Systemic Mastocytosis Treatment Market Overview
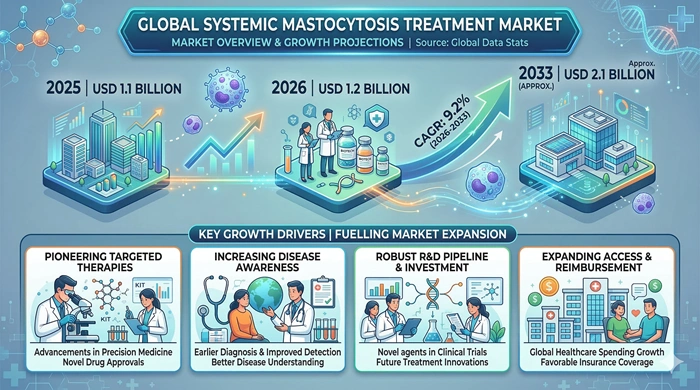
The global systemic mastocytosis treatment market size is valued at USD 1.1 billion in 2025 and is predicted to increase from USD 1.2 billion in 2026 to approximately USD 2.1 billion by 2033, growing at a CAGR of 9.2% from 2026 to 2033.
Systemic mastocytosis is a rare and complex hematological disorder characterized by the abnormal accumulation of clonal mast cells in organs such as the bone marrow, liver, spleen, and skin. The discovery of targeted KIT inhibitors and the growing understanding of the disease's molecular biology have transformed the treatment landscape significantly over the past decade. As pharmaceutical companies continue to invest in rare disease drug development and regulatory agencies expand orphan drug incentives, the systemic mastocytosis treatment market is entering a period of accelerated clinical and commercial progress.
AI Impact on the Systemic Mastocytosis Treatment Industry
Artificial Intelligence Is Accelerating Drug Discovery, Biomarker Identification, and Personalized Treatment Protocols for Systemic Mastocytosis, Reshaping the Future of Rare Disease Oncology Management
Artificial intelligence is playing a growing role in transforming how systemic mastocytosis is diagnosed and treated. AI-driven genomic analysis tools are helping researchers identify novel mutations beyond the well-known KIT D816V variant, enabling pharmaceutical scientists to design next-generation targeted therapies with greater precision and a broader patient applicability. Machine learning algorithms are also being applied to clinical trial data to identify patient subgroups most likely to respond to specific KIT inhibitor regimens, improving trial design efficiency and accelerating the time from drug discovery to regulatory submission in this rare disease space.
Beyond drug development, AI is improving diagnostic accuracy and treatment monitoring for patients with systemic mastocytosis. Advanced imaging analysis platforms integrated with AI can detect subtle changes in bone marrow mast cell burden earlier than conventional methods, supporting timely treatment adjustments. Natural language processing tools are helping clinicians mine electronic health records for symptom patterns associated with disease progression, enabling more proactive clinical management. As AI capabilities continue to mature within rare disease medicine, they are expected to meaningfully reduce the diagnostic odyssey that many systemic mastocytosis patients experience before receiving an accurate diagnosis and appropriate therapy.
Growth Factors
Breakthrough Targeted Therapies, Expanding Orphan Drug Designations, and Rising Rare Disease Awareness Are the Core Pillars Fueling Growth in the Systemic Mastocytosis Treatment Market
The most transformative growth driver in the systemic mastocytosis treatment market is the commercial success and clinical validation of targeted KIT inhibitors. Avapritinib, approved by the FDA in 2021 for advanced systemic mastocytosis, represented a landmark in rare disease oncology, delivering response rates far superior to prior standard-of-care therapies. Its continued commercial uptake, combined with expanding label indications and ongoing Phase III trials exploring its utility in indolent forms of the disease, is directly driving prescription volumes and market revenue growth. Similarly, midostaurin — the first approved therapy for advanced systemic mastocytosis — continues to contribute meaningfully to the treatment revenue base in markets where avapritinib access remains limited.
The broader rare disease drug development ecosystem is also creating powerful tailwinds for this market. Regulatory incentives including orphan drug designation, priority review vouchers, and accelerated approval pathways in the United States and Europe have significantly lowered the commercial risk for companies investing in systemic mastocytosis therapies. Growing patient advocacy organizations are increasing disease awareness among both patients and physicians, shortening diagnostic delays and expanding the diagnosed patient population. As more patients receive timely diagnoses and are channeled into appropriate specialty care pathways, the addressable market for systemic mastocytosis treatment continues to broaden meaningfully.

Market Outlook
The Systemic Mastocytosis Treatment Market Is Poised for Strong Expansion Through 2033, Driven by a Deep Clinical Pipeline, Expanding Treatment Access, and Increasing Diagnosis Rates Globally
The outlook for the systemic mastocytosis treatment market through 2033 is compelling and well-supported by multiple converging forces. A robust clinical pipeline featuring next-generation KIT inhibitors, combination therapy protocols, and novel immunotherapeutic approaches is expected to deliver multiple new treatment approvals over the forecast period. These innovations will not only expand therapeutic options for patients with advanced and aggressive disease but are also expected to address the large unmet need in indolent systemic mastocytosis — a patient population that has historically had limited pharmacological options beyond symptom management.
Geographic expansion of treatment access represents another major driver of long-term market growth. While North America currently dominates clinical and commercial activity, European markets are rapidly catching up as healthcare systems incorporate KIT inhibitors into national formularies and reimbursement frameworks. Emerging markets in Asia Pacific are also expected to contribute incrementally as rare disease awareness grows and specialized hematology centers expand their diagnostic and treatment capabilities. Overall, the systemic mastocytosis treatment market is entering a phase where science, regulatory support, and commercial infrastructure are all aligned to drive sustained revenue growth through the end of the forecast period.
Expert Speaks
-
Blueprint Medicines CEO Kate Haviland stated that the rare disease community has never been better positioned to deliver transformative therapies for conditions like systemic mastocytosis, and that Blueprint's ongoing commitment to precision medicine is centered on reaching every patient who can benefit from highly selective KIT inhibition, regardless of disease subtype.
-
Novartis CEO Vas Narasimhan emphasized that targeted therapies for rare hematological disorders are a strategic priority for Novartis, and that the clinical outcomes delivered by midostaurin in systemic mastocytosis patients have reinforced the company's belief that precision oncology approaches can fundamentally change the trajectory of rare blood diseases.
-
Pfizer CEO Albert Bourla noted that the expanding landscape of approved rare disease treatments, including therapies for advanced systemic mastocytosis, reflects the pharmaceutical industry's deepening commitment to patient populations that were historically overlooked, and that continued investment in rare oncology drug development remains a core part of Pfizer's long-term growth strategy.
Key Report Takeaways
-
North America leads the systemic mastocytosis treatment market, holding the largest revenue share driven by early access to approved KIT inhibitors, strong hematology specialty infrastructure, and the highest rate of diagnosed systemic mastocytosis patients globally.
-
Europe is the fastest-growing region, as healthcare systems across Germany, France, and the United Kingdom accelerate reimbursement approvals for targeted systemic mastocytosis therapies and rare disease clinical trial activity intensifies across the continent.
-
Hospitals and specialty hematology clinics are the dominant end users, managing the vast majority of diagnosed systemic mastocytosis cases due to the complexity of disease management and the need for specialist-level monitoring and drug administration.
-
Targeted therapy — particularly KIT inhibitors — is the dominant treatment type, accounting for the largest share of market revenue, driven by the clinical superiority of avapritinib and midostaurin over conventional chemotherapy for advanced systemic mastocytosis.
-
Avapritinib (Blueprint Medicines) is the most clinically adopted targeted agent, recognized for its high selectivity for KIT D816V-mutant mast cells and its superior clinical response rates across multiple systemic mastocytosis subtypes.
-
Indolent systemic mastocytosis is the fastest-growing disease type segment, expected to grow at a CAGR above the market average through 2033, with approximately 19% market share, as clinical trials actively evaluate avapritinib and novel agents in this large but historically undertreated patient population.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 2.1 Billion |
| Market Size by 2025 | USD 1.1 Billion |
| Market Size by 2026 | USD 1.2 Billion |
| Market Growth Rate (2026–2033) | CAGR of 9.2% |
| Dominating Region | North America |
| Fastest Growing Region | Europe |
| Base Year | 2025 |
| Forecast Period | 2026 to 2033 |
| Segments Covered | Treatment Type, Disease Type, End User |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
FDA-Approved KIT Inhibitors, Orphan Drug Incentives, and Growing Rare Disease Patient Advocacy Are the Most Powerful Drivers Propelling the Systemic Mastocytosis Treatment Market Forward
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Commercial success of avapritinib and midostaurin | ~34% | North America, Europe | Near to Long-term |
| Expanding orphan drug designations and regulatory support | ~26% | Global | Near to Mid-term |
| Growing rare disease awareness and patient advocacy | ~20% | North America, Europe | Mid to Long-term |
| Increasing diagnosis rates through improved testing | ~13% | Global | Mid-term |
| Rising investment in rare hematological drug R&D | ~7% | North America, Europe | Long-term |
The commercial breakthrough achieved by avapritinib has fundamentally reshaped the systemic mastocytosis treatment market. Its highly selective mechanism of action against the KIT D816V mutation — present in over 95% of systemic mastocytosis patients — delivers response rates that were previously unattainable with non-selective agents. The drug's approval has validated the commercial viability of the systemic mastocytosis space and has encouraged additional pharmaceutical investment in both competing KIT inhibitors and combination regimens designed to deepen and prolong clinical responses in advanced disease patients.
Regulatory incentives are amplifying this commercial momentum significantly. In the United States, orphan drug designation provides seven years of market exclusivity, reducing competitive pressure and extending the revenue window for approved therapies. Priority review and breakthrough therapy designations have also accelerated the development timelines of multiple pipeline agents targeting systemic mastocytosis. These policy frameworks, combined with growing patient advocacy organizations such as The Mastocytosis Society that are actively shortening the average diagnostic delay, are collectively expanding the diagnosed and treated patient population that forms the revenue foundation of this market.

Restraints Impact Analysis
Extremely Small Patient Population, High Drug Pricing Pressure, and Complex Differential Diagnosis Continue to Constrain Broader Market Expansion for Systemic Mastocytosis Treatments
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Very small and heterogeneous patient population | ~38% | Global | Ongoing |
| High cost of targeted therapies and reimbursement barriers | ~30% | Emerging Markets, Global | Near to Mid-term |
| Complex diagnosis and prolonged diagnostic delays | ~20% | Global, especially Emerging Markets | Ongoing |
| Limited specialist hematology access in rural areas | ~12% | Global | Long-term |
The exceptionally rare nature of systemic mastocytosis remains the most fundamental structural constraint on market size. With a global prevalence estimated at fewer than 1 in 10000 people, the overall patient pool is extremely limited, capping the maximum addressable market for any single systemic mastocytosis treatment. While the approved drugs command premium pricing justified by orphan drug status and clinical value, the small patient numbers mean that even high per-patient revenue cannot generate blockbuster-scale market volumes comparable to more common oncology indications.
Diagnosis complexity compounds this challenge in a meaningful way. Systemic mastocytosis presents with a wide range of non-specific symptoms — including skin lesions, gastrointestinal distress, anaphylaxis, and bone pain — that overlap with numerous other conditions, leading to diagnostic delays that commonly exceed five to ten years. During this period, patients are often misdiagnosed and treated for other conditions without receiving the targeted therapies that could meaningfully improve their disease course. Expanding access to sensitive diagnostic tools, including next-generation sequencing for KIT D816V detection and sensitive serum tryptase testing, is critical to addressing this bottleneck and growing the treated patient population in this market.
Opportunities Impact Analysis
Indolent Systemic Mastocytosis Drug Development, Combination Therapy Innovation, and Asia Pacific Market Entry Present the Most Significant Untapped Opportunities in This Market
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Clinical development for indolent systemic mastocytosis | ~35% | North America, Europe | Mid to Long-term |
| Combination therapy protocols with KIT inhibitors | ~28% | Global | Mid to Long-term |
| Expanding reimbursement in European markets | ~22% | Europe | Near to Mid-term |
| Emerging market disease awareness and diagnosis growth | ~15% | Asia Pacific, Latin America | Long-term |
The indolent systemic mastocytosis segment represents perhaps the largest untapped commercial opportunity in the entire market. With tens of thousands of patients globally suffering from chronic symptoms without access to disease-modifying therapy, the potential approval of avapritinib or novel KIT inhibitors for this indication would dramatically expand the treatable patient population. Blueprint Medicines and other developers are actively pursuing clinical data to support regulatory submissions for indolent disease, and positive trial outcomes could unlock a patient population several times larger than the current advanced disease market, fundamentally changing the commercial scale of the systemic mastocytosis treatment landscape.
Combination therapy development is creating additional opportunities for revenue growth and clinical differentiation. Preclinical and early clinical data suggest that combining KIT inhibitors with agents targeting complementary disease mechanisms — such as BET inhibitors, BCL-2 inhibitors, or immunotherapy — may achieve deeper and more durable responses than monotherapy alone, particularly in mast cell leukemia and systemic mastocytosis with associated hematological neoplasm. Companies investing in combination regimen development are positioning themselves to capture a larger share of the high-acuity, high-value patient segments within this market as evidence matures and clinical practice guidelines evolve.

Segment Analysis
By Treatment Type
Targeted Therapy — Particularly Highly Selective KIT Inhibitors — Dominates the Systemic Mastocytosis Treatment Market, Delivering Unprecedented Clinical Response Rates and Reshaping the Standard of Care
Targeted therapy accounts for approximately 58% of the total systemic mastocytosis treatment market revenue and represents the fastest-growing and most commercially significant treatment segment throughout the forecast period. This category is dominated by avapritinib and midostaurin, both of which have demonstrated clinically meaningful reductions in mast cell burden and symptom improvement in patients with advanced and aggressive disease. The targeted therapy segment is expected to grow at a CAGR of approximately 10.1% through 2033, driven by expanding clinical use, label extensions into earlier disease stages, and a pipeline of next-generation KIT inhibitors entering late-stage clinical development. North America leads this segment, where robust insurance reimbursement frameworks and high specialty physician density support broad patient access. Blueprint Medicines and Novartis are the primary commercial players in this segment, with Blueprint's avapritinib increasingly becoming the preferred first-line choice for advanced systemic mastocytosis among U.S. hematologists.
Europe is also a high-growth geography for targeted therapy adoption within the systemic mastocytosis treatment market, driven by accelerating regulatory approvals and expanding national formulary inclusions across Germany, France, the United Kingdom, and Italy. The European Medicines Agency has been proactive in granting conditional and accelerated approvals for KIT inhibitors in rare hematological indications, reducing the gap between U.S. and European patient access. Regional academic centers of excellence in mastocytosis — including those affiliated with the European Competence Network on Mastocytosis — are playing a key role in generating real-world evidence that supports payer reimbursement decisions and physician treatment adoption. This growing clinical infrastructure in Europe is expected to drive targeted therapy segment revenue at a CAGR meaningfully above the overall market average through 2033.
By Disease Type
Advanced Systemic Mastocytosis Remains the Highest-Revenue Disease Segment, While Indolent Systemic Mastocytosis Emerges as the Fastest-Growing and Most Commercially Promising Category
Advanced systemic mastocytosis — encompassing aggressive systemic mastocytosis, mast cell leukemia, and systemic mastocytosis with associated hematological neoplasm — currently contributes the largest share of the systemic mastocytosis treatment market revenue at approximately 44%. These aggressive subtypes carry the most severe prognosis and the greatest unmet treatment need, justifying the use of high-cost targeted therapies that drive disproportionately high per-patient revenue. The advanced disease segment is expected to maintain steady growth at a CAGR of approximately 8.7% through 2033, underpinned by expanding KIT inhibitor use and the entry of novel combination regimens into clinical practice. North America and Europe together account for the vast majority of diagnosed and treated advanced systemic mastocytosis patients, with Blueprint Medicines, Novartis, and AB Science maintaining the strongest commercial footprints in these geographies.
Indolent systemic mastocytosis is the fastest-growing disease type segment, expected to register a CAGR of approximately 10.8% through 2033 and holding approximately 19% of total market share in 2026. While this patient population currently has limited approved pharmacological options, ongoing Phase III clinical trials evaluating avapritinib in indolent disease are anticipated to generate pivotal data within the forecast period, potentially unlocking regulatory approvals that would transform the commercial size of this segment. The patient population with indolent systemic mastocytosis is significantly larger than that with advanced disease, meaning a single approval in this indication could more than double the total addressable market for targeted therapies in systemic mastocytosis globally.

Regional Insights
North America
North America Leads the Global Systemic Mastocytosis Treatment Market With the Highest Diagnosis Rates, Most Approved Therapies, and the Strongest Rare Disease Commercial Infrastructure in the World
North America holds approximately 46% of the global systemic mastocytosis treatment market share and is projected to maintain its dominant position through 2033, growing at a CAGR of approximately 8.9%. The United States is the cornerstone of this leadership, driven by the early commercial availability of both avapritinib and midostaurin, strong hematology-oncology referral networks, and a well-funded rare disease ecosystem that actively supports patient identification, diagnosis, and treatment initiation. The U.S. FDA's orphan drug framework continues to incentivize domestic pharmaceutical investment in systemic mastocytosis, with multiple clinical-stage companies headquartered in the United States. Key players in this region include Blueprint Medicines (U.S.), Novartis (Switzerland, with major U.S. commercial operations), and Cogent Biosciences (U.S.).
Canada contributes meaningfully to North America's regional leadership through its expanding rare disease drug reimbursement programs and growing hematology research infrastructure. Canadian academic medical centers are active participants in multinational systemic mastocytosis clinical trials, and Health Canada has been progressively aligning its rare disease approval pathways with FDA standards. Together, the U.S. and Canada provide a mature and commercially well-supported environment for systemic mastocytosis treatment that will continue to attract pharmaceutical investment and clinical research activity throughout the forecast period.
Europe
Europe Is the Fastest-Growing Region in the Systemic Mastocytosis Treatment Market, Driven by Accelerating Reimbursement Approvals, Strong Academic Research Networks, and the World's Most Active Mastocytosis Patient Registry Infrastructure
Europe is the fastest-growing region in the systemic mastocytosis treatment market, expected to register a CAGR of approximately 10.3% through 2033. The continent benefits from a dense network of specialized mastocytosis centers affiliated with the European Competence Network on Mastocytosis, which has been instrumental in standardizing diagnosis, treatment protocols, and patient data collection across member states. Germany, France, Spain, and the Netherlands are among the leading European markets where KIT inhibitors have received national reimbursement approval, enabling broader patient access to targeted therapies. Key companies with strong European presence include Novartis (Switzerland), AB Science (France), and Blueprint Medicines, which has been actively expanding its European commercial infrastructure to support avapritinib adoption.
The European Medicines Agency's progressive stance on rare disease approvals and its well-established adaptive pathways framework are enabling faster market entry for novel systemic mastocytosis therapies compared to previous regulatory cycles. European payers are also increasingly accepting real-world evidence generated through the continent's robust patient registries as supplementary support for reimbursement decisions, reducing the post-approval market access timeline. This combination of clinical expertise, regulatory efficiency, and growing payer alignment is positioning Europe as the most dynamic growth market globally for systemic mastocytosis treatment through the end of the forecast period.
Top Key Players
-
Blueprint Medicines Corporation (United States)
-
Novartis AG (Switzerland)
-
AB Science S.A. (France)
-
Cogent Biosciences Inc. (United States)
-
Deciphera Pharmaceuticals (United States)
-
Syndax Pharmaceuticals (United States)
-
Stemline Therapeutics (Menarini Group) (United States / Italy)
-
Pfizer Inc. (United States)
-
Bristol Myers Squibb (United States)
-
AbbVie Inc. (United States)
-
Celldex Therapeutics (United States)
-
Daiichi Sankyo Co. Ltd. (Japan)
Recent Developments
-
Blueprint Medicines (2025): Blueprint Medicines announced positive topline results from its PIONEER Phase III trial evaluating avapritinib in patients with indolent systemic mastocytosis, demonstrating statistically significant and clinically meaningful reductions in total symptom score compared to placebo, potentially supporting a new FDA supplemental approval in this larger patient population.
-
Cogent Biosciences (2024): Cogent Biosciences reported encouraging Phase II clinical data for bezuclastinib, its highly selective KIT D816V inhibitor, in patients with advanced systemic mastocytosis, with the drug demonstrating strong response rates and a differentiated safety profile, and advanced the program into a Phase III pivotal trial.
-
Novartis (2024–2025): Novartis continued to expand midostaurin's commercial reach in key European and Asian markets through new national reimbursement agreements, reinforcing its position as a foundational systemic mastocytosis treatment option in geographies where next-generation KIT inhibitors have not yet received regulatory approval.
-
AB Science (2024): AB Science reported updated clinical data from its masitinib trials in indolent systemic mastocytosis, showing sustained symptom control benefits in a long-term follow-up analysis, and continued to pursue regulatory submissions in key European markets to secure commercial approval for this patient population.
-
Deciphera Pharmaceuticals (2025): Deciphera Pharmaceuticals, following its acquisition by Winnebago Industries' healthcare division, continued to advance its KIT inhibitor pipeline for mast cell disorders, with preclinical data supporting differentiated activity against multiple KIT mutation variants beyond D816V, potentially expanding the addressable patient population in future clinical development stages.
Market Trends
Next-Generation KIT Inhibitors, Combination Therapy Protocols, and the Expansion of Systemic Mastocytosis Treatment Into Earlier Disease Stages Are the Defining Trends Shaping This Market Through 2033
The most consequential trend reshaping the systemic mastocytosis treatment market is the rapid advancement of next-generation, highly selective KIT D816V inhibitors in clinical development. While avapritinib has established a high clinical bar, compounds such as bezuclastinib are demonstrating differentiated pharmacological profiles — including improved CNS penetration and potentially more favorable drug interaction profiles — that could offer meaningful clinical advantages for specific patient subpopulations. The competitive intensification within the KIT inhibitor class is expected to drive pricing pressure while simultaneously improving patient outcomes through broader prescriber choice and enhanced clinical trial options for patients who have progressed on current standard-of-care therapies.
Simultaneously, the field is moving meaningfully toward treating systemic mastocytosis earlier in its disease course. The historically reactive treatment paradigm — where pharmacological intervention was largely reserved for advanced or aggressive disease — is being challenged by accumulating evidence that targeted KIT inhibition in indolent systemic mastocytosis can substantially reduce symptom burden, improve quality of life, and potentially slow disease progression. This paradigm shift, if validated by ongoing Phase III trials, would fundamentally expand the treated patient population and the commercial scope of the systemic mastocytosis treatment market, while also driving demand for more sophisticated diagnostic tools that can identify patients earlier in their disease trajectory.
Segments Covered in the Report
By Treatment Type
-
Targeted Therapy
-
KIT Inhibitors
-
Midostaurin
-
Avapritinib
-
Chemotherapy
-
Immunotherapy
-
Stem Cell Transplantation
-
Supportive Care
-
Antihistamines
-
Corticosteroids
-
Epinephrine
-
Others
By Disease Type
-
Indolent Systemic Mastocytosis
-
Smoldering Systemic Mastocytosis
-
Aggressive Systemic Mastocytosis
-
Mast Cell Leukemia
-
Systemic Mastocytosis with Associated Hematological Neoplasm
By End User
-
Hospitals & Specialty Clinics
-
Academic & Research Institutes
-
Ambulatory Surgical Centers
-
Others
By Region
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the global systemic mastocytosis treatment market and what growth is projected by 2033?
Answer: The global systemic mastocytosis treatment market is valued at USD 1.1 billion in 2025 and is projected to reach USD 2.1 billion by 2033. It is expected to grow at a CAGR of 9.2% from 2026 to 2033.
Question 2: What are the key drivers of growth in the systemic mastocytosis treatment market?
Answer: The primary growth drivers include the commercial success of approved KIT inhibitors such as avapritinib and midostaurin and the expanding pipeline of next-generation targeted agents. Favorable orphan drug regulatory incentives and growing rare disease awareness are also accelerating market expansion significantly.
Question 3: Which treatment type dominates the systemic mastocytosis treatment market?
Answer: Targeted therapy — specifically KIT inhibitors — dominates the systemic mastocytosis treatment market, accounting for approximately 58% of total market revenue. Avapritinib has become the preferred standard of care for advanced systemic mastocytosis due to its highly selective mechanism of action and superior clinical response rates.
Question 4: Which region leads the systemic mastocytosis treatment market and which is growing the fastest?
Answer: North America currently leads the systemic mastocytosis treatment market with approximately 46% of global revenue share. Europe is the fastest-growing region, projected to register a CAGR of approximately 10.3% through 2033, driven by accelerating reimbursement approvals and a strong academic mastocytosis research network.
Question 5: What is the difference between indolent and aggressive systemic mastocytosis in the context of treatment market opportunities?
Answer: Aggressive systemic mastocytosis currently generates the highest treatment revenue due to the use of high-cost targeted therapies in a severely ill patient population. Indolent systemic mastocytosis represents the fastest-growing opportunity, as ongoing clinical trials evaluating targeted therapy in this larger but historically undertreated patient group could dramatically expand the overall systemic mastocytosis treatment market upon potential approval.
