiation without requiring centralized laboratory infrastructure. Portable molecular diagnostic platforms including handheld devices and battery-powered systems bring sophisticated nucleic acid amplification testing to primary care clinics, rural health posts, and community screening programs in settings lacking electricity or reliable cold chain for reagent storage. Microfluidic chip-based diagnostics miniaturize complex laboratory processes into compact cartridges requiring minimal sample volumes and operator training, democratizing access to advanced molecular testing. Lateral flow assays and rapid immunochromatographic tests provide results within minutes at point of care, though sensitivity limitations compared to molecular methods constrain their use to screening rather than definitive diagnosis. Ultra-portable chest X-ray systems combined with AI interpretation algorithms create screening capabilities deployable to remote communities through mobile health vans and temporary field clinics.
Digital health integration and connectivity solutions are transforming tuberculosis diagnostics market dynamics by enabling remote expert consultation, real-time data reporting to public health authorities, and comprehensive disease surveillance systems tracking TB epidemiology patterns. Cloud-connected diagnostic instruments automatically transmit test results to electronic health records and national TB registries, eliminating delays from manual data entry and paper-based reporting systems. Telemedicine platforms allow frontline healthcare workers in remote locations to share patient chest X-rays and clinical information with specialist radiologists and pulmonologists located in urban referral centers, receiving guidance on interpretation and patient management within hours. Smartphone-based microscopy attachments convert mobile phones into portable diagnostic tools capable of imaging sputum smears for TB bacilli detection, with images uploaded to cloud servers for AI-assisted analysis or expert review. Digital adherence technologies including video-observed therapy and electronic medication monitors support treatment completion after diagnosis, generating real-world data on treatment outcomes. Blockchain applications create tamper-proof audit trails for diagnostic test results and ensure supply chain integrity for diagnostic reagents. These digital innovations expand diagnostic reach, improve data quality, enhance disease surveillance, and strengthen connections between diagnostic services and patient care delivery.
Expert Speaks
-
Robert Ford, CEO of Abbott, emphasized during 2024 that Abbott remains committed to developing innovative diagnostic solutions addressing global health challenges including infectious diseases, leveraging the company's expertise in molecular diagnostics, point-of-care testing, and laboratory automation to improve access to quality healthcare worldwide and support public health initiatives combating major disease burdens.
-
Thomas Schinecker, CEO of Roche, highlighted throughout 2024 and early 2025 that Roche continues investing in next-generation diagnostic technologies and strengthening partnerships with health authorities worldwide, demonstrating the company's dedication to improving patient outcomes through innovative solutions that enable earlier disease detection and personalized treatment approaches across diverse therapeutic areas including infectious diseases.
-
Tom Polen, former CEO of Becton Dickinson, stated during his tenure that BD maintains strategic focus on advancing diagnostic innovation, expanding access to essential medical technologies in underserved markets, and collaborating with global health organizations to address critical public health priorities including tuberculosis through development of integrated diagnostic solutions improving disease detection and management capabilities.
Key Report Takeaways
-
Asia Pacific dominates the tuberculosis diagnostics market with approximately 40.3% market share in 2025, driven by the highest global TB burden concentrated in countries including India, China, Indonesia, and the Philippines, large populations, expanding healthcare infrastructure, government initiatives supporting TB elimination programs, growing awareness of early diagnosis importance, and increasing adoption of molecular diagnostic technologies in public health laboratories and private healthcare facilities.
-
North America represents the fastest-growing regional market with projected CAGR of 5.5% during the forecast period from 2026 to 2033, fueled by advanced healthcare infrastructure, high awareness levels, robust government funding for TB control, strong emphasis on identifying and treating latent TB infections, technological innovation in diagnostic platforms, comprehensive screening programs for high-risk populations, and established reimbursement systems supporting adoption of sophisticated molecular tests.
-
Nucleic acid testing segment leads the market with commanding 36.45% market share in 2025 due to WHO recommendations prioritizing molecular diagnostics over traditional microscopy, superior sensitivity and specificity detecting TB bacteria and drug resistance mutations, rapid turnaround times enabling same-day diagnosis, ability to test directly from clinical specimens without culture requirement, widespread adoption of GeneXpert systems, and continuous innovation in molecular testing platforms.
-
Active TB disease segment accounts for the largest share representing the primary focus of diagnostic efforts globally, reflecting urgent clinical and public health priorities surrounding prompt detection and treatment of infectious cases to prevent disease transmission, reduce mortality, and support TB elimination goals through early identification of symptomatic patients requiring immediate anti-tuberculosis therapy.
-
Diagnostic laboratories segment dominates end-user category with approximately 49% market share in 2025, attributed to centralized laboratory networks processing high testing volumes, specialized equipment and trained personnel required for complex molecular and culture-based diagnostics, quality assurance programs ensuring result accuracy, integration with public health TB control programs, and established referral systems connecting clinical facilities to diagnostic laboratories.
-
Cytokine detection tests projected to achieve fastest growth throughout the forecast period, expanding rapidly as interferon-gamma release assays demonstrate superior performance for latent TB infection diagnosis compared to traditional tuberculin skin tests, offering objective interpretation without reader variability, requiring single patient visit eliminating need for return reading appointments, and providing reliable results in BCG-vaccinated populations where skin tests produce false-positive reactions.
Market Scope
| Report Coverage Details | |
|---|---|
| Market Size by 2033 | USD 3.49 Billion |
| Market Size by 2025 | USD 2.28 Billion |
| Market Size by 2026 | USD 2.41 Billion |
| Market Growth Rate from 2026 to 2033 | CAGR of 5.27% |
| Dominating Region | Asia Pacific |
| Fastest Growing Region | North America |
| Base Year | 2025 |
| Forecast Period | 2026 to 2033 |
| Segments Covered | Test Type, Disease Stage, End User, Region |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
Market Dynamics
Drivers Impact Analysis
Technological Advancements and Expanding Healthcare Access Accelerate Market Growth
| Impact Factor | Details |
|---|---|
| ≈ Impact on CAGR Forecast | +1.2% to +1.6% |
| Geographic Relevance | Global with strongest impact in developing markets |
| Impact Timeline | Immediate and sustained through 2033 |
Rapid technological advancement in molecular diagnostics represents a fundamental driver propelling the tuberculosis diagnostics market forward through development of nucleic acid amplification tests delivering accurate results within hours compared to weeks required for traditional culture methods. GeneXpert MTB/RIF systems from Cepheid have revolutionized TB diagnosis by simultaneously detecting Mycobacterium tuberculosis and rifampicin resistance in under two hours using automated cartridge-based platforms requiring minimal technical expertise, enabling deployment in peripheral healthcare facilities lacking sophisticated laboratory infrastructure. Next-generation sequencing technologies provide comprehensive drug susceptibility testing identifying resistance mutations to multiple anti-tuberculosis medications, guiding personalized treatment regimens particularly important for managing multidrug-resistant and extensively drug-resistant cases. Loop-mediated isothermal amplification assays offer simplified molecular testing suitable for field settings without requiring thermal cycling equipment. Digital PCR platforms deliver enhanced sensitivity for detecting low bacterial loads in paucibacillary TB cases including pediatric and HIV-associated disease where diagnosis proves particularly challenging. Advances in sample processing including liquefaction, concentration, and purification methods improve test performance from various specimen types including sputum, urine, stool, and blood.
Expanding healthcare infrastructure in high-burden countries creates essential foundation supporting tuberculosis diagnostics market growth by establishing laboratory networks, training diagnostic personnel, and implementing quality assurance programs ensuring reliable test results. National TB control programs invest in strengthening laboratory capacity through procurement of modern diagnostic equipment, renovation of testing facilities to meet biosafety standards, and development of specimen transportation systems connecting peripheral health centers to reference laboratories. International development assistance from organizations including USAID, PEPFAR, and the Global Fund finances laboratory strengthening activities in resource-limited settings, subsidizing equipment costs and operational expenses. Public health laboratories expand testing menus to include molecular diagnostics and drug susceptibility testing previously available only at central reference facilities, improving geographic access and reducing diagnostic delays. Private sector laboratory chains establish branches in secondary cities and rural areas, creating competitive markets driving quality improvement and cost reduction. Mobile laboratory units bring diagnostic services directly to underserved communities through outreach programs. Telemedicine connectivity enables remote technical support and quality monitoring. These infrastructure investments create enabling environments for adoption of advanced diagnostic technologies and expansion of testing coverage supporting TB case detection.

Restraints Impact Analysis
High Costs and Technical Complexity Present Implementation Challenges
| Impact Factor | Details |
|---|---|
| ≈ Impact on CAGR Forecast | -0.5% to -0.8% |
| Geographic Relevance | Particularly acute in low-income countries |
| Impact Timeline | Ongoing challenge throughout forecast period |
The substantial capital investment and operational costs associated with advanced molecular diagnostic platforms represent significant restraints limiting tuberculosis diagnostics market penetration in resource-constrained settings where TB burden concentrates most heavily. GeneXpert systems require initial equipment purchases costing tens of thousands of dollars per instrument, with ongoing expenses for proprietary test cartridges, calibration materials, maintenance contracts, and replacement parts creating recurring financial obligations straining limited public health budgets. Electricity supply interruptions common in many developing countries damage sensitive diagnostic equipment and interrupt testing services, necessitating expensive backup power systems. Cold chain requirements for reagent storage and transport add logistical complexity and costs in tropical climates and remote locations. Laboratory infrastructure upgrades including biosafety cabinets, ventilation systems, and specimen processing areas require capital investments many facilities cannot afford. Training costs for laboratory personnel operating sophisticated instruments and interpreting complex results consume scarce human resource development budgets. External quality assurance program participation fees and proficiency testing materials represent additional expenses. Patent protections and proprietary technologies limit competition that could reduce prices through generic alternatives.
Technical complexity and infrastructure requirements for implementing advanced TB diagnostic technologies create operational challenges restraining market growth in settings with limited technical expertise and weak health systems. Molecular diagnostic platforms require stable electricity supply, temperature-controlled storage, regular calibration, preventive maintenance, and technical troubleshooting capabilities often unavailable in peripheral health facilities serving rural populations. Laboratory information systems for managing specimens, tracking results, and generating reports demand IT infrastructure and trained personnel scarce in many developing countries. Biosafety requirements for handling potentially infectious TB specimens necessitate specialized facilities, personal protective equipment, and trained staff following proper procedures. Sample transportation networks connecting collection sites to testing laboratories require reliable logistics systems, standardized specimen containers, and cold chain capacity. Quality assurance programs ensuring result accuracy demand ongoing staff competency assessment, equipment performance verification, and participation in external proficiency testing schemes. Regulatory approval processes for new diagnostic technologies vary across countries, creating delays in product availability. Technology dependence on external suppliers for consumables, spare parts, and technical support creates vulnerabilities to supply chain disruptions. These technical and operational challenges slow the tuberculosis diagnostics market expansion in precisely those settings where improved diagnosis could deliver greatest public health impact.
Opportunities Impact Analysis
Pipeline Innovations and Integration Strategies Create Growth Avenues
| Impact Factor | Details |
|---|---|
| ≈ Impact on CAGR Forecast | +0.9% to +1.3% |
| Geographic Relevance | Global with emphasis on emerging markets |
| Impact Timeline | Medium to long term (2027-2033) |
The robust pipeline of next-generation diagnostic technologies under development presents substantial opportunities for the tuberculosis diagnostics market as innovations address current limitations in sensitivity, cost, turnaround time, and ease of use. Breath-based diagnostic systems detecting volatile organic compounds produced by TB bacteria offer completely non-invasive sampling eliminating difficulties obtaining quality sputum specimens from children, HIV-positive patients, and individuals with extrapulmonary disease. CRISPR-based diagnostics leverage gene-editing technology for ultrasensitive pathogen detection at fraction of current molecular test costs, potentially enabling widespread deployment in resource-limited settings. Biosensor platforms incorporating nanomaterials provide rapid pathogen detection through electrical or optical signals generated by specific TB antigen binding. Smartphone-based microscopy and AI image analysis democratize diagnostic capabilities by converting ubiquitous mobile devices into portable diagnostic tools. Multiplexed assays simultaneously detect TB alongside other respiratory pathogens including COVID-19, influenza, and bacterial pneumonia, improving differential diagnosis and operational efficiency. Host biomarker signatures in blood identifying active TB versus latent infection or other diseases improve diagnostic specificity reducing false-positive results.
Strategic integration of TB diagnostics into broader healthcare delivery systems and disease control programs creates opportunities for market expansion beyond traditional vertical TB services toward comprehensive respiratory health platforms. Integration of TB testing into HIV care programs captures patients requiring routine TB screening, with point-of-care molecular platforms enabling same-day diagnosis during clinic visits eliminating loss to follow-up before results return from central laboratories. Diabetes screening programs offer opportunities for active TB case finding among patients with elevated diabetes-related TB risks. Maternal and child health services provide platforms for pediatric TB screening and preventive therapy initiation. Prison health programs address high TB rates in correctional facilities through systematic screening and treatment. Migrant health services identify imported TB cases at borders or settlement camps. Corporate wellness programs conduct employee TB screening particularly in industries with occupational exposure risks. Integration strategies leverage existing health infrastructure, reduce stigma associated with TB-specific services, improve patient convenience, and create sustainable diagnostic service delivery models. Public-private partnerships bringing together governments, multilateral organizations, and commercial diagnostic companies accelerate innovation adoption, reduce prices through volume commitments, and expand market access in underserved populations. These integration and partnership opportunities position the tuberculosis diagnostics market for sustained growth addressing unmet diagnostic needs while supporting broader health system strengthening goals.

Segment Analysis
By Test Type
Nucleic Acid Testing Dominates Market Through Superior Performance and WHO Endorsement
The nucleic acid testing segment commands the tuberculosis diagnostics market with an impressive 36.45% market share in 2025, driven by World Health Organization policy recommendations prioritizing molecular diagnostic methods as preferred initial diagnostic tests replacing traditional sputum smear microscopy in TB diagnostic algorithms. Nucleic acid amplification tests including GeneXpert MTB/RIF, GeneXpert Ultra, and other molecular platforms detect Mycobacterium tuberculosis DNA with sensitivity exceeding 95% in smear-positive pulmonary TB and reaching 60 to 80% in smear-negative cases where traditional microscopy fails to identify infection. Simultaneous detection of rifampicin resistance mutations provides critical information guiding treatment decisions and identifying multidrug-resistant TB cases requiring specialized management with second-line medications. Rapid turnaround times delivering results within two hours enable same-day diagnosis and treatment initiation, dramatically reducing patient loss to follow-up between diagnosis and treatment compared to culture-based methods requiring weeks. Automated platforms minimize hands-on technical work and biosafety risks compared to culture methods involving manipulation of live mycobacteria. The tests operate from unprocessed sputum specimens eliminating complex sample preparation procedures, and closed cartridge systems reduce laboratory contamination and cross-contamination risks.
North America and Europe demonstrate strong adoption of molecular TB diagnostics supported by advanced laboratory infrastructure, favorable reimbursement policies covering expensive molecular tests, and clinical practice guidelines recommending molecular testing as first-line diagnostics. Asia Pacific represents the fastest-growing region for nucleic acid testing as countries including India, China, Indonesia, and the Philippines rapidly scale up GeneXpert networks through government procurement programs and international donor support addressing their high TB burdens. Leading manufacturers including Cepheid (Danaher Corporation), Roche Diagnostics, Abbott Laboratories, and Becton Dickinson maintain extensive product portfolios and continue innovation developing next-generation platforms with improved sensitivity, expanded drug resistance detection, and lower costs. Recent developments include WHO prequalification of Cepheid's Xpert MTB/XDR test in October 2025 detecting resistance to multiple TB medications beyond rifampicin, significantly expanding molecular diagnostic capabilities for comprehensively characterizing drug resistance patterns. The segment benefits from continued research and development producing novel molecular technologies, expanding indications to pediatric TB and extrapulmonary disease, and ongoing price reductions through competitive procurement improving affordability in resource-limited settings.
By End User
Diagnostic Laboratories Lead Market Through Specialized Testing Infrastructure and High Volumes
The diagnostic laboratories segment dominates the tuberculosis diagnostics market end-user category with approximately 49% market share in 2025, reflecting the essential role of specialized laboratory facilities equipped with sophisticated instrumentation, trained personnel, and quality assurance systems ensuring accurate and reliable TB test results. Public health laboratory networks operated by government TB control programs process the majority of diagnostic specimens in high-burden countries, performing microscopy, culture, molecular testing, and drug susceptibility testing supporting clinical care and disease surveillance. These centralized facilities manage high testing volumes achieving economies of scale, maintain expensive equipment including automated culture systems and molecular platforms, and employ specialized medical laboratory scientists and technicians with expertise in mycobacteriology. Reference laboratories provide specialized testing services including second-line drug susceptibility testing, TB genotyping, and resolution of ambiguous results not available at peripheral facilities. Private diagnostic laboratory chains serve paying patients and health insurance networks, investing in state-of-the-art technology and competing on turnaround time and service quality. Academic medical center laboratories combine diagnostic services with research and training missions.
The United States diagnostic laboratory segment benefits from extensive commercial and public health laboratory capacity, advanced automation, comprehensive test menus, and quality standards enforced through Clinical Laboratory Improvement Amendments regulations and College of American Pathologists accreditation. Asia Pacific countries are rapidly expanding laboratory networks through government investments in TB diagnostic infrastructure as part of national elimination strategies, with India deploying thousands of GeneXpert machines across public sector laboratories and establishing culture and drug susceptibility testing laboratories covering all states. Companies including Abbott Laboratories, Roche Diagnostics, Becton Dickinson, bioMérieux, and Thermo Fisher Scientific supply diagnostic platforms, reagents, and technical support to laboratory networks worldwide. The segment is experiencing strong growth as testing volumes increase with intensified case-finding activities, molecular test adoption expands, and laboratories add drug susceptibility testing capabilities. The hospitals and clinics end-user segment is projected to achieve fastest growth throughout the forecast period as point-of-care molecular testing platforms enable decentralization of sophisticated diagnostics from central laboratories directly to clinical care settings, reducing patient travel, accelerating treatment initiation, and improving retention in care particularly in remote and underserved areas.

Regional Insights
Asia Pacific
High Disease Burden and Government Initiatives Drive Regional Market Leadership
Asia Pacific maintains commanding dominance in the tuberculosis diagnostics market with approximately 40.3% of global market share in 2025, driven by the region's overwhelming concentration of global TB disease burden with countries including India, China, Indonesia, Philippines, Pakistan, and Bangladesh collectively accounting for more than half of worldwide TB cases. India alone reports over 2.5 million TB cases annually representing the world's largest national TB burden, creating massive diagnostic testing demand across public and private healthcare sectors. The region's large populations, high population density in urban areas facilitating transmission, poverty concentrations, malnutrition, and HIV co-infection rates sustain endemic TB and generate continuous diagnostic service requirements. Government commitment to TB elimination reflected in national strategic plans, dedicated funding allocations, and systematic screening programs targeting high-risk groups drives diagnostic testing volumes. The private healthcare sector provides substantial diagnostic capacity complementing public health laboratories, particularly in countries like India where private providers deliver majority of TB care.
Asia Pacific countries are rapidly modernizing TB diagnostic infrastructure through massive investments in molecular testing platforms, with India deploying over 1,000 GeneXpert machines and establishing culture and drug susceptibility testing facilities nationwide. China's TB control program integrates advanced diagnostics throughout its extensive healthcare system serving 1.4 billion people. Leading global diagnostic companies including Cepheid, Roche Diagnostics, Abbott Laboratories, Becton Dickinson, and bioMérieux maintain strong commercial presence across the region through local subsidiaries, distribution partnerships, and participation in government procurement programs. Regional manufacturers including Indian diagnostic companies contribute to market dynamics through affordable alternative products. The region is projected to grow at approximately 5.6% CAGR from 2026 to 2033, sustained by continuing high disease burden, expanding healthcare access in rural areas, government policy support for diagnostic scale-up, increasing private sector participation, and technological innovations enabling point-of-care testing deployment in peripheral facilities bringing diagnostics closer to patients requiring services.
North America
Advanced Infrastructure and Comprehensive Programs Position Region for Fastest Growth
North America represents the fastest-growing regional market in the tuberculosis diagnostics market, projected to achieve approximately 5.5% CAGR during the forecast period from 2026 to 2033, driven by comprehensive TB control programs emphasizing identification and treatment of latent TB infections preventing progression to active disease. The United States maintains sophisticated TB surveillance and control infrastructure through state and local health departments operating specialized TB clinics, contact investigation programs, and targeted screening initiatives identifying infections among foreign-born individuals, healthcare workers, prisoners, homeless populations, and other high-risk groups. Canada implements similar systematic approaches through provincial TB control programs. While absolute case numbers remain relatively low compared to high-burden countries, the emphasis on latent TB infection testing generates substantial diagnostic demand for interferon-gamma release assays and tuberculin skin tests. Active TB case investigation requires confirmatory molecular testing, culture, and drug susceptibility analysis performed by specialized public health laboratories equipped with state-of-the-art instrumentation.
The United States market benefits from advanced diagnostic infrastructure including commercial reference laboratories, hospital laboratories, and public health laboratories networked through standardized reporting systems and quality assurance programs ensuring result reliability. Leading diagnostic companies including Abbott Laboratories, Becton Dickinson, Qiagen, and others maintain headquarters or major operations in North America, driving innovation and early adoption of novel diagnostic technologies. Favorable reimbursement from Medicare, Medicaid, and commercial insurance supports utilization of expensive molecular tests and specialized assays. The Centers for Disease Control and Prevention provides technical guidance, funding support, and laboratory capacity building strengthening national TB diagnostic capabilities. Recent market dynamics include increasing adoption of molecular diagnostics replacing traditional methods, growing emphasis on interferon-gamma release assays for latent TB screening due to advantages over skin testing, and integration of TB testing into broader respiratory pathogen panels. The region's growth trajectory reflects sustained government investment in TB elimination efforts, technological advancement adoption, comprehensive screening program expansion, and ongoing research and development producing diagnostic innovations.
Top Key Players
-
Abbott Laboratories (United States)
-
Becton, Dickinson and Company (United States)
-
F. Hoffmann-La Roche AG (Switzerland)
-
bioMérieux SA (France)
-
Thermo Fisher Scientific Inc. (United States)
-
Cepheid Inc. (Danaher Corporation) (United States)
-
QIAGEN N.V. (Germany)
-
Hologic, Inc. (United States)
-
Hain Lifescience GmbH (Germany)
-
Oxford Immunotec (PerkinElmer) (United Kingdom)
-
Sansure Biotech Inc. (China)
-
Siemens Healthineers AG (Germany)
-
Quidel Corporation (QuidelOrtho Corporation) (United States)
-
Meridian Bioscience Inc. (United States)
-
Creative Diagnostics (United States)
Recent Developments
-
In October 2025, Cepheid announced World Health Organization prequalification of the Xpert MTB/XDR test, representing a significant advancement in rapid molecular diagnostics for extensively drug-resistant tuberculosis by detecting resistance to isoniazid, fluoroquinolones, ethionamide, and second-line injectable agents within 90 minutes on GeneXpert systems, enabling clinicians to quickly identify patients requiring specialized treatment regimens and infection control measures.
-
In August 2023, Danaher Corporation completed acquisition of Abcam plc for approximately USD 5.7 billion, strengthening the company's life sciences portfolio with Abcam's extensive collection of high-quality antibodies and reagents useful in tuberculosis diagnostic development, enhancing research capabilities supporting innovation in TB detection technologies and expanding the scientific tools available for advancing diagnostic assay development.
-
In February 2023, Roche Diagnostics announced enhancement of its partnership with the U.S. Centers for Disease Control and Prevention, collaborating to strengthen laboratory capacity in countries heavily impacted by tuberculosis through technical assistance, equipment provision, training programs, and quality assurance support, demonstrating commitment to improving global TB diagnostic infrastructure and advancing public health goals.
-
In March 2023, PerkinElmer Oxford Immunotec received FDA approval for use of Auto-Pure 24 and Auto-Pure 20B platforms with its T-SPOT.TB test, expanding the cell isolation instrument options compatible with this interferon-gamma release assay for latent TB infection diagnosis, improving laboratory workflow efficiency and accessibility of this advanced diagnostic technology in clinical and public health settings across the United States.
-
In January 2025, AlterDiag partnered with Institut Pasteur to develop next-generation rapid diagnostic tests, utilizing single-domain antibodies and lateral flow immunochromatography technology to create affordable, high-performance point-of-care tests for infectious diseases including tuberculosis, aiming to improve rapid TB detection accuracy and accessibility particularly in resource-limited settings where sophisticated laboratory infrastructure remains unavailable.
Market Trends
Point-of-Care Testing and Digital Health Integration Transform Diagnostic Service Delivery
The tuberculosis diagnostics market is experiencing a pronounced shift toward point-of-care testing solutions that decentralize sophisticated molecular diagnostics from centralized laboratories directly to clinical care settings, enabling same-day diagnosis and treatment initiation that improves patient outcomes and reduces loss to follow-up. Portable molecular platforms including battery-powered GeneXpert Edge systems and other field-deployable devices bring nucleic acid amplification testing to remote health posts, mobile clinics, and community outreach programs in settings lacking reliable electricity, temperature-controlled storage, or technical expertise previously required for molecular diagnostics. Handheld diagnostic devices under development promise true bedside testing capabilities fitting in backpacks and operating from smartphone power supplies. Rapid lateral flow tests detecting TB antigens or antibodies provide screening results within 15 minutes at point of care, though sensitivity limitations compared to molecular methods restrict their role to preliminary screening requiring confirmatory testing. Ultra-portable chest X-ray systems weighing under 50 pounds enable radiographic screening in field settings, with AI interpretation algorithms providing immediate preliminary reads flagging abnormalities suggesting TB.
Digital health technologies and connectivity solutions are revolutionizing the tuberculosis diagnostics market by enabling remote expert consultation, real-time data transmission to public health authorities, and comprehensive disease surveillance systems informing TB control strategies. Cloud-connected diagnostic instruments automatically upload test results to centralized databases, electronic health records, and national TB case registries, eliminating transcription errors and reporting delays inherent in paper-based systems while enabling real-time epidemiological monitoring of case trends, geographic hotspots, and drug resistance patterns. Telemedicine platforms allow healthcare workers in peripheral facilities to share chest X-ray images with radiologists at referral hospitals, receiving expert interpretation guidance within hours rather than waiting days or weeks for patient referrals. Smartphone applications support diagnostic algorithms guiding clinicians through systematic TB evaluation including symptom screening, risk assessment, and appropriate test selection. Digital adherence technologies including video-observed therapy and electronic pill boxes ensure treatment completion after diagnosis, generating valuable outcome data. Blockchain applications create tamper-proof audit trails for diagnostic results and supply chain tracking. These digital innovations expand diagnostic access, improve data quality, enhance disease surveillance, and strengthen linkages between diagnosis and treatment supporting comprehensive TB care delivery.
Segments Covered in the Report
By Test Type
-
Radiographic Tests (Chest X-Ray, CT Scan)
-
Nucleic Acid Testing (NAT/Molecular Diagnostics)
-
Cytokine Detection Tests (IGRA Tests)
-
Drug Resistance Tests
-
Culture-Based Tests
-
Sputum Smear Microscopy
By Disease Stage
-
Latent TB Infection (LTBI)
-
Active TB Disease
By End User
-
Diagnostic Laboratories (Public Health Labs, Reference Labs, Private Labs)
-
Hospitals and Clinics
-
Research Institutes
-
Others
By Region
-
North America
-
United States
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
United Kingdom
-
France
-
Italy
-
Spain
-
Rest of Europe
-
-
Asia Pacific
-
India
-
China
-
Indonesia
-
Philippines
-
Japan
-
Australia
-
Rest of Asia Pacific
-
-
Latin America
-
Brazil
-
Argentina
-
Rest of Latin America
-
-
Middle East and Africa
-
South Africa
-
GCC Countries
-
Rest of Middle East and Africa
-
Frequently Asked Questions
Question 1: What is the current tuberculosis diagnostics market size and projected growth rate?
Answer: The global tuberculosis diagnostics market is valued at USD 2.28 billion in 2025 and is expected to reach USD 3.49 billion by 2033, expanding at a CAGR of 5.27% from 2026 to 2033. This growth is driven by rising TB prevalence and technological advancements in diagnostic methods.
Question 2: Which region dominates the tuberculosis diagnostics market globally?
Answer: Asia Pacific leads the tuberculosis diagnostics market with approximately 40.3% market share in 2025, driven by high disease burden in countries like India and China. North America represents the fastest-growing region with projected CAGR of 5.5% through 2033.
Question 3: What test type holds the largest share in the tuberculosis diagnostics market?
Answer: Nucleic acid testing dominates the tuberculosis diagnostics market with 36.45% share in 2025 due to WHO recommendations and superior sensitivity. Cytokine detection tests are projected to grow fastest driven by advantages of interferon-gamma release assays for latent TB diagnosis.
Question 4: Which end user segment accounts for the largest tuberculosis diagnostics market share?
Answer: Diagnostic laboratories command approximately 49% of the tuberculosis diagnostics market in 2025 due to specialized infrastructure and high testing volumes. Hospitals and clinics segment is expected to grow rapidly as point-of-care molecular testing platforms enable diagnostic decentralization.
Question 5: Who are the major companies operating in the tuberculosis diagnostics market?
Answer: Leading players in the tuberculosis diagnostics market include Abbott Laboratories, Becton Dickinson and Company, F. Hoffmann-La Roche AG, bioMérieux SA, and Thermo Fisher Scientific Inc. Other key companies include Cepheid Inc., QIAGEN N.V., Hologic Inc., and Hain Lifescience GmbH.
