Precision Medicine Market Overview
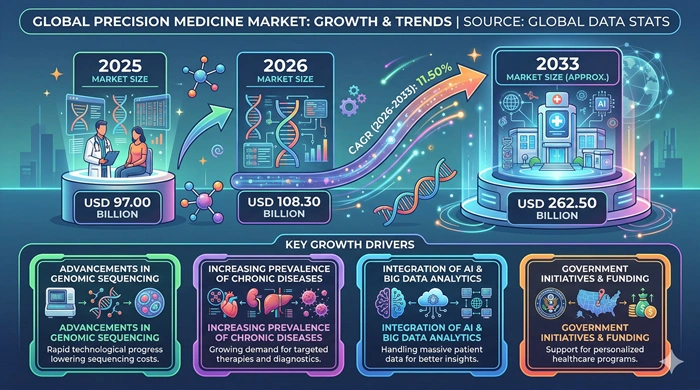
The global Precision Medicine market size is valued at USD 97.00 billion in 2025 and is predicted to increase from USD 108.30 billion in 2026 to approximately USD 262.50 billion by 2033, growing at a CAGR of 11.50% from 2026 to 2033.
Precision medicine represents one of the most transformative shifts in modern healthcare — moving away from generalized, population-wide treatment approaches toward highly individualized therapies that account for each patient's unique genetic makeup, molecular profile, lifestyle, and environmental exposure to deliver more targeted, effective, and safer medical interventions. This approach integrates advanced genomics, proteomics, metabolomics, bioinformatics, and companion diagnostics to identify the specific biological mechanisms underlying a patient's disease — enabling clinicians to select treatments most likely to succeed for that individual while avoiding those most likely to cause harm. The Precision Medicine market is experiencing robust and accelerating growth, driven by the rapidly falling cost of genomic sequencing, explosive growth in oncology precision therapy approvals, expanding pharmacogenomics adoption in drug development and clinical care, and the deepening integration of artificial intelligence into genomic data interpretation and treatment decision support across global healthcare systems.
AI Impact on the Precision Medicine Industry
Artificial Intelligence Is Transforming Genomic Data Interpretation, Drug Target Discovery, Biomarker Identification, Clinical Decision Support, and Real-World Evidence Generation in Ways That Are Fundamentally Accelerating the Pace and Commercial Scale of Personalized Medicine Adoption Across the Global Healthcare Ecosystem*
Artificial intelligence is delivering its most profound and commercially impactful contributions to the Precision Medicine market through the application of deep learning algorithms to genomic and multi-omic data interpretation — where AI systems analyze whole genome sequences, transcriptomes, proteomes, and clinical data simultaneously to identify clinically actionable genetic variants, predict disease progression trajectories, and match individual patients to optimal targeted therapies with a speed, consistency, and analytical depth that far exceeds what human clinical genomic interpretation can achieve at population scale. These AI-powered genomic interpretation platforms — developed by companies including Tempus, Foundation Medicine, Illumina DRAGEN, and IBM Watson for Genomics — are enabling clinical oncology programs and precision medicine centers to derive actionable therapeutic insights from complex multi-omic patient data within clinically meaningful timeframes, progressively transforming genomic sequencing from a research tool into a routine clinical decision support resource in oncology and other precision-amenable therapeutic areas. The FDA and global regulatory agencies have progressively recognized AI-assisted genomic interpretation and companion diagnostic platforms as clinically valid decision support tools — creating a favorable regulatory pathway that is accelerating institutional adoption of AI-powered precision medicine tools as the evolving standard of genomically-informed clinical care.
AI is simultaneously reshaping the pharmaceutical dimension of precision medicine through machine learning-powered drug target discovery and biomarker identification programs — where AI systems mine large genomic, proteomic, and clinical datasets to identify novel disease-associated molecular targets, predict compound-target binding affinity, and prioritize biomarker-stratified patient subpopulations most likely to respond to investigational precision therapies before costly clinical trials begin. Companies including BenevolentAI, Recursion Pharmaceuticals, Exscientia, and Insilico Medicine are demonstrating that AI-guided precision target discovery can significantly compress pharmaceutical development timelines and improve the probability of clinical success by ensuring that precision medicine candidates enter clinical development with stronger biological rationale and more precisely defined patient selection biomarkers. This AI acceleration of precision drug discovery is generating growing pharmaceutical industry investment in AI-enabled precision medicine platform development — creating a virtuous cycle of expanding AI capability investment, improving drug discovery productivity, and growing commercial validation of AI's role as a central enabler of precision medicine advancement.
Growth Factors
Falling Genomic Sequencing Costs, Expanding Precision Oncology Therapy Approvals, Growing Pharmacogenomics Clinical Adoption, Supportive Regulatory Biomarker Frameworks, and Rising Chronic Disease Burden Are the Core Growth Engines of the Precision Medicine Market*
The single most structurally important growth driver of the Precision Medicine market is the extraordinary and continuing decline in whole genome sequencing cost — from over USD 100 million per genome in 2001 to approximately USD 200 today — a price reduction of unprecedented scale in the history of medical technology that has transformed genomic sequencing from an experimental research tool accessible only to elite academic centers into an affordable clinical diagnostic tool deployable across mainstream healthcare systems globally. This genomic sequencing cost revolution is generating a cascade of downstream precision medicine market growth effects — enabling routine tumor genomic profiling in oncology, pharmacogenomic testing in drug prescribing, newborn genomic screening programs, preconception carrier screening, and liquid biopsy cancer detection — each representing large and growing commercial opportunities within the broader precision medicine ecosystem that collectively sustain the market's strong double-digit revenue growth trajectory. The simultaneous expansion of next-generation sequencing instrument capabilities — with platforms from Illumina, Pacific Biosciences, and Oxford Nanopore achieving increasingly long read lengths, higher throughput, and lower per-base sequencing costs — is progressively expanding the clinical applications addressable by routine genomic testing and broadening the commercial market for precision medicine products and services.
The extraordinary commercial momentum of precision oncology — where targeted therapies and immunotherapies guided by companion diagnostic biomarker testing have progressively become the standard of care across breast cancer, lung cancer, colorectal cancer, melanoma, leukemia, and a growing number of other tumor types — represents the most commercially mature and revenue-significant driver of current precision medicine market growth. The FDA has approved more than 300 targeted therapies with companion diagnostic indications — each representing a mandated genomic or molecular testing requirement that generates recurring precision medicine diagnostic revenue alongside therapeutic sales — with the oncology precision medicine pipeline containing hundreds of additional targeted therapy candidates in clinical development that will generate further companion diagnostic market expansion as they achieve regulatory approval. The growing adoption of comprehensive genomic profiling panels — where a single tumor tissue or liquid biopsy test simultaneously evaluates hundreds of clinically actionable genomic alterations — is both expanding the clinical utility of precision oncology testing and increasing average revenue per precision medicine diagnostic test in the oncology market.

Market Outlook
With Whole Genome Sequencing Becoming a Clinical Standard, Multi-Cancer Early Detection Liquid Biopsy Approaching Commercial Mainstream, AI Drug Discovery Accelerating, and Emerging Market Genomic Infrastructure Expanding Rapidly, the Precision Medicine Market Outlook Is Exceptionally Strong Through 2033*
The long-term outlook for the Precision Medicine market is firmly positive, supported by an exceptionally strong combination of technology maturation, regulatory momentum, pharmaceutical pipeline growth, and geographic expansion catalysts expected to sustain robust double-digit revenue growth throughout the 2026–2033 forecast period. The most commercially transformative near-term clinical development within precision medicine is the emergence of multi-cancer early detection liquid biopsy tests — where cell-free DNA methylation pattern analysis enables simultaneous screening for multiple cancer types from a single blood draw — with Grail's Galleri test and competing platforms from Exact Sciences, Guardant Health, and Foundation Medicine approaching commercial mainstream adoption as large prospective clinical trial evidence supporting their clinical utility progressively accumulates. Successful mainstream adoption of multi-cancer early detection liquid biopsy would represent one of the most commercially significant precision medicine market expansions in history — potentially adding tens of millions of annual tests globally and generating a new multi-billion-dollar revenue category within the precision medicine diagnostic segment that does not yet exist at commercial scale.
The progressive expansion of precision medicine beyond oncology into cardiovascular disease, neurological conditions, rare diseases, metabolic disorders, and immune-mediated inflammatory diseases represents the most important long-term commercial growth opportunity for the broader Precision Medicine market — as the genomic and molecular insights that have transformed oncology treatment are progressively being translated into actionable therapeutic frameworks across additional high-prevalence and high-burden disease areas. Polygenic risk score-guided cardiovascular disease prevention programs, pharmacogenomics-guided antidepressant and antipsychotic prescribing, genomics-guided rare disease diagnosis and treatment, and molecular subtype-guided inflammatory bowel disease therapy selection are each in various stages of clinical validation and early commercial adoption — collectively representing enormous future market expansion opportunities that will progressively materialize throughout the 2026–2033 forecast period as clinical evidence accumulates and payer reimbursement frameworks develop. Emerging market healthcare system investment in genomic infrastructure — particularly in China, India, the Middle East, and Southeast Asia — is additionally creating large new geographic precision medicine markets that are currently in early development but carry transformative long-term revenue potential.
Expert Speaks
-
Albert Bourla, CEO, Pfizer Inc. — "Precision medicine is not the future of healthcare — it is the present, and the pace of transformation is accelerating beyond what most of the industry anticipated even five years ago. The combination of affordable genomic sequencing, AI-powered data interpretation, and a rapidly expanding library of targeted therapies with validated biomarker selection criteria is creating a genuinely new paradigm of individualized medicine that is delivering measurably better patient outcomes and reshaping every dimension of pharmaceutical development, clinical practice, and healthcare system design. Pfizer is deeply committed to building our therapeutic portfolio around precision medicine principles — ensuring that every drug we develop is matched with the biomarker selection framework needed to identify the patients most likely to benefit."
-
Chris Boerner, CEO, Bristol-Myers Squibb — "Immuno-oncology and targeted therapy have already demonstrated definitively that precision medicine approaches — where treatment selection is guided by the molecular characteristics of each patient's disease rather than anatomical tumor location alone — deliver superior clinical outcomes compared to conventional empirical treatment selection strategies. BMS is investing at scale in the genomic, proteomic, and immune profiling technologies required to identify new precision medicine biomarkers across our oncology and immunology pipeline — and we see the continued expansion of companion diagnostic-guided therapy as the most important driver of durable long-term value creation in pharmaceutical development."
-
Pascal Soriot, CEO, AstraZeneca — "AstraZeneca's commercial success in oncology has been built on precision medicine principles — identifying the genomic and molecular patient subpopulations most likely to respond to our targeted therapies and ensuring that companion diagnostic testing reaches those patients efficiently within clinical practice. We see the continued evolution of precision medicine technology — including liquid biopsy early detection, multi-omic tumor profiling, and AI-guided treatment selection — as central to our strategy for developing the next generation of transformative medicines that deliver truly meaningful improvements in patient survival and quality of life."
Key Report Takeaways
-
North America dominates the Precision Medicine market, holding approximately 44.80% of global market share in 2025, driven by the world's highest clinical genomic testing adoption rates, the most extensive precision oncology companion diagnostic regulatory approval framework under FDA oversight, the largest pharmaceutical and biotechnology company investment in precision medicine drug and diagnostic development, the strongest concentration of genomics technology companies including Illumina, Foundation Medicine, Guardant Health, and Tempus, and the most mature payer reimbursement infrastructure for precision medicine diagnostic tests.
-
Asia Pacific is the fastest-growing regional market for precision medicine, projected to expand at a CAGR of approximately 13.40% from 2026 to 2033, driven by China's extraordinary government investment in genomic medicine infrastructure through the Precision Medicine Initiative and China Precision Medicine Initiative programs, India's rapidly growing cancer patient population generating precision oncology testing demand, and South Korea and Japan's progressive integration of genomic testing into national healthcare reimbursement frameworks that are expanding clinical precision medicine adoption across Asian healthcare systems.
-
Oncology is the dominant application segment, accounting for approximately 46.20% of total market revenue in 2025, reflecting precision oncology's position as the most clinically mature and commercially developed precision medicine application — where hundreds of FDA-approved targeted therapies with mandatory companion diagnostic testing requirements generate large and continuously growing precision medicine diagnostic revenue alongside the targeted therapy pharmaceutical market that collectively represents the largest single commercial expression of precision medicine principles in clinical practice.
-
Genomics is the dominant technology segment, contributing approximately 38.60% of total market revenue in 2025, driven by next-generation sequencing's foundational role as the primary technology platform enabling comprehensive genomic profiling across oncology, rare disease, pharmacogenomics, and population health precision medicine applications — with NGS instrument and reagent revenue from platforms including Illumina NovaSeq, Illumina NextSeq, and emerging long-read platforms from Pacific Biosciences and Oxford Nanopore collectively representing the largest single technology revenue category within the precision medicine market.
-
Pharmaceutical and biotechnology companies represent the largest end-user segment, contributing approximately 41.30% of total market revenue in 2025, driven by the massive and continuously growing investment these organizations make in precision medicine drug discovery, biomarker development, companion diagnostic co-development, and pharmacogenomics programs across their clinical development pipelines.
-
Liquid biopsy and multi-cancer early detection is the fastest-growing future segment, expected to expand at a CAGR of approximately 18.60% through 2033, driven by the progressive commercialization of cell-free DNA methylation-based multi-cancer early detection tests — including Grail's Galleri test and competing platforms — and the expanding clinical validation of liquid biopsy for minimal residual disease monitoring, treatment response assessment, and resistance mutation detection in precision oncology programs that are generating growing clinical adoption and payer reimbursement coverage expansion across North American and European healthcare markets.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 262.50 Billion |
| Market Size by 2025 | USD 97.00 Billion |
| Market Size by 2026 | USD 108.30 Billion |
| Market Growth Rate (2026–2033) | CAGR of 11.50% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Segments Covered | Technology, Application, End User |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Falling Genomic Sequencing Costs, Expanding Precision Oncology Approvals, AI-Powered Genomic Interpretation, Pharmacogenomics Clinical Adoption, and Rising Chronic Disease Burden Are Collectively Powering the Precision Medicine Market's Strong Double-Digit Revenue Growth*
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Continuously falling genomic sequencing costs expanding clinical adoption | ~31% | Global | Short to Long-term |
| Expanding precision oncology targeted therapy and companion diagnostic approvals | ~28% | North America, Europe | Short to Long-term |
| AI-powered genomic data interpretation and drug discovery acceleration | ~24% | North America, Europe, Asia Pacific | Short to Medium-term |
| Growing pharmacogenomics clinical adoption in drug prescribing | ~17% | North America, Europe | Short to Medium-term |
The most foundational and commercially transformative driver sustaining the Precision Medicine market's strong growth trajectory is the continuing democratization of genomic sequencing technology — where the combination of dramatically lower sequencing costs, higher instrument throughput, and more clinically actionable bioinformatics interpretation pipelines is progressively enabling genomic testing to transition from specialized academic and reference laboratory settings into mainstream clinical diagnostic workflows at hospitals, oncology centers, and large physician practice groups globally. This genomic testing democratization is simultaneously expanding the clinical indications where precision medicine approaches are validated and reimbursed — generating new precision medicine market revenue across oncology molecular profiling, pharmacogenomics drug selection, rare disease genomic diagnosis, reproductive genomics, and population health genomic screening applications that each represent meaningful commercial market segments within the broader precision medicine ecosystem. The accelerating pace of precision therapy FDA approvals — with targeted therapy and immunotherapy approvals increasingly requiring validated companion diagnostic tests — is generating a growing mandatory testing market that sustains predictable precision medicine diagnostic revenue streams regardless of broader healthcare spending variability.
The extraordinary pace of AI advancement in genomic and multi-omic data interpretation is simultaneously enabling precision medicine to address molecular complexity that has historically exceeded human analytical capabilities — where AI systems can identify clinically meaningful patterns within whole genome, transcriptome, proteome, and clinical data combinations that would require impractically large human analytical teams to process at the individual patient level. This AI-powered analytical capability expansion is progressively enabling precision medicine principles to penetrate clinical applications — including polygenic risk score-guided disease prevention, AI-guided treatment response prediction, and molecular subtype-based therapy selection in complex immune-mediated diseases — that the existing evidence base and analytical infrastructure had not previously been able to support at scale. The combination of AI analytical power, continuously falling sequencing costs, and an expanding library of validated precision medicine biomarkers is creating a compounding market growth dynamic where each new precision medicine clinical validation generates new testing volume, new therapeutic development investment, and new downstream market expansion.

Restraints Impact Analysis
High Genomic Test Reimbursement Uncertainty, Data Privacy Regulatory Complexity, Genomic Data Infrastructure Investment Requirements, and Clinician Education Gaps Restrain Faster Precision Medicine Market Penetration*
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Inconsistent payer reimbursement coverage for genomic and precision medicine tests | ~35% | Global — especially emerging markets | Short to Long-term |
| Genomic data privacy regulation complexity across multiple jurisdictions | ~27% | Europe, North America, Asia Pacific | Short to Long-term |
| High infrastructure investment requirements for genomic medicine program establishment | ~24% | Asia Pacific, Latin America, MEA | Short to Medium-term |
| Clinician genomic literacy and genetic counseling capacity gaps | ~14% | Global | Short to Medium-term |
The most broadly experienced and commercially consequential barrier constraining faster precision medicine adoption is the highly inconsistent and often inadequate payer reimbursement coverage for genomic and molecular diagnostic tests across healthcare systems globally — where the clinical utility evidence required by payers for reimbursement coverage decisions frequently lags behind the pace of genomic testing innovation, creating significant access barriers for patients in markets where precision medicine tests are clinically available but not reimbursed. The United States Medicare coverage landscape for genomic tests — governed by Local Coverage Determinations that vary by contractor jurisdiction — creates a fragmented reimbursement environment where identical genomic tests may be covered for some patient populations and clinical indications but not others, complicating precision medicine program adoption decisions for health systems and ordering physicians. European national healthcare systems present an equally complex payer landscape — where individual country health technology assessment processes assess genomic test clinical utility and cost-effectiveness independently, creating a balkanized European precision medicine reimbursement environment that limits the commercial scale achievable within the region relative to its clinical infrastructure capabilities.
The growing complexity of genomic data privacy regulation — including GDPR genomic data special category protections in Europe, HIPAA genetic information provisions in the United States, GINA genetic discrimination protections, and a proliferating landscape of national genomic data sovereignty requirements across Asian markets — is creating substantial compliance burden for precision medicine companies operating multi-country genomic testing, data analysis, and research programs. Healthcare organizations seeking to build precision medicine programs across multiple jurisdictions must navigate a complex web of genomic data consent requirements, cross-border data transfer restrictions, and genomic data localization mandates that disproportionately burden smaller precision medicine organizations lacking dedicated regulatory and privacy compliance infrastructure. The combination of reimbursement uncertainty and data privacy complexity is creating a challenging commercial operating environment that moderates the precision medicine market's growth rate below the trajectory that the strength of its clinical evidence and technological capabilities would otherwise support.
Opportunities Impact Analysis
Multi-Cancer Early Detection Liquid Biopsy Commercialization, Rare Disease Genomic Diagnosis Expansion, Polygenic Risk Score Clinical Adoption, and Emerging Market Genomic Infrastructure Investment Create Major Precision Medicine Market Opportunities*
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Multi-cancer early detection liquid biopsy mainstream commercialization | ~34% | North America, Europe | Short to Medium-term |
| Rare disease genomic diagnosis and precision therapy development expansion | ~26% | North America, Europe | Short to Long-term |
| Pharmacogenomics mainstream adoption in drug prescribing across therapeutic areas | ~25% | North America, Europe, Asia Pacific | Short to Medium-term |
| Emerging market genomic medicine infrastructure establishment | ~15% | Asia Pacific, Middle East, Latin America | Medium to Long-term |
The single most commercially transformative near-term opportunity within the Precision Medicine market is the successful mainstream commercialization of multi-cancer early detection liquid biopsy tests — where the ability to detect multiple cancer types simultaneously from a single blood sample at early, more treatable stages represents a fundamentally new and enormously large precision medicine market category that could generate tens of millions of annual tests globally if clinical validation evidence, physician adoption, and payer reimbursement coverage develop favorably over the 2026–2033 forecast period. Grail's Galleri test — currently commercially available in the United States and undergoing reimbursement coverage evaluation — represents the most advanced commercial expression of multi-cancer early detection, with NHS England's Galleri trial generating important clinical evidence supporting population-level liquid biopsy screening program adoption that could establish the clinical and economic validation framework required for national healthcare system adoption across major markets. The multi-cancer early detection liquid biopsy opportunity represents a market expansion of potentially comparable commercial significance to the prior development of precision oncology molecular profiling — introducing a large new population of asymptomatic individuals as precision medicine test recipients rather than limiting precision medicine testing to already-diagnosed cancer patients.
The rare disease genomic medicine opportunity represents an equally compelling and commercially significant precision medicine market expansion — where the approximately 10,000 known rare genetic diseases affecting more than 300 million people globally represent a massive underserved patient population whose diagnostic odysseys and treatment options are being transformed by comprehensive genomic sequencing that identifies causative variants and, increasingly, targeted precision therapies designed to address specific genetic mechanisms. The FDA's accelerated approval pathway for precision rare disease therapies — enabled by genomic biomarker patient selection and supported by the FDA's rare pediatric disease and orphan drug designation programs — is generating a rapidly growing portfolio of approved precision rare disease therapies including gene therapies, antisense oligonucleotides, and small molecule precision treatments that create new companion diagnostic and genomic testing market segments alongside each therapeutic approval. Precision medicine companies that develop comprehensive rare disease genomic testing and interpretation capabilities are well-positioned to capture a disproportionate share of the growing rare disease precision medicine market throughout the forecast period.

Segment Analysis
By Application
Precision Oncology Commands the Largest Application Share in the Precision Medicine Market While Multi-Cancer Liquid Biopsy and CNS Precision Applications Register the Fastest Growth*
Oncology holds the dominant position in the Precision Medicine market, accounting for approximately 46.20% of total global market revenue in 2025, reflecting precision oncology's status as the most clinically mature, commercially validated, and therapeutically transformative application of precision medicine principles — where hundreds of approved targeted therapies with companion diagnostic biomarker requirements collectively generate mandatory molecular testing demand across lung cancer, breast cancer, colorectal cancer, melanoma, hematologic malignancies, and a growing spectrum of other tumor types that cannot be optimally managed without genomic and molecular characterization of each patient's specific cancer biology. Comprehensive genomic profiling panels — where a single tumor tissue or liquid biopsy test evaluates hundreds of clinically actionable genomic alterations simultaneously — are progressively becoming standard of care across major oncology centers in North America and Europe, driving per-test revenue expansion alongside growing total test volume as precision oncology's clinical standard progressively shifts from single-gene companion diagnostic testing toward multi-gene panel and whole exome approaches. Leading precision oncology companies including Foundation Medicine, Guardant Health, Tempus, Illumina, and Myriad Genetics dominate North American precision oncology market revenue — with Foundation Medicine and Guardant Health commanding particularly strong commercial positions through comprehensive genomic profiling panel products validated across multiple tumor types and supported by extensive clinical evidence publications that drive clinical adoption. Asia Pacific is the fastest-growing precision oncology market — driven by the rapidly expanding cancer patient population, progressive adoption of international precision oncology testing standards, and growing pharmaceutical company clinical trial activity generating comprehensive genomic profiling requirements at Asian research sites.
Pharmacogenomics represents the second-largest and increasingly important application segment in the Precision Medicine market, accounting for approximately 18.40% of total market revenue in 2025 and growing at a CAGR of approximately 13.20% through 2033 as the clinical evidence base supporting pharmacogenomics-guided drug prescribing progressively expands across psychiatry, cardiology, oncology supportive care, pain management, and infectious disease therapeutic areas. Pharmacogenomics testing — which analyzes an individual patient's genetic variants in drug-metabolizing enzyme genes including CYP2D6, CYP2C19, CYP2C9, and DPYD to predict drug response, toxicity risk, and optimal dosing — is gaining clinical momentum as health systems recognize the patient safety, treatment effectiveness, and healthcare cost benefits of replacing empirical drug selection with genomically-guided prescribing for high-risk medications. Myriad Genetics' GeneSight pharmacogenomics test, Mayo Clinic's RIGHT Protocol, and Genomind's pharmacogenomics panel represent the most commercially advanced pharmacogenomics platforms in North America — each generating meaningful commercial revenue from psychiatry and pain management applications where drug selection variability and adverse effect burden create compelling clinical motivation for genomic guidance. Europe is the fastest-growing pharmacogenomics market — driven by the Dutch Pharmacogenomics Working Group's comprehensive prescribing guidelines that cover over 100 drug-gene pairs and are progressively being integrated into electronic prescribing systems across European healthcare organizations that are establishing pharmacogenomics-guided prescribing as a mainstream clinical practice.
By Technology
Next-Generation Sequencing Leads the Precision Medicine Technology Market While AI-Powered Bioinformatics Platforms and Liquid Biopsy Technologies Register the Fastest Commercial Expansion*
Genomics — led by next-generation sequencing technology — holds the dominant position in the Precision Medicine market's technology segmentation, accounting for approximately 38.60% of total global market revenue in 2025, reflecting NGS's foundational role as the primary enabling technology for virtually all clinical precision medicine applications from comprehensive cancer genomic profiling and pharmacogenomics through rare disease diagnosis, reproductive genomics, and population health genomic screening. The NGS instrument and reagent market is dominated by Illumina — whose NovaSeq, NextSeq, and MiSeq platforms represent the most widely deployed NGS infrastructure in clinical and research precision medicine programs globally — with Illumina commanding approximately 70–75% of global NGS instrument market share while facing emerging competition from Pacific Biosciences' long-read SMRT sequencing platform and Oxford Nanopore's real-time portable sequencing technology that address specific clinical applications where Illumina's short-read technology has limitations. North America leads genomics technology revenue globally — driven by the highest NGS instrument deployment density, most mature clinical genomics laboratory infrastructure, and largest concentration of genomics technology company R&D investment — while Asia Pacific is the fastest-growing genomics technology region at approximately 14.80% CAGR through 2033, driven by China's government-funded genomic medicine initiative investments and India's expanding clinical genomics laboratory sector. Key players including Illumina, Pacific Biosciences, Thermo Fisher Scientific, and Agilent Technologies dominate the North American genomics technology segment through comprehensive instrument, reagent, and bioinformatics software portfolio offerings.
Bioinformatics and big data analytics represent the fastest-growing technology segment within the Precision Medicine market, projected to grow at a CAGR of approximately 16.40% from 2026 to 2033 — reflecting the growing recognition that the most commercially valuable component of precision medicine programs is not the sequencing instrument itself but the analytical software, clinical interpretation algorithms, and AI-powered decision support platforms that transform raw genomic data into actionable clinical insights that physicians can use to guide treatment decisions. The bioinformatics segment encompasses a diverse and rapidly expanding commercial landscape including genomic variant interpretation software, clinical decision support platforms, AI-powered tumor mutation burden and microsatellite instability callers, population genomics analysis tools, and integrated multi-omic data analysis platforms that collectively represent the software infrastructure layer enabling precision medicine data to translate into clinical value. Tempus, Flatiron Health (Roche), Foundation Medicine, and specialized AI bioinformatics companies including SOPHiA GENETICS are among the most commercially dynamic bioinformatics and precision medicine analytics platform providers — with SOPHiA GENETICS' cloud-based genomic data analysis platform commanding particularly strong commercial traction among hospital clinical genomics laboratories in Europe and North America seeking scalable and cost-effective bioinformatics infrastructure for their growing precision medicine testing programs.

Regional Insights
North America
North America Commands the Global Precision Medicine Market With the World's Most Mature Precision Oncology Ecosystem, Most Progressive FDA Companion Diagnostic Regulatory Framework, and the Highest Concentration of Genomics Technology Innovation and Commercial Leadership*
North America holds the dominant position in the global Precision Medicine market, accounting for approximately 44.80% of total global market revenue in 2025, and is projected to sustain steady market leadership at a CAGR of approximately 10.80% from 2026 to 2033. The United States drives the overwhelming majority of North American revenue — where the FDA's robust companion diagnostic approval framework, the National Cancer Institute's comprehensive genomic data programs, the NIH Precision Medicine Initiative's All of Us Research Program, and the world's highest clinical genomic testing adoption rates collectively create the most commercially developed and clinically advanced precision medicine ecosystem globally. Leading companies commanding the North American Precision Medicine market include Illumina, Foundation Medicine, Guardant Health, Tempus, Myriad Genetics, Grail, Exact Sciences, Thermo Fisher Scientific, and Roche Diagnostics — each maintaining strong commercial positions across their respective precision medicine technology and application segments through extensive clinical evidence portfolios, validated companion diagnostic regulatory approvals, and deep health system customer relationships.
Canada contributes meaningfully to North American precision medicine revenue — with Cancer Care Ontario's precision oncology programs, Genome Canada's investment in clinical genomics research, and the progressive integration of genomic testing into provincial cancer care guidelines collectively establishing a well-developed Canadian precision medicine clinical infrastructure. North America's precision medicine ecosystem is evolving most rapidly in the liquid biopsy and multi-cancer early detection segment — where Grail, Guardant Health, Foundation Medicine, and Exact Sciences are competing intensely for first-mover commercial advantage in what is widely recognized as the largest near-term precision medicine market expansion opportunity globally. The combination of regulatory leadership, commercial infrastructure maturity, and technology innovation concentration positions North America to maintain clear precision medicine market leadership throughout 2033.
Asia Pacific
Asia Pacific Is the Fastest-Growing Precision Medicine Market, Driven by China's Government Genomic Medicine Investment, India's Growing Cancer Burden, and Progressive Integration of Genomic Testing Into Asian Healthcare Reimbursement Frameworks*
Asia Pacific is the fastest-growing regional segment in the Precision Medicine market, projected to expand at a CAGR of approximately 13.40% from 2026 to 2033 — the highest regional growth rate globally. China is the most commercially significant and rapidly growing Asia Pacific precision medicine market — where government investment through the China Precision Medicine Initiative has funded genomic infrastructure development at major academic medical centers and established national genomic data platforms enabling population-scale genomic research that is accelerating domestic precision medicine clinical capability development. India represents the second-largest and equally dynamic Asia Pacific precision medicine market — where the country's enormous and rapidly growing cancer patient population, combined with government healthcare quality improvement programs and the progressive adoption of international oncology treatment guidelines incorporating precision medicine testing requirements, is generating fast-growing precision oncology testing demand that is attracting investment from global genomics technology companies seeking Asian market expansion. Key companies serving the Asia Pacific Precision Medicine market include Illumina, Thermo Fisher Scientific, Roche Diagnostics, and regional Asian genomics companies including Berry Genomics, WuXi AppTec, and GenomicsEngland partners serving domestic precision medicine program development.
Japan and South Korea represent the most technically sophisticated and commercially mature Asia Pacific precision medicine markets — with well-established national precision medicine research programs, progressive integration of companion diagnostic testing into national health insurance reimbursement frameworks, and strong domestic pharmaceutical and genomics company investment in precision medicine capability development. Southeast Asian markets — particularly Singapore, Australia, and Taiwan — are developing rapidly as regional precision medicine hubs — with Singapore's Precision Health Research Singapore program and Australia's national genomics initiative each investing in the clinical, research, and regulatory infrastructure required to support mainstream precision medicine adoption. The extraordinary scale of Asia Pacific's cancer patient population, combined with rapidly improving genomic testing infrastructure and growing government precision medicine investment, positions the region as the most commercially dynamic and fastest-expanding precision medicine market globally through 2033.
Top Key Players
-
Illumina Inc. (United States)
-
Thermo Fisher Scientific Inc. (United States)
-
Roche Diagnostics (Switzerland)
-
Foundation Medicine Inc. — Roche (United States)
-
Guardant Health Inc. (United States)
-
Myriad Genetics Inc. (United States)
-
Tempus AI Inc. (United States)
-
Exact Sciences Corporation (United States)
-
Grail Inc. — Illumina (United States)
-
SOPHiA GENETICS SA (Switzerland)
-
Pacific Biosciences of California Inc. (United States)
-
Agilent Technologies Inc. (United States)
Recent Developments
-
Illumina Inc. (2025) — Completed the divestiture of Grail Inc. following regulatory mandates — with Grail re-emerging as an independent company focused on accelerating the commercialization of its Galleri multi-cancer early detection liquid biopsy test — while Illumina simultaneously announced the commercial launch of its NovaSeq X Plus instrument platform featuring significantly reduced per-genome sequencing costs and expanded throughput capabilities that are expected to accelerate clinical whole genome sequencing adoption across precision medicine programs globally.
-
Guardant Health (2025) — Received FDA approval for the Guardant Shield blood-based colorectal cancer screening test — representing a landmark regulatory approval establishing liquid biopsy as a validated primary colorectal cancer screening modality — and simultaneously reported strong commercial revenue growth for its Guardant360 and Guardant Infinity precision oncology profiling platforms driven by expanding comprehensive genomic profiling adoption across community oncology practices and academic cancer centers in North America.
-
Foundation Medicine (2024) — Launched FoundationOne Liquid CDx expanded indications — adding new FDA-approved companion diagnostic claims for additional targeted therapies across lung cancer, breast cancer, and other solid tumor types — progressively expanding the clinical utility of its comprehensive liquid biopsy platform and reinforcing Foundation Medicine's position as the market leader in FDA-approved companion diagnostic liquid biopsy testing for precision oncology treatment selection across North American and European markets.
-
Tempus AI Inc. (2025) — Completed its initial public offering and began trading as a publicly listed company — raising significant capital to accelerate its AI-powered precision medicine platform development — while reporting strong commercial momentum for its TIME clinical sequencing platform, Lens multimodal data analytics service, and newly launched Tempus Next AI clinical decision support tool that integrates genomic sequencing results with multimodal clinical data to generate AI-powered precision medicine treatment recommendations for oncology providers.
-
Exact Sciences Corporation (2024) — Announced the commercial expansion of its Oncotype DX breast cancer genomic assay into new Asian markets and reported strong revenue growth for its Cologuard colorectal cancer screening test — while simultaneously advancing clinical development of its multi-cancer early detection liquid biopsy platform designed to detect multiple cancer types simultaneously from a blood sample — positioning Exact Sciences to compete in the emerging multi-cancer early detection market alongside Grail and Guardant Health as clinical validation evidence matures.
Market Trends
The Convergence of Multi-Cancer Liquid Biopsy Early Detection Commercialization and the Mainstream Integration of AI-Powered Precision Medicine Clinical Decision Support Are the Two Most Commercially Defining Trends Reshaping the Precision Medicine Market Through 2033*
The most commercially transformative structural trend reshaping the Precision Medicine market is the progressive commercialization of multi-cancer early detection liquid biopsy technology — where the ability to detect multiple cancer types simultaneously at early stages from a single blood sample is transitioning from a clinical research concept toward a commercially available product with growing clinical evidence support, regulatory engagement, and payer reimbursement evaluation. The NHS England Galleri trial, the NCI's PATHFINDER study, and multiple other large prospective clinical studies are generating the population-level clinical evidence required to support national healthcare system adoption of multi-cancer early detection screening programs — with the magnitude of the potential market impact comparable in scale to the transformative commercial impact of PSA testing, mammography, and colonoscopy in their respective cancer screening contexts. As clinical evidence matures and reimbursement decisions develop favorably, multi-cancer early detection is positioned to become the single largest new revenue category in precision medicine history — introducing a healthy screening population at scale as precision medicine test recipients and generating a fundamentally new and commercially enormous market dimension within the broader precision medicine ecosystem.
The mainstream integration of AI-powered clinical decision support into precision medicine programs — where AI systems synthesize genomic sequencing results, multi-omic biomarker profiles, real-world clinical outcome data, and published precision medicine evidence to generate individualized treatment recommendations for clinicians — is simultaneously transforming precision medicine from a primarily diagnostic activity into a comprehensive clinical intelligence service that creates substantially greater clinical value than genomic testing alone. Precision medicine companies that develop validated AI clinical decision support capabilities alongside their genomic testing products are progressively differentiating their commercial offerings and capturing higher per-patient revenue by delivering not just molecular data but actionable clinical intelligence that directly informs treatment decisions. This AI-enhanced precision medicine value proposition is creating important competitive differentiation between platform providers that offer integrated genomic testing plus AI clinical intelligence and those delivering genomic data alone — reshaping the competitive landscape of the precision medicine industry and accelerating investment in AI clinical decision support capability development across leading precision medicine companies.
Segments Covered in the Report
By Technology:
-
Genomics
-
Next-Generation Sequencing (NGS)
-
Polymerase Chain Reaction (PCR)
-
Microarray
-
Proteomics
-
Metabolomics
-
Pharmacogenomics
-
Bioinformatics and Big Data Analytics
-
Others
By Application:
-
Oncology
-
Pharmacogenomics
-
CNS (Central Nervous System)
-
Immunology
-
Cardiovascular Diseases
-
Rare Diseases
-
Others
By End User:
-
Hospitals and Healthcare Providers
-
Pharmaceutical and Biotechnology Companies
-
Diagnostic Laboratories
-
Academic and Research Institutes
-
Contract Research Organizations (CROs)
By Region:
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the Precision Medicine market and what is its projected value by 2033?
Answer: The Precision Medicine market was valued at USD 97.00 billion in 2025 and is projected to reach USD 262.50 billion by 2033, growing at a CAGR of 11.50% from 2026 to 2033. This growth is driven by falling genomic sequencing costs, expanding precision oncology approvals, AI-powered data analytics adoption, and growing pharmacogenomics integration across clinical practice globally.
Question 2: What is precision medicine and how is it different from traditional medicine?
Answer: Precision medicine is a medical approach that tailors treatment and prevention strategies to the individual patient's unique genetic, molecular, environmental, and lifestyle characteristics — rather than applying a one-size-fits-all treatment based on population-average responses to therapy. The Precision Medicine market encompasses the technologies, diagnostics, and therapeutics that enable this individualized approach — including genomic sequencing, companion diagnostics, targeted therapies, and AI-powered clinical decision support platforms.
Question 3: Which region leads the Precision Medicine market and which is growing the fastest?
Answer: North America leads the Precision Medicine market with approximately 44.80% of global revenue in 2025 — driven by the most mature precision oncology ecosystem, the most progressive FDA companion diagnostic approval framework, and the highest clinical genomic testing adoption rates globally. Asia Pacific is the fastest-growing region at approximately 13.40% CAGR through 2033 — driven by China's government genomic medicine investment, India's rapidly growing cancer patient population, and progressive integration of genomic testing into Asian healthcare reimbursement frameworks.
Question 4: How is artificial intelligence accelerating growth in the Precision Medicine market?
Answer: AI is accelerating precision medicine growth through deep learning-powered genomic variant interpretation that extracts clinically actionable insights from complex multi-omic datasets at unprecedented speed and scale, and through machine learning drug target discovery programs that identify novel precision medicine targets and biomarker-stratified patient populations before clinical trials begin — improving development efficiency and probability of success. The integration of AI clinical decision support into precision medicine platforms is progressively transforming genomic testing from a data generation activity into a comprehensive clinical intelligence service that delivers directly actionable treatment recommendations to clinicians.
Question 5: What are the key challenges facing the Precision Medicine market growth?
Answer: The most significant challenges constraining faster precision medicine market growth include inconsistent payer reimbursement coverage for genomic and molecular diagnostic tests — where clinical utility evidence requirements frequently lag behind testing innovation — and the growing complexity of genomic data privacy regulation across multiple jurisdictions that creates substantial compliance burden for organizations operating multinational precision medicine programs. Clinician genomic literacy gaps and genetic counseling capacity limitations additionally moderate the pace at which precision medicine test results can be effectively interpreted and applied in routine clinical practice outside specialized genomic medicine centers.
