Theranostics Market Overview
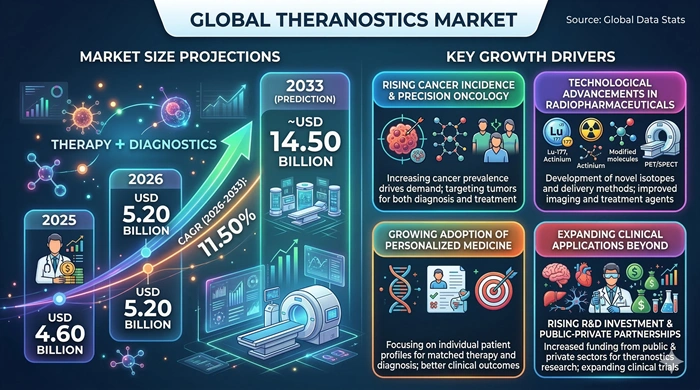
The global Theranostics market size is valued at USD 4.60 billion in 2025 and is predicted to increase from USD 5.20 billion in 2026 to approximately USD 14.50 billion by 2033, growing at a CAGR of 11.50% from 2026 to 2033.
Theranostics — the integration of diagnostics and therapeutics into a single, unified platform — is emerging as one of the most transformative approaches in modern precision medicine. The growing clinical adoption of radiopharmaceutical-based theranostic pairs, particularly in oncology, combined with increasing regulatory approvals and expanding healthcare infrastructure globally, is driving consistent and broad-based growth across this market. The convergence of molecular imaging and targeted therapy is reshaping how cancers and other chronic diseases are detected and treated simultaneously.
AI Impact on the Theranostics Industry
Artificial Intelligence Is Accelerating the Identification of Theranostic Targets, Enhancing Imaging Precision, and Enabling Truly Personalized Treatment Planning at an Unprecedented Scale
Artificial intelligence is fundamentally transforming the Theranostics market by dramatically improving the speed and accuracy with which theranostic agents are discovered, validated, and applied clinically. AI-powered imaging analysis tools are now capable of identifying subtle molecular targets from diagnostic scans with greater sensitivity than traditional reading methods, allowing physicians to better select patients who will benefit most from targeted theranostic therapies. These capabilities are particularly valuable in oncology, where treatment response prediction and dosimetry optimization are critical to achieving optimal patient outcomes.
On the drug development side, machine learning algorithms are being applied to identify novel radioligand and nanoparticle-based theranostic candidates from large biological datasets far more efficiently than conventional screening approaches. AI-driven platforms are also enabling more precise radiation dosimetry calculations in targeted radionuclide therapy, reducing side effects and improving therapeutic windows. As these tools mature and become integrated into clinical workflows, they are expected to meaningfully expand the reach of theranostic medicine into community hospitals and regional cancer centers — not just specialized academic institutions.
Growth Factors
Surging Oncology Disease Burden, Expanding Radiopharmaceutical Approvals, and the Accelerating Global Shift Toward Precision Medicine Are the Core Pillars Driving Theranostics Market Growth
The primary growth driver of the Theranostics market is the extraordinary and growing global burden of cancer. As cancer incidence continues to rise across all major geographies, the clinical need for more targeted, effective, and less toxic treatment strategies is intensifying. Theranostic approaches — which allow the same molecular target to be used for both imaging and therapy — offer precisely this advantage, enabling physicians to confirm target expression before committing a patient to treatment and to monitor therapeutic response in real time. This capability is driving increasing adoption across oncology centers worldwide.
Regulatory milestones are also playing a pivotal role. The FDA's approval of several landmark radiopharmaceutical theranostic agents in recent years — particularly in prostate cancer and neuroendocrine tumors — has validated the clinical and commercial viability of this approach and catalyzed a wave of investment and pipeline development. Pharmaceutical companies are now actively building theranostic portfolios, and a growing number of radioligand therapy programs are advancing through late-stage clinical trials, with several additional approvals expected during the 2026–2033 forecast period. This regulatory momentum is one of the most important structural tailwinds supporting the market's double-digit growth trajectory.

Market Outlook
With a Deep and Maturing Clinical Pipeline, Expanding Manufacturing Infrastructure, and Growing Payer Acceptance, the Theranostics Market Is Positioned for a Decade of Strong and Diversifying Growth
The long-term outlook for the Theranostics market is highly favorable. The forecast period from 2026 to 2033 is expected to see a significant broadening of the theranostic application landscape beyond its current oncology core. Researchers are actively investigating theranostic platforms in cardiology, neurology, and inflammatory disease — and early clinical data in these areas is promising. As the scientific and clinical evidence base expands, payer acceptance and reimbursement coverage for theranostic procedures is also expected to improve, removing a key barrier to broader adoption in both developed and emerging markets.
The scaling of radiopharmaceutical manufacturing infrastructure is another critical element of the market's future trajectory. Major pharmaceutical companies and specialized contract manufacturing organizations are investing heavily in new production facilities for short-lived radionuclides and radioligand therapies. This infrastructure build-out is essential to meeting the anticipated surge in clinical demand and will also help reduce per-unit costs over time, improving accessibility. The combination of deepening clinical adoption, an expanding pipeline, and growing manufacturing capacity makes the Theranostics market one of the most compelling growth stories in the global healthcare sector through 2033.
Expert Speaks
-
Emma Walmsley, CEO, GSK — "Precision medicine, including theranostic approaches that combine diagnosis and therapy, is central to the future of oncology care. We are investing significantly in building radioligand therapy capabilities as part of our long-term oncology strategy, and the clinical results we are seeing are genuinely encouraging."
-
Christoph Franz, Chairman, Roche — "The theranostic model represents exactly the kind of integrated, personalized approach that modern medicine needs. Our diagnostics and pharmaceutical divisions working together on companion diagnostic and targeted therapy combinations is a perfect example of how this framework creates real value for patients."
-
David Ricks, CEO, Eli Lilly — "Targeted therapies that can be guided and monitored by molecular imaging are a natural evolution of precision oncology. We see the theranostics framework as increasingly relevant to our pipeline strategy, particularly as we build out capabilities in radioligand therapy and next-generation molecular targeting."
Key Report Takeaways
-
North America dominates the Theranostics market, accounting for approximately 42.30% of global market share in 2025, supported by a robust regulatory framework, high cancer incidence rates, strong pharmaceutical R&D investment, and well-developed nuclear medicine infrastructure across major hospital networks in the U.S. and Canada.
-
Asia Pacific is the fastest-growing regional market, expanding at an estimated CAGR of approximately 13.20% from 2026 to 2033, driven by rapidly rising cancer burden, expanding nuclear medicine departments, increasing government healthcare investment, and a growing number of clinical trial programs in China, Japan, South Korea, and India.
-
Oncology is the dominant application segment, contributing approximately 68.40% of total market revenue in 2025, as cancer remains the primary clinical domain for theranostic agents, with prostate cancer and neuroendocrine tumor programs representing the largest current commercial applications.
-
Nuclear medicine and radiopharmaceutical-based theranostics is the leading technology segment, commanding approximately 54.70% of market share in 2025, owing to the clinical maturity of radioligand therapy pairs such as PSMA-targeted diagnostics and therapeutics and SSTR-targeted somatostatin analogs.
-
Hospitals and specialty clinics are the primary end-user segment, accounting for approximately 46.20% of total revenue in 2025, as these settings house the nuclear medicine and radiation oncology departments where the majority of clinical theranostic procedures are performed.
-
Nanoparticle-based theranostics is the fastest-growing technology sub-segment, projected to expand at a CAGR of approximately 14.30% from 2026 to 2033, driven by intense academic and pharmaceutical R&D interest in engineered nanocarriers that simultaneously deliver imaging contrast agents and therapeutic payloads to tumor sites with high specificity.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 14.50 Billion |
| Market Size by 2025 | USD 4.60 Billion |
| Market Size by 2026 | USD 5.20 Billion |
| Market Growth Rate (2026–2033) | CAGR of 11.50% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Segments Covered | Type, Technology, Application, End User |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
FDA Approvals of Radioligand Therapies, Rising Cancer Incidence, and Growing Investment in Nuclear Medicine Infrastructure Are Collectively Accelerating the Theranostics Market's Expansion
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Regulatory approvals of radiopharmaceutical theranostic agents | ~34% | North America, Europe | Short to Long-term |
| Rising global cancer incidence and oncology treatment demand | ~30% | Global | Short to Long-term |
| Growing adoption of precision and personalized medicine | ~22% | North America, Europe, Asia Pacific | Medium to Long-term |
| Expanding nuclear medicine infrastructure globally | ~14% | Asia Pacific, Europe, Latin America | Medium-term |
The series of regulatory approvals for radiopharmaceutical theranostic pairs in oncology — particularly for prostate-specific membrane antigen (PSMA)-targeted agents in prostate cancer and somatostatin receptor (SSTR)-targeted agents in neuroendocrine tumors — has been the most catalytic single driver of the Theranostics market in recent years. These approvals have not only validated the clinical model but have also triggered a wave of pharmaceutical investment in building radioligand therapy manufacturing capacity, clinical trial programs, and commercial infrastructure. The established reimbursement pathways being created in the wake of these approvals are also removing economic barriers for patients and providers, accelerating broad clinical uptake.
Rising global cancer incidence continues to generate an ever-expanding patient population for theranostic interventions. The WHO estimates that global cancer cases will continue to rise significantly over the next two decades, driven by population aging, lifestyle factors, and environmental exposures. This demographic and epidemiological reality creates a durable, structural demand base for theranostic diagnostics and therapeutics that is not subject to the cyclical fluctuations that can affect other healthcare market segments. The Theranostics market benefits from this steady demand foundation while also gaining from pharmaceutical industry investment cycles and improving reimbursement environments.

Restraints Impact Analysis
High Production Costs for Radiotheranostic Agents, Complex Regulatory Pathways, and Limited Access to Nuclear Medicine Infrastructure Remain Meaningful Barriers to Theranostics Market Expansion
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High manufacturing cost and complexity of radiopharmaceuticals | ~35% | Global — disproportionate in emerging markets | Short to Medium-term |
| Limited nuclear medicine infrastructure in developing regions | ~28% | Latin America, MEA, parts of Asia Pacific | Medium-term |
| Complex regulatory requirements for combined diagnostic-therapeutic agents | ~25% | North America, Europe | Medium to Long-term |
| Short shelf life of radionuclides creating logistics challenges | ~12% | Global | Short-term |
One of the most significant structural restraints facing the Theranostics market is the high cost and technical complexity of manufacturing radiopharmaceutical agents. Short-lived radionuclides require on-site or near-site production and rapid distribution logistics, making supply chains inherently complex and capital-intensive. This limits market accessibility primarily to large, well-funded hospital networks and specialized treatment centers, creating geographic and institutional disparities in theranostic availability that prevent the market from reaching its full potential patient population in the near term.
The regulatory pathway for combined diagnostic-therapeutic agents is also more complex and time-consuming than for standalone diagnostics or therapeutics. Regulatory bodies require demonstration of both diagnostic accuracy and therapeutic efficacy, as well as appropriate dosimetry data and safety profiles, which increases development costs and timelines. In developing regions, the lack of nuclear medicine cyclotrons, radiopharmacy infrastructure, and trained nuclear medicine physicians further constrains market penetration, creating a meaningful gap between the clinical potential of theranostics and its actual accessibility in lower- and middle-income healthcare settings.
Opportunities Impact Analysis
Expanding Theranostic Applications Beyond Oncology, Rising Nanoparticle Research Investment, and Emerging Market Healthcare Development Are Creating Substantial Untapped Growth Opportunities
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Expansion of theranostic applications into cardiology and neurology | ~36% | North America, Europe | Medium to Long-term |
| Nanoparticle-based theranostic platform development | ~30% | North America, Europe, Asia Pacific | Medium to Long-term |
| Healthcare infrastructure investment in emerging markets | ~22% | Asia Pacific, Latin America, MEA | Short to Medium-term |
| Growing CRO and CDMO involvement in radiopharmaceutical development | ~12% | Global | Short-term |
The expansion of the Theranostics market beyond oncology into cardiovascular and neurological disease represents perhaps the most significant medium-to-long-term commercial opportunity. Theranostic agents targeting amyloid plaques in Alzheimer's disease, inflammation markers in atherosclerosis, and fibrosis pathways in cardiac disease are in active preclinical and early clinical development. Successful clinical validation in even one of these high-prevalence disease areas could add an entirely new, large commercial dimension to the market, substantially expanding the addressable patient population beyond the oncology-focused base that currently dominates revenue.
Nanoparticle-based theranostic platforms are simultaneously creating a parallel innovation track with immense commercial potential. Engineered nanoparticles — including lipid nanoparticles, polymeric nanoparticles, and inorganic nanostructures — can be designed to carry imaging agents and therapeutic payloads simultaneously, offering versatile, modular platforms that can be adapted to multiple targets and disease indications. Academic institutions and startups in North America, Europe, and Asia Pacific are producing a growing body of preclinical evidence supporting nanoparticle-based theranostic efficacy, and pharmaceutical companies are beginning to acquire or partner with these innovators to build next-generation theranostic portfolios.

Segment Analysis
By Technology
Nuclear Medicine and Radiopharmaceutical Theranostics Leads With Clinical Maturity, While Nanoparticle-Based Platforms Rapidly Emerge as the Most Innovative and Fastest-Growing Technology Segment
Nuclear medicine and radiopharmaceutical-based theranostics commands the largest share of the technology segment, accounting for approximately 54.70% of global market revenue in 2025. This segment's leadership is built on decades of clinical experience with radiotracer diagnostics and the recent landmark approvals of radioligand therapies in prostate cancer and neuroendocrine tumors, which have firmly established radiopharmaceutical theranostic pairs as a standard-of-care option in these indications. North America is the dominant region for this technology segment, where major cancer centers and academic hospitals have the nuclear medicine infrastructure, regulatory support, and clinical expertise to deploy these therapies at scale. Novartis — through its Endocyte and Advanced Accelerator Applications acquisitions — is the clear global leader in this segment, with its PSMA-targeted and SSTR-targeted theranostic products generating substantial commercial revenue. The segment is growing at a healthy CAGR supported by a pipeline of new radioligand therapy programs targeting additional solid tumor types including breast cancer, glioblastoma, and hepatocellular carcinoma.
Nanoparticle-based theranostics is the technology segment generating the most scientific excitement and investment, with a projected CAGR of approximately 14.30% from 2026 to 2033. This sub-segment encompasses a wide range of engineered nanocarrier platforms — including iron oxide nanoparticles for MRI-guided therapy, gold nanoparticles for photothermal theranostics, and lipid nanoparticles for combined nucleic acid therapy and imaging. Asia Pacific, particularly China and South Korea, is emerging as a key innovation hub for nanoparticle-based Theranostics, with prolific academic research output and a growing number of startups translating laboratory discoveries into clinical-stage programs. Companies such as Nanobiotix (France) and a number of Chinese nanoparticle biotechnology firms are among the most active players developing commercially viable nanoparticle theranostic products, with several candidates progressing through early to mid-stage clinical trials during the current forecast period.
By Application
Oncology Dominates as the Definitive Application Segment for Theranostic Approaches, With Prostate Cancer and Neuroendocrine Tumor Programs Anchoring Commercial Revenue Globally
Oncology is the overwhelming leader in the application segment, contributing approximately 68.40% of total Theranostics market revenue in 2025. The dominance of oncology reflects the fact that cancer has both the greatest clinical need for targeted, personalized treatment approaches and the most advanced theranostic science base, with well-characterized molecular targets such as PSMA, SSTR, HER2, and EGFR that can serve as anchors for combined diagnostic-therapeutic agent design. North America holds the largest oncology theranostics market share globally, driven by high cancer incidence, strong healthcare spending, established nuclear medicine infrastructure, and active clinical trial enrollment at major NCI-designated cancer centers. Novartis, Lantheus Holdings, and Point Biopharma are among the key companies generating significant commercial oncology theranostics revenue in the region, with robust sales of approved PSMA and SSTR theranostic agents. This segment is growing at a CAGR consistent with the overall market, sustained by new indication expansions, label approvals, and increasing real-world adoption at community oncology centers.
Cardiology is the fastest-growing application segment beyond oncology, attracting increasing research and early clinical investment as the theranostic framework is applied to cardiovascular disease management. Molecular imaging of atherosclerotic plaque inflammation, cardiac fibrosis, and vulnerable plaque characteristics using theranostic agents that can simultaneously deliver local therapy is a compelling and actively investigated clinical concept. Europe is a key region for early-stage cardiovascular theranostics research, with several leading academic centers in Germany, the Netherlands, and the U.K. running investigator-initiated clinical programs. The convergence of cardiology's large patient population, significant unmet therapeutic need, and the availability of well-established cardiac imaging infrastructure creates a promising long-term growth pathway for the application of Theranostics beyond oncology.

Regional Insights
North America
North America Holds Commanding Leadership in the Global Theranostics Market, Anchored by Landmark Regulatory Approvals, World-Class Cancer Centers, and Dominant Pharmaceutical Investment
North America accounts for approximately 42.30% of global Theranostics market share in 2025 and is projected to maintain its leadership position throughout the forecast period at a CAGR of approximately 10.80% from 2026 to 2033. The United States is the primary revenue contributor, driven by the FDA's approvals of landmark radioligand therapies, the highest per-capita oncology spending globally, and the presence of major pharmaceutical and radiopharmaceutical companies that are actively scaling theranostic commercial operations. Leading companies with strong North American presence and operations include Novartis, Lantheus Holdings, Bristol-Myers Squibb, Eli Lilly, and Point Biopharma — all of which are either commercializing approved theranostic agents or advancing late-stage pipeline programs. The U.S. also hosts the majority of global theranostic clinical trials, reinforcing its position as the primary driver of both scientific progress and commercial adoption in this field.
Canada contributes meaningfully to North America's regional market, with several major academic cancer centers at institutions such as Princess Margaret Cancer Centre and the University of British Columbia operating advanced nuclear medicine and radiopharmacy programs. The region's well-developed reimbursement infrastructure for nuclear medicine procedures is an important enabler of market growth, as it ensures that approved theranostic therapies can reach commercially viable patient volumes. The overall North American Theranostics market is expected to more than double in value between 2025 and 2033, driven by new indication approvals, pipeline maturation, and expanding access to theranostic procedures at community and regional cancer centers beyond the current concentration at major academic institutions.
Asia Pacific
Asia Pacific Is Rapidly Emerging as the Theranostics Market's Fastest-Growing Region, Powered by Rising Cancer Burden, Infrastructure Investment, and a Maturing Nuclear Medicine Ecosystem
Asia Pacific is the fastest-growing regional segment in the Theranostics market, projected to expand at a CAGR of approximately 13.20% from 2026 to 2033 — the highest of any region globally. China is the single largest national market within the region and is experiencing rapid expansion in nuclear medicine departments, radiopharmacy infrastructure, and clinical trial activity in oncology theranostics. The Chinese government has identified nuclear medicine and advanced cancer therapies as strategic healthcare priorities, and both public investment and private pharmaceutical development are accelerating accordingly. Key players operating in Asia Pacific include Siemens Healthineers (Germany, with strong regional presence), Fujifilm Toyama Chemical (Japan), and a growing number of domestic Chinese radiopharmaceutical companies such as HTA Co. Ltd. that are developing theranostic agents specifically for Asian patient populations and market needs.
Japan and South Korea represent the most mature sub-markets within Asia Pacific for Theranostics, where nuclear medicine has a long clinical history and where regulatory pathways for new radiopharmaceutical agents are becoming increasingly streamlined. Japan's aging population and high cancer incidence are creating sustained demand growth, while South Korea's dynamic biotech sector is producing innovative theranostic research programs with global commercialization ambitions. India represents a large and rapidly developing theranostics market, with significant government investment in establishing new nuclear medicine centers under national cancer control programs. The breadth and diversity of growth drivers across Asia Pacific — from infrastructure build-out in developing economies to clinical innovation in advanced ones — makes this region the most dynamic and strategically important growth frontier for theranostics market participants through 2033.
Top Key Players
-
Novartis AG (Switzerland)
-
Lantheus Holdings, Inc. (United States)
-
Siemens Healthineers AG (Germany)
-
General Electric Healthcare (United States)
-
Bristol-Myers Squibb Company (United States)
-
Eli Lilly and Company (United States)
-
Point Biopharma Global Inc. (United States)
-
Nanobiotix S.A. (France)
-
Fusion Pharmaceuticals Inc. (Canada)
-
RadioMedix Inc. (United States)
-
Clarity Pharmaceuticals (Australia)
-
Actinium Pharmaceuticals, Inc. (United States)
-
Blue Earth Diagnostics (United Kingdom)
-
Sofie Biosciences (United States)
Recent Developments
-
Novartis AG (2025) — Expanded its radioligand therapy portfolio with the submission of regulatory applications for a new PSMA-targeted theranostic agent for earlier-stage prostate cancer, building on the commercial success of its existing approved radioligand therapies and reinforcing its position as the global leader in the theranostics segment.
-
Eli Lilly and Company (2024) — Completed the acquisition of Point Biopharma Global Inc. in a transaction valued at approximately USD 1.4 billion, significantly strengthening its radiopharmaceutical theranostics capabilities and manufacturing infrastructure across North America and positioning Lilly as a major competitor in the radioligand therapy space.
-
Lantheus Holdings (2025) — Announced a strategic collaboration with a major European pharmaceutical company to co-develop next-generation theranostic agents targeting novel oncology biomarkers, expanding its pipeline beyond its established PSMA imaging franchise and reinforcing its commitment to the broader theranostics platform.
-
Siemens Healthineers (2024) — Launched an upgraded PET/CT imaging platform with enhanced molecular imaging capabilities specifically optimized for theranostic dosimetry applications, including improved quantification algorithms for radiopharmaceutical biodistribution analysis, targeting the growing clinical demand from oncology centers deploying radioligand therapy programs.
-
Fusion Pharmaceuticals (2024) — Was acquired by AstraZeneca in a deal valued at approximately USD 2.4 billion, marking one of the largest theranostics-focused acquisitions of the year and signaling major pharma's deepening commitment to building radioligand therapy capabilities for a broad range of oncology indications.
Market Trends
Radioligand Therapy Pipeline Expansion and the Emergence of Artificial Intelligence-Guided Theranostic Dosimetry Are the Two Most Consequential Trends Reshaping the Theranostics Market Today
The most significant commercial trend reshaping the Theranostics market is the extraordinary expansion of the radioligand therapy clinical pipeline across multiple tumor types. Following the landmark approvals in prostate cancer and neuroendocrine tumors, pharmaceutical companies are now advancing theranostic programs targeting breast cancer, glioblastoma, colorectal cancer, and hepatocellular carcinoma — among others. This pipeline breadth means that the next five to seven years are likely to see multiple additional approvals, each opening new patient populations and generating new commercial channels. The scale of pharmaceutical investment in radioligand therapy manufacturing — with billions of dollars being committed to new production facilities for actinium-225, lutetium-177, and other therapeutic radionuclides — reflects industry confidence that the theranostic model will become a mainstream pillar of oncology treatment.
A parallel and increasingly important trend is the integration of AI-powered dosimetry and treatment planning tools into clinical theranostic workflows. Precise dosimetry — calculating exactly how much radiation each organ and tumor receives during radioligand therapy — is essential for optimizing therapeutic efficacy and minimizing toxicity, but has historically been complex, time-consuming, and inconsistent across institutions. AI algorithms trained on large clinical imaging and dosimetry datasets are now beginning to automate and standardize this process, making personalized theranostic dosimetry feasible at scale in community hospitals rather than just specialized academic centers. This development is expected to meaningfully expand the addressable patient population for theranostic procedures and accelerate market penetration throughout the 2026–2033 forecast period.
Segments Covered in the Report
By Type:
-
Diagnostics
-
Therapeutics
-
Combined Theranostics
By Technology:
-
Nuclear Medicine / Radiopharmaceuticals
-
Optical Imaging-Based Theranostics
-
Ultrasound-Based Theranostics
-
Nanoparticle-Based Theranostics
-
Other Technologies
By Application:
-
Oncology
-
Cardiology
-
Neurology
-
Orthopedics
-
Other Applications
By End User:
-
Hospitals and Specialty Clinics
-
Diagnostic Imaging Centers
-
Pharmaceutical and Biotechnology Companies
-
Academic and Research Institutes
-
Contract Research Organizations
By Region:
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the Theranostics market and what is its growth forecast through 2033?
Answer: The Theranostics market was valued at USD 4.60 billion in 2025 and is projected to reach USD 14.50 billion by 2033. It is expected to grow at a CAGR of 11.50% from 2026 to 2033, driven by expanding radioligand therapy approvals and rising oncology demand.
Question 2: What are the key factors driving growth in the Theranostics market globally?
Answer: Rising global cancer incidence and landmark FDA approvals of radioligand therapies are the two most powerful drivers of the Theranostics market. Growing adoption of precision medicine frameworks and expanding nuclear medicine infrastructure across emerging markets are also contributing meaningfully to sustained market expansion.
Question 3: Which region leads the Theranostics market and which is growing the fastest?
Answer: North America dominates the Theranostics market with approximately 42.30% of global share in 2025, supported by strong pharmaceutical R&D and regulatory activity. Asia Pacific is the fastest-growing region, expanding at approximately 13.20% CAGR from 2026 to 2033 due to rising cancer burden and healthcare infrastructure investment.
Question 4: Which application and technology segments dominate the Theranostics market?
Answer: Oncology is the leading application segment, contributing approximately 68.40% of total Theranostics market revenue in 2025. Nuclear medicine and radiopharmaceutical-based theranostics is the dominant technology segment, while nanoparticle-based theranostics is the fastest-growing sub-segment at approximately 14.30% CAGR through 2033.
Question 5: Who are the leading companies competing in the Theranostics market?
Answer: Key players in the Theranostics market include Novartis AG, Lantheus Holdings, Eli Lilly, Siemens Healthineers, and Fusion Pharmaceuticals (acquired by AstraZeneca). These companies are driving growth through pipeline expansion, strategic acquisitions, and significant investments in radiopharmaceutical manufacturing infrastructure globally.
