Pharmaceutical Manufacturing Market Overview
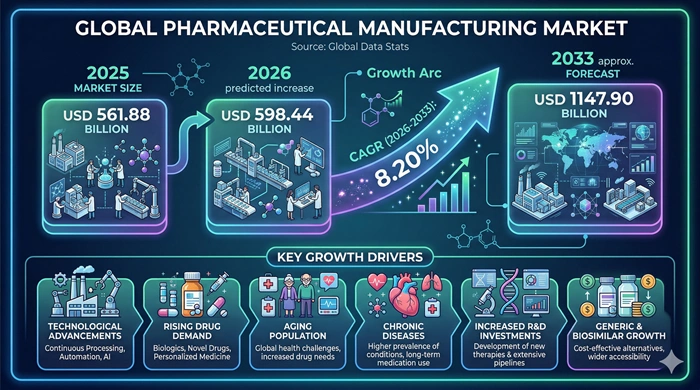
The global Pharmaceutical Manufacturing market size is valued at USD 561.88 billion in 2025 and is predicted to increase from USD 598.44 billion in 2026 to approximately USD 1147.90 billion by 2033, growing at a CAGR of 8.20% from 2026 to 2033.
Pharmaceutical manufacturing encompasses the complete spectrum of processes, technologies, and facilities involved in producing medicinal products — from active pharmaceutical ingredient (API) synthesis and purification through drug product formulation, fill-finish operations, quality control testing, and commercial packaging. This highly regulated and technically complex industry forms the foundational production backbone of the global healthcare system — ensuring the reliable supply of small molecule drugs, biologics, vaccines, generics, biosimilars, and over-the-counter medicines to patients worldwide. The Pharmaceutical Manufacturing market is experiencing a period of exceptional growth and transformation, driven by rising global disease burden, the biologics and biosimilars revolution, expanding contract manufacturing sector, and the progressive adoption of continuous manufacturing and digitalization technologies across the industry.
AI Impact on the Pharmaceutical Manufacturing Industry
Artificial Intelligence Is Transforming Drug Formulation Development, Manufacturing Process Optimization, Quality Control Automation, and Supply Chain Management in Ways That Are Meaningfully Accelerating Production Efficiency, Product Quality, and Regulatory Compliance*
Artificial intelligence is making a profound and rapidly accelerating impact across the Pharmaceutical Manufacturing market, most immediately through its application in process analytical technology (PAT), real-time release testing, and AI-driven quality control systems that are enabling pharmaceutical manufacturers to detect and correct production deviations in real time rather than through traditional end-of-batch testing. Machine learning models trained on large multivariate process datasets are being deployed to identify the complex, non-linear relationships between raw material properties, process parameters, and finished product quality attributes — enabling manufacturers to establish more robust design spaces, reduce batch failures, and achieve consistent product quality with greater confidence and efficiency. Leading pharmaceutical manufacturers including Pfizer, Johnson & Johnson, and Novartis are actively deploying AI-powered manufacturing analytics platforms that monitor production processes in real time, predict quality deviations before they occur, and generate automated recommendations for process corrections that maintain product within specification throughout the production campaign.
Beyond quality management, AI is transforming pharmaceutical manufacturing through its application in predictive maintenance of critical production equipment, AI-assisted regulatory submission preparation, and machine learning-driven supply chain optimization. Predictive maintenance algorithms — analyzing vibration data, temperature profiles, pressure signatures, and other equipment sensor outputs — are enabling pharmaceutical manufacturers to anticipate and prevent critical equipment failures before they cause costly production shutdowns or product loss events. AI-powered natural language processing tools are simultaneously accelerating regulatory submission preparation by automating the extraction, organization, and cross-referencing of manufacturing data for regulatory filings — reducing the time and resource burden of pharmaceutical manufacturing regulatory compliance. These combined AI applications are progressively elevating the operational efficiency, quality assurance capability, and regulatory performance of pharmaceutical manufacturing operations globally.
Growth Factors
Rising Global Disease Burden, Biologics and Biosimilars Pipeline Expansion, Growing Contract Manufacturing Outsourcing, and Continuous Manufacturing Technology Adoption Are the Core Drivers of Pharmaceutical Manufacturing Market Growth*
The most fundamental and enduring driver of the Pharmaceutical Manufacturing market is the continuously rising global burden of chronic and infectious diseases, which is expanding the patient population requiring pharmaceutical treatment and driving sustained growth in global medicine consumption volumes. An aging global population — with the proportion of people aged 65 and older growing across virtually every major healthcare market — is disproportionately increasing demand for medicines treating cardiovascular diseases, diabetes, cancer, respiratory conditions, and neurological disorders that require long-term pharmaceutical management. Simultaneously, the ongoing threat of emerging infectious diseases — as dramatically demonstrated by the COVID-19 pandemic — has reinforced global commitment to maintaining robust vaccine and antiviral drug manufacturing capabilities, driving significant infrastructure and capacity investment across both government-supported and private-sector pharmaceutical production networks.
The pharmaceutical industry's ongoing transition from small molecule-dominated drug pipelines toward an increasing proportion of large molecule biologics — including monoclonal antibodies, recombinant proteins, cell therapies, gene therapies, and mRNA-based medicines — is creating both significant manufacturing capacity expansion requirements and important technology investment demand within the pharmaceutical manufacturing sector. Biologic drug manufacturing requires substantially more complex, capital-intensive, and technically demanding production infrastructure than traditional small molecule chemical synthesis — including bioreactor systems, cell culture processes, chromatography purification platforms, and specialized fill-finish capabilities for sensitive biological drug products. The rapid growth of the biosimilars sector — as patents expire on major biologic reference products — is simultaneously driving significant new manufacturing capacity investment by both established pharmaceutical companies and specialized biosimilar manufacturers building the production infrastructure required to serve expanding biosimilar market opportunities globally.

Market Outlook
With Global Medicine Demand Growing Steadily, Biologics Manufacturing Capacity Expanding Rapidly, Contract Manufacturing Sector Scaling, and Continuous Manufacturing Adoption Accelerating, the Pharmaceutical Manufacturing Market Outlook Is Strongly Positive Through 2033*
The long-term outlook for the Pharmaceutical Manufacturing market is firmly positive and supported by multiple converging structural growth drivers that are expected to sustain strong market expansion throughout the 2026–2033 forecast period. The global biologics manufacturing sector is experiencing an exceptional capacity build-out cycle — with leading pharmaceutical companies, contract development and manufacturing organizations (CDMOs), and emerging biotech manufacturers collectively investing tens of billions of dollars in new bioreactor capacity, fill-finish infrastructure, and advanced cell and gene therapy manufacturing facilities to serve the rapidly expanding biologics drug pipeline. This manufacturing investment wave is creating both significant near-term capital expenditure demand for pharmaceutical manufacturing equipment and facility suppliers and robust long-term production capacity that will support continued market revenue growth through the forecast period.
The progressive adoption of continuous manufacturing technology — which replaces traditional batch-by-batch production with uninterrupted, flow-based drug synthesis and processing — represents a particularly important technology-driven growth and efficiency improvement driver for the Pharmaceutical Manufacturing market. Continuous manufacturing delivers significant advantages over conventional batch methods — including smaller facility footprint, reduced production cycle times, lower work-in-process inventory, improved process consistency, real-time quality monitoring, and reduced manufacturing cost per unit — making it an increasingly attractive operational upgrade for pharmaceutical producers seeking competitive cost efficiency and manufacturing agility. As regulatory agencies including the FDA actively encourage continuous manufacturing adoption and as more products receive regulatory approval through continuous manufacturing processes, the penetration of this technology across the pharmaceutical industry is expected to accelerate meaningfully during the forecast period, driving important equipment and technology investment within the manufacturing sector.
Expert Speaks
-
Albert Bourla, CEO, Pfizer — "Investing in pharmaceutical manufacturing capabilities — including both our internal production network and our partnerships with contract manufacturing organizations — is a core strategic priority that directly determines our ability to deliver medicines and vaccines reliably and at scale to patients globally. The adoption of advanced manufacturing technologies including continuous processing and AI-powered quality systems is fundamental to making pharmaceutical production more efficient, resilient, and responsive to evolving patient needs."
-
Joaquin Duato, CEO, Johnson & Johnson — "The transformation of pharmaceutical manufacturing through advanced technologies — from continuous manufacturing and single-use systems to artificial intelligence and digitalized quality management — is creating a fundamentally more capable and efficient industry that can better serve the growing global demand for innovative medicines. Johnson & Johnson is committed to leading this manufacturing transformation and to ensuring that our production capabilities match the ambition of our drug development pipeline."
-
Vasant Narasimhan, CEO, Novartis — "Pharmaceutical manufacturing excellence is not just an operational capability — it is a patient safety and public health imperative. At Novartis, we are investing significantly in next-generation manufacturing technologies and digital transformation of our production network to ensure that we can reliably supply high-quality medicines to patients worldwide while continuously improving efficiency, sustainability, and manufacturing agility across our global operations."
Key Report Takeaways
-
North America dominates the Pharmaceutical Manufacturing market, holding approximately 36.40% of global market share in 2025, driven by the world's largest pharmaceutical industry concentration — including the headquarters and major manufacturing facilities of industry leaders such as Pfizer, Johnson & Johnson, Merck, and Abbott — combined with the strongest biopharmaceutical R&D investment globally, robust FDA-regulated manufacturing infrastructure, and the largest per-capita medicine consumption market worldwide.
-
Asia Pacific is the fastest-growing regional market for pharmaceutical manufacturing, projected to expand at a CAGR of approximately 9.60% from 2026 to 2033, driven by India's dominant global generic drug and API manufacturing position, China's rapidly expanding biopharmaceutical production capabilities, and the progressive build-out of advanced pharmaceutical manufacturing infrastructure across South Korea, Japan, and Southeast Asian markets.
-
Finished dosage formulations are the dominant product type segment, accounting for approximately 48.30% of total market revenue in 2025, as the formulation, filling, and packaging of final drug products for patient administration represents the highest-value and most complex stage of the pharmaceutical production chain — encompassing tablets, capsules, injectables, topical preparations, and biological drug products across all therapeutic categories.
-
Biopharmaceuticals are the fastest-growing product segment, projected to expand at a CAGR of approximately 11.40% through 2033, driven by the extraordinary growth of the biologics drug pipeline — including monoclonal antibodies, recombinant proteins, cell and gene therapies, and mRNA-based products — combined with the massive biosimilar manufacturing sector expansion as major biologic patents expire.
-
Contract Manufacturing Organizations (CMOs) are the fastest-growing end-user segment, expected to expand at a CAGR of approximately 10.80% through 2033, driven by pharmaceutical and biotech companies' growing preference for outsourcing manufacturing to specialized CDMOs — enabling faster speed-to-market, capital-light expansion, access to specialized manufacturing expertise, and manufacturing flexibility that internal fixed-asset production networks often cannot provide as efficiently.
-
Continuous manufacturing is the fastest-growing technology segment, projected to grow at a CAGR of approximately 12.30% through 2033, as regulatory encouragement, significant operational cost advantages, and the growing body of successfully approved products manufactured through continuous processes accelerate industry adoption of this transformative production technology across both small molecule and increasingly biologic drug manufacturing applications.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 1147.90 Billion |
| Market Size by 2025 | USD 561.88 Billion |
| Market Size by 2026 | USD 598.44 Billion |
| Market Growth Rate (2026–2033) | CAGR of 8.20% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Segments Covered | Product Type, Manufacturing Process, Technology, End User |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Rising Global Disease Burden, Biologics Pipeline Expansion, CDMO Sector Growth, and Continuous Manufacturing Technology Adoption Are the Four Pillars Driving Pharmaceutical Manufacturing Market Growth*
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising global chronic and infectious disease burden expanding medicine demand | ~34% | Global — especially North America, Europe, Asia Pacific | Short to Long-term |
| Biologics and biosimilars manufacturing capacity expansion | ~28% | North America, Europe, Asia Pacific | Short to Long-term |
| Growing pharmaceutical and biotech outsourcing to specialized CDMOs | ~24% | North America, Europe, Asia Pacific | Short to Medium-term |
| Continuous manufacturing technology adoption improving production efficiency | ~14% | North America, Europe | Medium to Long-term |
The most foundational and structurally durable driver of the Pharmaceutical Manufacturing market is the continuously expanding global medicine demand driven by population aging, rising chronic disease prevalence, improving healthcare access in emerging markets, and the ongoing development of novel therapies for previously unmet medical needs. The global burden of chronic non-communicable diseases — including cardiovascular disease, type 2 diabetes, cancer, chronic respiratory conditions, and neurological disorders — is growing in absolute patient numbers as populations age and as improving longevity extends the duration of pharmaceutical treatment per patient. This demographic and epidemiological foundation creates a structurally growing and largely recession-resistant demand base for pharmaceutical production that will sustain market expansion reliably throughout the 2026–2033 forecast period.
The pharmaceutical industry's ongoing transition toward biologics-dominated drug pipelines is simultaneously creating an extraordinary manufacturing capacity investment cycle that is one of the most commercially significant dynamics in the pharmaceutical manufacturing sector today. Biologic drugs now represent the majority of new drug approvals in major markets, and the manufacturing infrastructure required to produce monoclonal antibodies, fusion proteins, cell therapies, and gene therapies at commercial scale is both enormously capital-intensive and highly technically specialized — driving tens of billions of dollars of annual manufacturing facility and equipment investment by pharmaceutical companies and CDMOs globally. The biosimilars sector adds a further powerful layer of manufacturing demand, as the expiry of patents on major reference biologics including adalimumab, etanercept, bevacizumab, and trastuzumab creates large commercial opportunities for biosimilar manufacturers investing in the specialized cell culture, purification, and fill-finish production capabilities required.

Restraints Impact Analysis
Stringent Regulatory Compliance Requirements, High Capital Intensity of Biologic Manufacturing, Drug Supply Chain Vulnerabilities, and Skilled Workforce Shortages Restrain Faster Pharmaceutical Manufacturing Market Growth*
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Stringent FDA, EMA, and global GMP regulatory compliance requirements | ~34% | Global | Short to Long-term |
| High capital cost of establishing and operating biologic manufacturing facilities | ~28% | North America, Europe, Asia Pacific | Short to Long-term |
| Drug supply chain concentration and raw material vulnerability risks | ~24% | North America, Europe | Short to Medium-term |
| Skilled manufacturing workforce shortages in advanced pharma production | ~14% | North America, Europe | Medium-term |
The pharmaceutical manufacturing sector operates under one of the most rigorous and demanding regulatory compliance frameworks of any industry globally — with FDA Current Good Manufacturing Practice (cGMP) regulations in the United States, EMA GMP guidelines in Europe, and equivalent national regulatory standards across all major markets imposing extensive facility qualification, process validation, documentation, testing, and quality management system requirements on every aspect of pharmaceutical production. These regulatory demands are both a necessary patient safety imperative and a significant operational cost driver — requiring substantial ongoing investment in quality assurance infrastructure, regulatory affairs expertise, and compliance program management that adds meaningfully to the total cost structure of pharmaceutical manufacturing operations. FDA warning letters and import alerts resulting from GMP compliance failures can have severe commercial consequences for pharmaceutical manufacturers — including facility shutdowns, product recalls, and loss of major customer contracts — making regulatory compliance management a mission-critical operational priority.
The growing geographic concentration of global API manufacturing in India and China — which together supply an estimated 60–80% of the world's active pharmaceutical ingredient production — has created meaningful drug supply chain vulnerability that became acutely apparent during the COVID-19 pandemic when supply disruptions threatened the availability of critical medicines across North America and Europe. This supply chain concentration risk is now motivating governments and pharmaceutical companies to invest in manufacturing reshoring and geographic diversification — with the U.S. BIOSECURE Act and European pharmaceutical strategy both advancing policies to expand domestic and allied-country pharmaceutical manufacturing capabilities. While this diversification investment is a long-term positive for the pharmaceutical manufacturing market, the short-term transition costs and operational complexity of establishing new manufacturing sites create near-term investment demands that can strain company capital allocation priorities.
Opportunities Impact Analysis
Cell and Gene Therapy Manufacturing Scaling, mRNA Platform Technology Expansion, Pharmaceutical Manufacturing Reshoring, and Digital Manufacturing Technology Adoption Create the Most Significant Growth Opportunities*
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Cell and gene therapy commercial manufacturing scale-up | ~32% | North America, Europe | Short to Medium-term |
| mRNA vaccine and therapeutic manufacturing platform expansion | ~28% | North America, Europe, Asia Pacific | Short to Medium-term |
| Pharmaceutical manufacturing reshoring and geographic supply chain diversification | ~26% | North America, Europe | Medium to Long-term |
| Digital manufacturing and Industry 4.0 technology adoption | ~14% | North America, Europe, Asia Pacific | Medium to Long-term |
The commercial-scale manufacturing of cell and gene therapies represents the most transformative and high-value growth opportunity in the Pharmaceutical Manufacturing market over the forecast period. These highly personalized, complex biological therapies — including CAR-T cell therapies, viral vector gene therapies, and stem cell-based products — require entirely new manufacturing paradigms, specialized facilities, and novel process technologies that are fundamentally different from conventional pharmaceutical production. With the cell and gene therapy clinical pipeline containing hundreds of candidates advancing toward commercialization, the demand for specialized manufacturing infrastructure, raw materials, analytical tools, and CDMO services tailored to these therapies is growing extraordinarily rapidly — creating a large and premium-value market expansion opportunity for manufacturers and equipment suppliers that can establish strong positions in this technically demanding sector.
The global pharmaceutical manufacturing reshoring movement — driven by government policy incentives, national security concerns about API supply chain vulnerability, and post-pandemic lessons about the risks of geographic manufacturing concentration — represents a substantial infrastructure investment opportunity for the pharmaceutical manufacturing sector. In the United States, the Biden and Trump administrations both advanced significant policy support for domestic pharmaceutical manufacturing investment, including direct government funding, tax incentives, and procurement preferences for domestically manufactured medicines. In Europe, the EU Pharmaceutical Strategy is advancing similar supply chain diversification initiatives. This policy-driven reshoring momentum is expected to drive significant new facility construction and capacity investment across North America and Europe during the forecast period — creating sustained demand for pharmaceutical manufacturing equipment, engineering services, and specialized facility components.

Segment Analysis
By Product Type
Finished Dosage Formulations Lead the Pharmaceutical Manufacturing Market Revenue While Biopharmaceuticals Emerge as the Highest-Growth Segment Driven by the Global Biologics Revolution*
Finished dosage formulations hold the dominant revenue position in the product type segment of the Pharmaceutical Manufacturing market, contributing approximately 48.30% of total global market revenue in 2025. This segment encompasses the final drug product manufacturing stage — including tablet and capsule production, sterile injectable filling, topical formulation manufacturing, and biological drug product fill-finish operations — where formulated drug substances are converted into the precise dosage forms administered to patients in clinical and community settings. The complexity, regulatory rigor, and value-add of finished dosage manufacturing make it the highest revenue-generating stage of the pharmaceutical production value chain. North America is the largest regional contributor to finished dosage formulation revenue, where advanced solid oral dose manufacturing facilities, injectable drug production infrastructure, and biological fill-finish capacity operated by industry leaders including Pfizer, Johnson & Johnson, Abbott, and Merck represent the highest-value and most technically sophisticated commercial drug product manufacturing base globally. Europe is the second-largest region, with major finished dosage manufacturing operations run by Novartis, Roche, AstraZeneca, and Sanofi across Switzerland, Germany, the United Kingdom, and Ireland.
Biopharmaceuticals — encompassing monoclonal antibodies, recombinant proteins, vaccines, cell therapies, and gene therapies — represent the fastest-growing product segment in the Pharmaceutical Manufacturing market, projected to expand at a CAGR of approximately 11.40% from 2026 to 2033. The biologics segment's extraordinary growth reflects both the dominance of biologic drugs in new pharmaceutical approvals and the massive manufacturing capacity investment cycle required to produce these complex products at commercial scale. North America leads biopharmaceutical manufacturing revenue globally — driven by the extraordinary concentration of innovative biotech and pharmaceutical companies with biologic drug portfolios including Amgen, Genentech/Roche, Regeneron, AbbVie, and Bristol-Myers Squibb — all of which operate major biological manufacturing facilities in the United States. Singapore, Ireland, and Switzerland are emerging as important additional biopharmaceutical manufacturing hubs, attracting major pharmaceutical company investments with favorable regulatory environments, skilled workforces, and strategic logistics advantages.
By End User
Pharmaceutical Companies Lead the Manufacturing Market Revenue While Contract Manufacturing Organizations Grow Fastest Driven by Industry Outsourcing Acceleration*
Large pharmaceutical companies — operating their own internal manufacturing networks — represent the dominant end-user segment in the Pharmaceutical Manufacturing market, accounting for approximately 54.60% of total global market revenue in 2025. Major integrated pharmaceutical manufacturers including Pfizer, Johnson & Johnson, Roche, Novartis, Merck, AbbVie, Bristol-Myers Squibb, and AstraZeneca operate extensive global manufacturing networks encompassing API production, biological manufacturing, finished dosage formulation, and quality control facilities — generating the largest absolute manufacturing expenditure volumes in the industry. These companies' internal manufacturing operations are supported by substantial ongoing capital investment in facility upgrades, technology modernization, continuous manufacturing implementation, and advanced quality management system deployment. North America and Europe collectively host the largest concentration of major pharmaceutical company manufacturing facilities globally, with Ireland emerging as a particularly significant European pharmaceutical manufacturing hub due to its favorable tax and regulatory environment.
Contract Manufacturing Organizations and Contract Development and Manufacturing Organizations represent the fastest-growing end-user segment in the pharmaceutical manufacturing market, projected to expand at a CAGR of approximately 10.80% from 2026 to 2033, driven by the pharmaceutical and biotech industry's accelerating preference for outsourcing manufacturing to specialized external partners. Emerging biotech companies — which typically lack the capital and scale to build internal manufacturing infrastructure — are the most intensive users of CDMO services, making pharmaceutical company CMO/CDMO outsourcing volume highly correlated with the continued strong growth of the global biotech industry. Leading CDMO organizations including Lonza Group, Catalent (acquired by Nova Holdings), Samsung Biologics, Boehringer Ingelheim Biopharmaceuticals, and Wuxi Biologics are investing aggressively in capacity expansion — particularly for biologic drug manufacturing, cell and gene therapy production, and highly potent API synthesis — to capture the growing outsourcing demand from both innovative pharmaceutical and emerging biotech clients.

Regional Insights
North America
North America Leads the Global Pharmaceutical Manufacturing Market With the World's Largest Drug Industry Concentration, Highest Biologics Manufacturing Investment, and Most Advanced Regulatory and Technology Infrastructure*
North America holds the dominant position in the global Pharmaceutical Manufacturing market, accounting for approximately 36.40% of total global market revenue in 2025, and is projected to maintain its regional leadership at a CAGR of approximately 7.80% from 2026 to 2033. The United States is the primary driver within the region — home to the world's largest pharmaceutical industry by revenue, the most extensive biologics manufacturing infrastructure, the highest per-capita medicine consumption, and the FDA regulatory framework that sets global pharmaceutical manufacturing quality standards. Major U.S.-based pharmaceutical manufacturers including Pfizer, Johnson & Johnson, Merck, AbbVie, Bristol-Myers Squibb, and Amgen operate some of the world's most advanced Pharmaceutical Manufacturing facilities for both small molecule and biologic drug production. The region is also experiencing a manufacturing reshoring investment surge — with significant new facility investments from both domestic companies and international pharmaceutical manufacturers establishing U.S. production presence — that is adding meaningful new manufacturing capacity and revenue to the North American market.
Canada contributes to regional market revenue through its growing generic pharmaceutical manufacturing sector and expanding contract manufacturing base, with companies including Apotex and several international CDMO operators building significant Canadian production capabilities. The North American pharmaceutical manufacturing outlook for 2026–2033 is strongly positive, reinforced by continued record levels of biopharmaceutical R&D spending generating growing pipeline molecules requiring clinical and commercial manufacturing, progressive cell and gene therapy commercialization creating new specialized manufacturing demand, and sustained policy support for domestic pharmaceutical supply chain strengthening that is motivating broad-based manufacturing investment across the region.
Asia Pacific
Asia Pacific Is the Fastest-Growing Pharmaceutical Manufacturing Market, Powered by India's Generic and API Manufacturing Dominance, China's Biopharmaceutical Expansion, and Southeast Asia's Emerging Production Capabilities*
Asia Pacific is the fastest-growing regional segment in the Pharmaceutical Manufacturing market, projected to expand at a CAGR of approximately 9.60% from 2026 to 2033 — the highest of any region globally. India is the single most important driver of Asia Pacific pharmaceutical manufacturing revenue and growth — holding the position as the world's largest exporter of generic medicines and a globally critical API production hub, with companies including Sun Pharmaceutical Industries, Dr. Reddy's Laboratories, Cipla, Lupin, and Aurobindo Pharma collectively supplying pharmaceutical products to over 200 countries and territories. China represents the region's most dynamic growth market for Pharmaceutical Manufacturing — with rapidly expanding biopharmaceutical production capabilities, aggressive domestic innovative drug pipeline development, and major CDMO operators including WuXi Biologics and WuXi AppTec building world-class biologics and specialty chemical manufacturing infrastructure that is attracting growing international pharmaceutical outsourcing volume.
South Korea and Japan are mature and high-quality pharmaceutical manufacturing markets within Asia Pacific, with South Korea's Samsung Biologics and Celltrion representing globally competitive biologics and biosimilar manufacturing capabilities, while Japan's established pharmaceutical industry hosts advanced solid dose and biological manufacturing operations by both domestic companies and major international pharma partners. Singapore has established itself as a premier pharmaceutical manufacturing hub in Southeast Asia — attracting major biologics manufacturing investments by Pfizer, GSK, Sanofi, and Takeda due to its world-class regulatory infrastructure, skilled workforce, and strategic supply chain position for serving Asia Pacific markets. The combined growth momentum of India, China, South Korea, Singapore, and Japan positions Asia Pacific as both the most commercially dynamic and fastest-expanding regional market in global pharmaceutical manufacturing through 2033.
Top Key Players
-
Pfizer Inc. (United States)
-
Johnson & Johnson (United States)
-
Roche Holding AG (Switzerland)
-
Novartis AG (Switzerland)
-
Merck & Co. Inc. (United States)
-
AbbVie Inc. (United States)
-
AstraZeneca plc (United Kingdom)
-
Bristol-Myers Squibb Company (United States)
-
Lonza Group AG — CDMO (Switzerland)
-
Samsung Biologics Co. Ltd. (South Korea)
-
WuXi Biologics Co. Ltd. (China)
-
Catalent Inc. — Nova Holdings (United States)
-
Sun Pharmaceutical Industries Ltd. (India)
-
Boehringer Ingelheim GmbH (Germany)
-
Sanofi S.A. (France)
Recent Developments
-
Pfizer Inc. (2025) — Announced a significant expansion of its biologics manufacturing capacity at its McPherson, Kansas facility — one of the company's largest global manufacturing investments — targeting increased production of injectable biologics and biosimilar products for the U.S. and international markets, reflecting Pfizer's strategic commitment to strengthening its internal biologics manufacturing infrastructure following the massive capacity scale-up experience gained during COVID-19 vaccine production.
-
Lonza Group (2024) — Completed the acquisition of a major cell and gene therapy manufacturing facility from a leading biotech company, significantly expanding Lonza's CDMO capabilities for advanced therapy medicinal products and reinforcing its position as the world's leading contract manufacturer for biopharmaceuticals — particularly in the fast-growing and commercially highly valuable cell and gene therapy manufacturing segment.
-
Samsung Biologics (2025) — Announced the initiation of construction on its fifth biomanufacturing facility in Incheon, South Korea — adding approximately 180000 liters of additional bioreactor capacity and making Samsung Biologics the world's single largest biologics contract manufacturer by total installed capacity — targeting the growing global demand for outsourced biologics and biosimilar manufacturing from major pharmaceutical and biotech clients worldwide.
-
AstraZeneca (2024) — Invested over USD 1.5 billion in expanding its biologics and small molecule pharmaceutical manufacturing capabilities across multiple global sites including the United States, Ireland, and Sweden — advancing both internal production capacity for its oncology, respiratory, and cardiovascular drug portfolios and strengthening the manufacturing infrastructure required to support its extensive late-stage clinical development pipeline approaching commercialization.
-
WuXi Biologics (2025) — Despite navigating the complex regulatory and geopolitical landscape arising from the U.S. BIOSECURE Act discussions, continued to advance capacity expansion at its European and Irish manufacturing facilities — strategically diversifying its geographic production footprint beyond China to serve international pharmaceutical customers seeking supply chain geographic diversification from their Chinese CDMO partners while maintaining uninterrupted biologics manufacturing service capabilities.
Market Trends
The Accelerating Commercialization of Cell and Gene Therapy Manufacturing and the Progressive Industry-Wide Adoption of Continuous Manufacturing and Digital Quality Systems Are the Two Defining Trends Reshaping the Pharmaceutical Manufacturing Market*
The most transformative trend reshaping the Pharmaceutical Manufacturing market is the accelerating transition from development-stage to commercial-scale manufacturing of cell and gene therapies — a class of highly personalized, complex biological medicines that require fundamentally new production paradigms, specialized closed-system manufacturing equipment, novel raw material supply chains, and entirely new quality control approaches relative to conventional pharmaceutical manufacturing. As an increasing number of CAR-T cell therapies, AAV gene therapies, and stem cell-based products receive regulatory approval and enter commercial production, the pharmaceutical manufacturing ecosystem is investing heavily in developing the specialized infrastructure, automated processes, and manufacturing expertise required to produce these highly complex, patient-specific or allogeneic therapies at both clinical and commercial scale. This technology transition is creating significant new manufacturing equipment demand, CDMO capacity investment, and workforce development requirements that will define important market growth dynamics through 2033.
The progressive pharmaceutical industry adoption of continuous manufacturing — replacing traditional batch production with uninterrupted, integrated flow-based drug synthesis and processing — represents the second major manufacturing technology trend that is steadily reshaping pharmaceutical production economics and capabilities. The FDA's active encouragement of continuous manufacturing adoption through regulatory guidance and manufacturing technology innovation partnerships is accelerating industry confidence in this approach, while the demonstrated economic benefits — including 30–50% reductions in production cycle times, improved process consistency, lower facility footprint, and significant cost per unit reductions — are making the business case for continuous manufacturing investment increasingly compelling for pharmaceutical companies of all sizes. As more products achieve regulatory approval through continuous manufacturing processes and as the equipment cost and complexity of transitioning from batch to continuous production decreases with technology maturity, adoption across both new facility investments and existing manufacturing line upgrades is expected to accelerate meaningfully during the forecast period.
Segments Covered in the Report
By Product Type:
-
Finished Dosage Formulations
-
Active Pharmaceutical Ingredients (API)
-
Biopharmaceuticals
-
Generics
-
Biosimilars
-
Vaccines
-
Over-the-Counter Products
By Manufacturing Process:
-
Batch Manufacturing
-
Continuous Manufacturing
-
Sterile Manufacturing
-
Non-Sterile Manufacturing
By Technology:
-
Traditional Manufacturing
-
Advanced Manufacturing Technologies
-
Single-Use Technologies
-
Continuous Processing
By End User:
-
Pharmaceutical Companies
-
Contract Manufacturing Organizations (CMOs)
-
Hospitals and Clinics
-
Academic and Research Institutions
By Region:
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the Pharmaceutical Manufacturing market and what is its projected value by 2033?
Answer: The Pharmaceutical Manufacturing market was valued at USD 561.88 billion in 2025 and is projected to reach USD 1147.90 billion by 2033. It is expected to grow at a CAGR of 8.20% from 2026 to 2033, driven by rising global disease burden, biologics manufacturing expansion, growing CDMO outsourcing, and continuous manufacturing technology adoption.
Question 2: What are the primary growth drivers of the Pharmaceutical Manufacturing market?
Answer: Rising global chronic and infectious disease burden expanding medicine demand and the pharmaceutical industry's transition toward biologics-dominated drug pipelines requiring specialized, capital-intensive manufacturing infrastructure are the two most powerful drivers of the Pharmaceutical Manufacturing market. The accelerating adoption of contract manufacturing outsourcing and continuous manufacturing technology are additional important structural growth contributors.
Question 3: Which region leads the Pharmaceutical Manufacturing market and which is the fastest growing?
Answer: North America dominates the Pharmaceutical Manufacturing market with approximately 36.40% of global revenue in 2025, led by the world's largest pharmaceutical industry concentration and most advanced biologics manufacturing infrastructure. Asia Pacific is the fastest-growing region at approximately 9.60% CAGR through 2033, driven by India's generic and API manufacturing dominance, China's expanding biopharmaceutical capabilities, and South Korea's world-class CDMO sector.
Question 4: Which product type and end-user segment lead the Pharmaceutical Manufacturing market?
Answer: Finished dosage formulations lead the Pharmaceutical Manufacturing market product segment with approximately 48.30% of total revenue in 2025, while biopharmaceuticals are the fastest-growing product type at approximately 11.40% CAGR through 2033. Pharmaceutical companies are the dominant end-user with approximately 54.60% market share, while contract manufacturing organizations are the fastest-growing segment at approximately 10.80% CAGR.
Question 5: How is the cell and gene therapy revolution impacting the Pharmaceutical Manufacturing market?
Answer: The commercialization of cell and gene therapies is creating an entirely new and highly specialized segment of the Pharmaceutical Manufacturing market — requiring novel closed-system manufacturing equipment, automated cell processing platforms, viral vector production infrastructure, and specialized CDMO services that command significant premium pricing relative to conventional pharmaceutical manufacturing. This technology transition is driving major capacity investment by leading CDMOs including Lonza, Samsung Biologics, and WuXi Biologics, and is expected to be one of the most commercially significant pharmaceutical manufacturing growth drivers through 2033.
