Nasal Aspirate Testing Market Overview
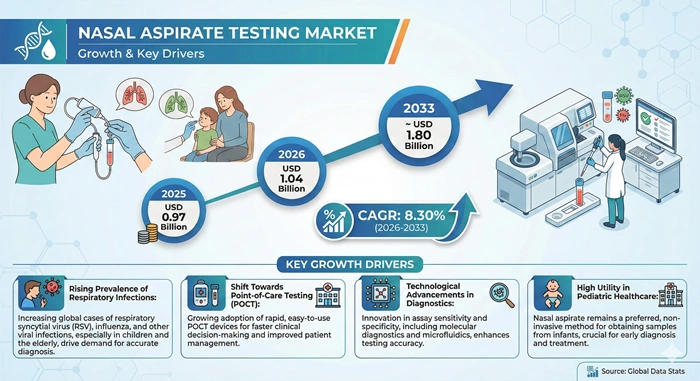
The global nasal aspirate testing market size is valued at USD 0.97 billion in 2025 and is predicted to increase from USD 1.04 billion in 2026 to approximately USD 1.80 billion by 2033, growing at a CAGR of 8.30% from 2026 to 2033.
Nasal aspirate testing represents a critical diagnostic method involving the collection of nasal secretions through gentle suction to identify respiratory pathogens causing infections. This non-invasive procedure provides healthcare professionals with valuable samples for detecting viruses, bacteria, and other microorganisms responsible for respiratory illnesses affecting millions globally. The technique proves particularly effective for pediatric patients, as it causes minimal discomfort compared to nasopharyngeal swabs while delivering highly accurate diagnostic results. Growing awareness about early disease detection, coupled with the rising burden of respiratory infections including influenza, respiratory syncytial virus, and emerging viral threats, continues driving substantial demand for nasal aspirate testing solutions across hospitals, diagnostic laboratories, and point-of-care settings.
AI Impact on the Nasal Aspirate Testing Industry
Revolutionizing Diagnostic Accuracy and Operational Efficiency Through Intelligent Technologies
Artificial intelligence and machine learning technologies are fundamentally transforming the nasal aspirate testing market by enhancing diagnostic speed, improving result accuracy, and streamlining laboratory workflows across healthcare facilities worldwide. AI-powered image recognition systems analyze microscopic samples from nasal aspirates with remarkable precision, identifying pathogen characteristics and distinguishing between viral, bacterial, and fungal infections faster than traditional manual examination methods. These intelligent algorithms process complex patterns in test results, reducing human error while enabling earlier detection of respiratory infections that require immediate medical intervention. Machine learning models trained on vast datasets of respiratory infection profiles help laboratories predict seasonal outbreak patterns, optimize testing capacity, and allocate resources efficiently during high-demand periods such as flu season.
Automation driven by AI integration has revolutionized sample processing procedures within diagnostic laboratories, with robotic systems handling specimen preparation, reagent dispensing, and result interpretation with minimal human intervention. These automated platforms reduce turnaround times from hours to minutes, enabling rapid clinical decision-making that improves patient outcomes while reducing hospital burden. AI-enhanced quality control systems continuously monitor testing processes, flagging potential errors or contamination issues before they compromise result reliability. Digital platforms incorporating AI analytics aggregate nasal aspirate testing data across healthcare networks, supporting epidemiological surveillance initiatives that track infection spread patterns and inform public health responses. The integration of AI with point-of-care testing devices enables real-time results delivery directly to clinicians' mobile devices, facilitating immediate treatment decisions particularly valuable in emergency and pediatric care settings.
Growth Factors
Rising Respiratory Infection Burden and Technological Advances Propelling Market Expansion
The nasal aspirate testing market experiences robust growth driven primarily by the escalating global prevalence of respiratory infections affecting populations across all age groups, with particular vulnerability among children, elderly individuals, and immunocompromised patients. Respiratory diseases remain among the leading causes of morbidity and mortality worldwide, with seasonal influenza alone affecting millions annually and respiratory syncytial virus causing severe illness in infants and young children. The COVID-19 pandemic fundamentally increased awareness about respiratory diagnostics importance, establishing testing infrastructure that continues supporting market expansion for broader respiratory pathogen detection. Healthcare systems worldwide recognize early accurate diagnosis as critical for appropriate treatment selection, infection control implementation, and prevention of disease transmission within communities and healthcare facilities.
Technological innovations in molecular diagnostics represent another critical growth catalyst, with next-generation PCR platforms and rapid molecular tests delivering results with unprecedented sensitivity and specificity while reducing testing timeframes. Multiplex testing capabilities enable simultaneous detection of multiple respiratory pathogens from single nasal aspirate samples, improving diagnostic efficiency and clinical utility. The development of portable point-of-care testing devices brings sophisticated diagnostic capabilities directly to patient bedsides, emergency departments, and remote healthcare facilities previously dependent on centralized laboratory services. Immunoassay rapid antigen tests offer cost-effective screening solutions with results available within minutes, supporting triage decisions and reducing unnecessary hospitalizations. Growing emphasis on personalized medicine and targeted antimicrobial therapy drives demand for accurate pathogen identification, as healthcare providers seek to optimize treatment selection while combating antimicrobial resistance through appropriate medication use.

Market Outlook
Promising Growth Trajectory Supported by Innovation and Expanding Clinical Applications
The nasal aspirate testing market outlook reflects exceptional growth prospects characterized by continuous technological advancement, expanding application beyond traditional respiratory diagnostics, and increasing adoption across diverse healthcare settings globally. North America maintains market leadership through 2033, supported by advanced diagnostic infrastructure, high testing volumes driven by robust disease surveillance programs, favorable reimbursement policies from government and private insurers, and presence of major diagnostic companies driving innovation. The region benefits from established clinical protocols recommending nasal aspirate testing for specific patient populations, particularly pediatric cases where sample collection methods significantly impact patient comfort and diagnostic yield. Strong research and development investments from biotechnology companies and academic institutions continuously advance testing methodologies, improving accuracy while reducing costs.
Asia Pacific region emerges as the fastest-growing market with projected CAGR substantially exceeding global averages, driven by rapidly expanding healthcare infrastructure, increasing respiratory disease burden across densely populated regions, rising healthcare expenditure, and growing awareness about early diagnostic benefits. Countries including China, India, Japan, and Southeast Asian nations demonstrate particularly strong growth dynamics, reflecting government initiatives strengthening infectious disease surveillance systems, expanding diagnostic laboratory networks, and improving healthcare access in rural areas. The region's large pediatric population creates substantial demand for child-friendly diagnostic methods like nasal aspirate testing. Market expansion also benefits from increasing adoption of point-of-care testing solutions in primary care settings, enabling earlier diagnosis and treatment initiation closer to patients' homes. Direct-to-consumer testing models and telemedicine integration represent emerging opportunities, as digital health platforms facilitate remote specimen collection guidance and results delivery.
Expert Speaks
-
Leadership from diagnostic testing companies emphasizes the critical importance of non-invasive sample collection methods particularly for pediatric respiratory infection diagnosis, highlighting how nasal aspirate testing delivers laboratory-quality specimens with significantly less patient discomfort compared to nasopharyngeal swabs, improving patient experience while maintaining diagnostic accuracy essential for appropriate treatment decisions.
-
Executives from molecular diagnostics companies discuss revolutionary advances in multiplex PCR testing platforms enabling simultaneous detection of 15-20 respiratory pathogens from single nasal aspirate samples, dramatically improving diagnostic efficiency while reducing unnecessary antibiotic prescriptions through accurate pathogen identification supporting antimicrobial stewardship initiatives across healthcare systems worldwide.
-
Clinical laboratory directors highlight expanding adoption of rapid molecular testing at point-of-care locations including emergency departments and urgent care clinics, noting that delivering accurate results within 15-30 minutes rather than 24-48 hours transforms clinical workflows, enables immediate treatment initiation, and reduces unnecessary hospital admissions particularly valuable during seasonal respiratory illness surges.
Key Report Takeaways
-
North America leads the nasal aspirate testing market with a dominant market share of approximately 44.10% in 2025, driven by advanced diagnostic infrastructure, high testing adoption rates, comprehensive insurance reimbursement coverage, and presence of major diagnostic companies including Abbott, Roche Diagnostics, and QuidelOrtho maintaining strong market positions.
-
Asia Pacific region demonstrates the fastest projected CAGR during the forecast period from 2026 to 2033, fueled by expanding healthcare infrastructure, rising respiratory disease burden across large population bases, increasing government investments in public health programs, improving diagnostic capabilities, and growing awareness about early infection detection benefits.
-
Molecular testing segment dominates the nasal aspirate testing market, accounting for substantial market share of approximately 58.40% in 2025, attributed to superior diagnostic accuracy, ability to detect multiple pathogens simultaneously, declining costs making advanced testing accessible, and growing clinical adoption of PCR-based diagnostics across healthcare facilities.
-
Viral infections application represents the largest clinical indication segment with considerable market share of approximately 72.60% in 2025, driven by high prevalence of respiratory viral infections including influenza, RSV, COVID-19, and other seasonal pathogens creating sustained demand for accurate diagnostic testing.
-
Immunoassay rapid antigen testing segment projects the fastest growth rate with anticipated high CAGR through 2033, supported by advantages including rapid results delivery within 10-15 minutes, cost-effectiveness compared to molecular tests, ease of use enabling point-of-care deployment, and expanding test availability for multiple respiratory pathogens.
-
Point-of-care testing centers end-user segment demonstrates significant growth potential with strong projected CAGR, driven by shift toward decentralized testing models, development of portable testing devices, advantages of immediate results supporting clinical decision-making, and expanding urgent care and retail clinic networks offering respiratory diagnostic services.
Market Scope
| Report Coverage Details | |
|---|---|
| Market Size by 2033 | USD 1.80 Billion |
| Market Size by 2025 | USD 0.97 Billion |
| Market Size by 2026 | USD 1.04 Billion |
| Market Growth Rate from 2026 to 2033 | CAGR of 8.30% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2033 |
| Segments Covered | Test Type, Application, Device Type, End User, Region |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
Market Dynamics
Drivers Impact Analysis
Escalating Respiratory Disease Prevalence and Diagnostic Innovation Accelerating Market Growth
| Drivers | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising respiratory infection prevalence | +2.8% | Global, particularly pediatric populations | 2026-2033 |
| Technological advances in molecular diagnostics | +2.2% | North America, Europe, Asia Pacific | 2026-2031 |
| Increasing point-of-care testing adoption | +1.7% | Global | 2027-2033 |
| Growing awareness of non-invasive testing benefits | +1.3% | Global, especially pediatric care | 2026-2030 |
The accelerating global burden of respiratory infections represents the primary driver propelling the nasal aspirate testing market forward, as viral and bacterial respiratory illnesses affect hundreds of millions of individuals annually across all age demographics. Influenza viruses cause seasonal epidemics affecting 5-15% of global population yearly, while respiratory syncytial virus remains the leading cause of lower respiratory tract infections in infants and young children, accounting for substantial hospitalizations and healthcare costs. The emergence of novel respiratory pathogens including new coronavirus variants and influenza strains creates ongoing demand for accurate diagnostic testing enabling appropriate clinical management and public health responses. Pediatric populations demonstrate particularly high respiratory infection rates, with children experiencing 6-10 upper respiratory infections annually on average, creating sustained testing demand across pediatric hospitals, urgent care facilities, and primary care practices.
Technological innovations in molecular diagnostics have dramatically transformed nasal aspirate testing capabilities, with advanced PCR platforms delivering highly sensitive pathogen detection from minimal sample volumes while reducing turnaround times. Multiplex molecular tests enable simultaneous identification of 15-20 respiratory pathogens from single specimens, improving diagnostic efficiency while reducing costs compared to sequential single-pathogen testing. The development of isothermal amplification technologies including loop-mediated isothermal amplification and recombinase polymerase amplification enables rapid molecular testing without expensive thermal cycler equipment, facilitating point-of-care deployment in resource-limited settings. Rapid antigen immunoassays incorporating improved antibody technologies demonstrate enhanced sensitivity approaching molecular test performance while maintaining speed and cost advantages. The integration of digital health platforms with testing devices enables seamless results transmission to electronic health records, supporting clinical decision support systems and epidemiological surveillance programs tracking infection patterns across populations.

Restraints Impact Analysis
Sample Quality Challenges and Cost Considerations Creating Market Headwinds
| Restraints | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Sample collection technique variability | -0.9% | Global | 2026-2033 |
| High costs of molecular testing equipment | -0.7% | Emerging markets | 2026-2029 |
| Limited reimbursement in certain regions | -0.5% | Latin America, Africa, parts of Asia | 2026-2030 |
| Competition from alternative sampling methods | -0.4% | Global | 2027-2033 |
Sample collection technique variability represents a significant challenge limiting nasal aspirate testing market expansion, as proper specimen collection requires trained personnel using appropriate equipment and standardized procedures to ensure adequate sample quality for accurate diagnosis. Inconsistent collection methods yield insufficient cellular material or inadequate viral loads, potentially causing false negative results that delay appropriate treatment and compromise patient care. Healthcare facilities must invest in staff training programs teaching proper nasal aspirate collection techniques, while equipment maintenance and quality control protocols require ongoing resource allocation. The learning curve associated with mastering collection procedures, particularly for inexperienced personnel, can initially reduce diagnostic yield until proficiency develops through repeated practice and supervision.
The high capital costs associated with advanced molecular testing platforms create barriers limiting market penetration particularly in resource-constrained healthcare settings including rural hospitals, small diagnostic laboratories, and facilities in developing regions. PCR instruments capable of processing nasal aspirate samples require investments ranging from tens of thousands to hundreds of thousands of dollars, while reagent costs and maintenance expenses create ongoing operational burdens. Smaller healthcare facilities often struggle to justify these investments given limited testing volumes, potentially limiting patient access to advanced diagnostic capabilities. Limited or inconsistent insurance reimbursement for nasal aspirate testing in certain regions and clinical scenarios creates financial barriers affecting test utilization, as healthcare providers may choose less expensive but potentially less accurate alternatives when reimbursement remains uncertain. Competition from alternative sampling methods including nasopharyngeal swabs, which require less equipment investment and demonstrate comparable diagnostic performance for certain applications, potentially limits nasal aspirate testing adoption particularly in non-pediatric populations.
Opportunities Impact Analysis
Emerging Technologies and Expanding Applications Opening New Growth Pathways
| Opportunities | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Home-based testing development | +1.4% | North America, Europe | 2028-2033 |
| Artificial intelligence integration | +1.1% | Global | 2027-2033 |
| Emerging pathogen surveillance | +0.9% | Global | 2026-2033 |
| Telemedicine integration | +0.7% | North America, Europe, Asia Pacific | 2026-2030 |
The development of home-based nasal aspirate testing represents a substantial emerging opportunity, as consumer demand for convenient at-home diagnostic solutions continues growing following COVID-19 pandemic experiences with self-testing. Companies developing user-friendly nasal aspirate collection kits with clear instructions enable patients to collect high-quality samples at home, shipping specimens to laboratories for analysis or using integrated rapid testing devices providing immediate results. This model particularly appeals to parents seeking convenient testing options for children experiencing respiratory symptoms, reducing emergency department visits and clinic exposures during infectious illness. Partnerships between diagnostic companies and telehealth platforms create integrated care pathways supporting remote specimen collection guidance, digital results delivery, and virtual physician consultations for treatment recommendations.
Artificial intelligence integration throughout testing workflows presents significant opportunities for market advancement, as machine learning algorithms enhance pathogen identification accuracy, optimize laboratory operations, and support predictive epidemiology enabling proactive public health responses. AI-powered digital pathology systems analyze nasal aspirate samples with superhuman speed and consistency, identifying subtle pathogen characteristics missed by human examination while reducing skilled technician workload. Predictive analytics leveraging historical testing data and real-time results enable healthcare systems to anticipate respiratory illness surges, optimize staffing levels, and ensure adequate testing supplies during high-demand periods. The growing focus on emerging pathogen surveillance creates opportunities for nasal aspirate testing expansion, as public health agencies establish sentinel surveillance networks monitoring respiratory infections for novel variants and zoonotic spillover events requiring rapid detection and characterization. Integration with blockchain technologies enables secure, transparent sample tracking and results verification supporting international health regulations and cross-border disease monitoring.

Segment Analysis
By Test Type
Molecular Testing Dominates with Superior Accuracy and Comprehensive Pathogen Detection
Molecular testing represents the largest test type segment within the nasal aspirate testing market, commanding substantial market share of approximately 58.40% in 2025 due to unparalleled diagnostic accuracy, ability to detect multiple respiratory pathogens simultaneously, and growing clinical adoption across hospital laboratories and diagnostic centers. These sophisticated tests utilize polymerase chain reaction technology and other nucleic acid amplification methods to identify genetic material from viruses, bacteria, and other microorganisms present in nasal aspirate samples with exceptional sensitivity and specificity. Real-time PCR platforms detect extremely low pathogen concentrations, enabling early diagnosis before symptoms fully develop or when viral loads decline during late infection stages. Multiplex PCR assays simultaneously test for 15-20 respiratory pathogens from single specimens, including influenza A and B, respiratory syncytial virus, human metapneumovirus, adenovirus, rhinovirus, and SARS-CoV-2, providing comprehensive diagnostic information guiding appropriate treatment decisions. North America leads the molecular testing segment, with major diagnostic companies including Abbott, Roche Diagnostics, Thermo Fisher Scientific, and Qiagen maintaining dominant market positions through advanced testing platforms, extensive laboratory service networks, and continuous innovation enhancing assay performance.
The molecular testing segment projects robust growth with CAGR approximating 8.0% through 2033, supported by declining test costs making advanced diagnostics accessible to broader healthcare facilities, technological improvements reducing turnaround times to under one hour for certain platforms, expanding test menus incorporating newly discovered pathogens, and growing recognition of molecular testing value for antimicrobial stewardship programs. Asia Pacific emerges as the fastest-growing regional market for molecular testing, driven by substantial government investments in laboratory infrastructure, increasing adoption of advanced diagnostics in tertiary hospitals, growing awareness among healthcare providers about molecular testing benefits, and expanding domestic manufacturing capabilities reducing import dependence. Leading regional players including Chinese and Indian diagnostic companies increasingly develop competitive molecular testing platforms tailored to local market needs and price points, while international companies including Illumina and Bio-Rad establish regional manufacturing and distribution partnerships. Recent innovations include development of isothermal amplification technologies enabling rapid molecular testing without thermal cycling equipment, portable testing devices suitable for point-of-care deployment, and integration with artificial intelligence for automated result interpretation.
By Application
Viral Infections Lead Market with High Prevalence and Testing Demand
The viral infections application segment dominates the nasal aspirate testing market, capturing considerable market share of approximately 72.60% in 2025 and representing the largest clinical indication category reflecting the overwhelming prevalence of viral respiratory infections compared to bacterial or fungal causes. Respiratory viruses including influenza, respiratory syncytial virus, rhinovirus, adenovirus, human metapneumovirus, parainfluenza, and coronaviruses account for the vast majority of upper and lower respiratory tract infections across all age groups globally. Seasonal influenza alone causes 3-5 million severe illnesses annually worldwide, while RSV hospitalizes 60,000-80,000 children under five years old in the United States each year. The COVID-19 pandemic dramatically increased viral respiratory testing volumes, establishing infrastructure and clinical protocols that continue supporting broader respiratory pathogen testing. Accurate viral identification enables appropriate clinical management including antiviral therapy when indicated, prevents unnecessary antibiotic prescriptions reducing antimicrobial resistance, and supports infection control measures limiting transmission in healthcare and community settings.
North America maintains the largest market share in the viral infections application segment, supported by comprehensive viral testing availability across hospital emergency departments, urgent care clinics, pediatric practices, and reference laboratories, established clinical guidelines recommending testing for specific patient populations and clinical presentations, and favorable insurance reimbursement for medically indicated viral diagnostics. Healthcare providers routinely order nasal aspirate testing for hospitalized patients with respiratory symptoms, immunocompromised individuals at high complication risk, and pediatric patients requiring accurate diagnosis guiding treatment decisions. The segment projects steady growth with CAGR approximating 8.5% through 2033, driven by continued high respiratory viral infection burden, expanding multiplex testing adoption enabling comprehensive pathogen panel screening, development of new antiviral therapies requiring diagnostic confirmation before treatment initiation, and growing recognition of long-term complications from certain viral infections including RSV. Asia Pacific demonstrates particularly strong growth dynamics, with increasing viral testing adoption driven by rising respiratory infection awareness, expanding pediatric healthcare services, government programs strengthening disease surveillance capabilities, and improving laboratory infrastructure supporting advanced molecular diagnostics across urban and rural regions.

Regional Insights
North America
Market Leadership Through Advanced Diagnostic Infrastructure and Strong Clinical Adoption
North America maintains its position as the dominant regional market for nasal aspirate testing, commanding approximately 44.10% market share in 2025 and generating the highest absolute revenue among all geographic regions globally. This leadership position reflects the region's sophisticated diagnostic testing infrastructure characterized by extensive hospital laboratory networks, numerous independent diagnostic facilities, widespread point-of-care testing deployment, and advanced molecular diagnostics capabilities serving diverse clinical needs. The United States represents the largest country market within North America, distinguished by high respiratory testing volumes driven by robust infectious disease surveillance programs, established clinical practice guidelines recommending nasal aspirate testing for specific patient populations particularly pediatric cases, comprehensive insurance reimbursement from Medicare, Medicaid, and private payers supporting test accessibility, and presence of major diagnostic companies driving continuous innovation. Emergency departments, urgent care clinics, pediatric hospitals, and primary care practices routinely utilize nasal aspirate testing for rapid respiratory pathogen identification, supporting immediate treatment decisions and infection control measures. The region projects moderate CAGR approximating 7.80% through 2033, driven by aging population increasing respiratory infection vulnerability, expanding point-of-care testing adoption, technological innovations enhancing test performance and ease of use, and growing integration with digital health platforms.
Leading diagnostic and pharmaceutical companies headquartered or maintaining substantial operations across North America dominate the regional nasal aspirate testing market, with Abbott (United States), Roche Diagnostics (United States operations), QuidelOrtho (United States), Thermo Fisher Scientific (United States), Becton Dickinson (United States), and Bio-Rad Laboratories (United States) representing key market participants. These companies leverage established distribution networks spanning hospital systems, reference laboratories, physician offices, and retail clinics, strong brand recognition built through decades of diagnostic testing leadership, comprehensive regulatory expertise navigating FDA clearance processes, and substantial research investments advancing testing technologies. Academic medical centers including Mayo Clinic, Cleveland Clinic, and Johns Hopkins maintain specialized respiratory diagnostics laboratories conducting testing while advancing clinical research validating new methodologies. Canada represents another important market within the region, characterized by universal healthcare coverage ensuring diagnostic test accessibility, growing respiratory testing adoption particularly during seasonal illness periods, and government support for infectious disease surveillance programs. Recent market dynamics include Abbott's USD 23 billion acquisition of Exact Sciences announced in November 2025, strengthening diagnostic capabilities and laboratory infrastructure supporting potential nasal aspirate testing expansion.
Asia Pacific
Fastest Growth Driven by Healthcare Infrastructure Expansion and Rising Disease Burden
Asia Pacific emerges as the fastest-growing regional market for nasal aspirate testing, projected to expand at the highest CAGR from 2026 to 2033, substantially exceeding growth rates in mature markets and establishing this region as the critical focus area for diagnostic companies pursuing expansion strategies. This exceptional growth trajectory reflects multiple converging factors including rapidly increasing respiratory disease burden across densely populated regions, substantial government investments in healthcare infrastructure development, rising healthcare expenditure enabling advanced diagnostic adoption, expanding diagnostic laboratory networks serving urban and rural communities, and growing awareness among healthcare providers and patients about early respiratory infection detection benefits. Countries across the region demonstrate varied development stages creating diverse market opportunities, with established markets including Japan, South Korea, and Australia maintaining advanced diagnostic capabilities while emerging markets including China, India, Indonesia, Philippines, and Vietnam experience rapid healthcare sector expansion and diagnostic testing infrastructure development.
Major international diagnostic companies and regional manufacturers operating across Asia Pacific play pivotal roles in market development, with Abbott, Roche Diagnostics, Thermo Fisher Scientific, and Becton Dickinson maintaining strong regional presence through local partnerships, distribution agreements, and manufacturing facilities supporting growing testing demand. Domestic companies based in countries including China, India, Japan, and South Korea increasingly develop competitive nasal aspirate testing solutions leveraging cost-effective production capabilities, deep understanding of local healthcare systems and market dynamics, and government support for domestic medical device industry development. India demonstrates particularly strong growth potential, driven by large pediatric population creating substantial respiratory testing demand, expanding private hospital networks offering advanced diagnostic services, government initiatives improving rural healthcare access, and growing medical tourism sector. The region's healthcare systems increasingly prioritize infectious disease surveillance following pandemic experiences, supporting nasal aspirate testing adoption for routine respiratory pathogen monitoring. Traditional cultural acceptance of proactive healthcare and preventive medicine in many Asian societies facilitates diagnostic testing adoption, while expanding middle-class populations gain improved healthcare access and willingness to invest in quality medical services.
Top Key Players
-
Abbott Laboratories (United States)
-
F. Hoffmann-La Roche Ltd (Switzerland)
-
QuidelOrtho Corporation (United States)
-
Thermo Fisher Scientific Inc. (United States)
-
Becton, Dickinson and Company (United States)
-
Bio-Rad Laboratories Inc. (United States)
-
Qiagen N.V. (Netherlands)
-
Hologic Inc. (United States)
-
bioMérieux SA (France)
-
Luminex Corporation (United States)
-
Cepheid (United States)
-
Mesa Laboratories Inc. (United States)
Recent Developments
-
Abbott (2025) – Announced agreement to acquire cancer diagnostics company Exact Sciences for approximately USD 23 billion in November 2025, significantly expanding Abbott's diagnostic testing capabilities and laboratory infrastructure that could support enhanced respiratory diagnostics portfolio including potential nasal aspirate testing service expansion across broader geographic markets.
-
QuidelOrtho (2025) – Announced strategic initiative to accelerate growth in molecular diagnostics including intent to acquire full ownership of LEX Diagnostics after receiving FDA 510(k) clearance in March 2025, strengthening the company's molecular testing platform capabilities supporting respiratory pathogen detection including nasal aspirate sample processing.
-
Abbott (2024) – Continued expanding point-of-care rapid molecular testing solutions throughout 2024, with Abbott ID NOW platform widely deployed across pharmacies, urgent care clinics, and physician offices enabling rapid respiratory pathogen detection from nasal samples within 15 minutes, supporting immediate clinical decision-making and reducing unnecessary antibiotic prescriptions.
-
Roche Diagnostics (2024) – Advanced cobas respiratory testing panels throughout 2024, offering comprehensive multiplex PCR assays detecting 15-20 respiratory pathogens simultaneously from nasal aspirate and other specimen types, enabling efficient laboratory workflows while providing clinicians complete diagnostic information guiding optimal patient management decisions.
-
Multiple Diagnostic Companies (2024) – Expanded COVID-19/Flu/RSV triple combination rapid antigen test availability throughout 2024, with improved formulations demonstrating enhanced sensitivity particularly for nasal sample types, supporting efficient screening during respiratory illness season and enabling simultaneous detection of three most common viral respiratory pathogens.
Market Trends
Point-of-Care Testing Expansion and Multiplex Diagnostics Reshaping Clinical Practice
The nasal aspirate testing market experiences significant transformation driven by accelerating shift toward point-of-care testing models bringing sophisticated diagnostic capabilities directly to patient encounter locations including emergency departments, urgent care facilities, physician offices, and retail clinics. Portable molecular testing devices delivering laboratory-quality results within 15-30 minutes enable immediate clinical decision-making, reducing unnecessary hospitalizations and emergency department wait times while improving patient satisfaction through faster diagnosis and treatment initiation. This decentralization trend particularly impacts pediatric respiratory care, as parents increasingly seek convenient testing options close to home rather than traveling to centralized hospital laboratories. Rapid antigen tests continue improving sensitivity and specificity through enhanced antibody technologies and optimized sample processing methods, narrowing performance gaps with molecular tests while maintaining speed and cost advantages. The COVID-19 pandemic accelerated point-of-care testing infrastructure development and clinical acceptance, with established workflows and reimbursement models now supporting broader respiratory pathogen testing applications.
Multiplex diagnostic testing adoption represents another major trend, as healthcare providers recognize value of comprehensive respiratory pathogen panels identifying multiple organisms simultaneously from single nasal aspirate samples. These panels eliminate diagnostic uncertainty by testing for all common respiratory viruses and bacteria in parallel rather than sequential testing based on clinical suspicion, improving diagnostic accuracy while reducing costs and specimen requirements compared to multiple individual tests. The integration of digital health technologies including electronic health record connections, mobile results delivery, and telemedicine platforms enhances testing workflows and patient communication. Laboratory information systems automatically transmit results to clinicians' devices, triggering clinical decision support alerts for abnormal findings requiring immediate intervention. Consumer-facing mobile applications enable patients to schedule testing appointments, receive results notifications, and access educational resources about respiratory infections and prevention strategies. Blockchain integration supporting secure sample tracking and results verification addresses privacy concerns while ensuring data integrity for regulatory compliance and public health reporting.
Segments Covered in the Report
By Test Type
-
Molecular Testing (PCR, Isothermal Amplification, Next-Generation Sequencing)
-
Immunoassay Testing (ELISA, Chemiluminescence)
-
Rapid Antigen Testing (Lateral Flow Assays, Immunochromatography)
-
PCR Testing (Real-Time PCR, Multiplex PCR)
-
Others (Culture-Based Methods, Direct Fluorescent Antibody)
By Application
-
Viral Infections (Influenza, RSV, COVID-19, Rhinovirus, Adenovirus, Human Metapneumovirus)
-
Bacterial Infections (Streptococcus Pneumoniae, Haemophilus Influenzae, Mycoplasma Pneumoniae)
-
Fungal Infections
-
Others
By Device Type
-
Manual Aspirators
-
Electric Aspirators
-
Others
By End User
-
Hospitals (Emergency Departments, Pediatric Units, General Wards)
-
Diagnostic Laboratories (Reference Labs, Hospital Labs, Independent Labs)
-
Point-of-Care Testing Centers (Urgent Care Clinics, Physician Offices, Retail Clinics)
-
Homecare Settings
-
Others
By Region
-
North America (United States, Canada, Mexico)
-
Europe (Germany, United Kingdom, France, Italy, Spain, Rest of Europe)
-
Asia Pacific (China, India, Japan, South Korea, Australia, Southeast Asia, Rest of Asia Pacific)
-
Latin America (Brazil, Argentina, Rest of Latin America)
-
Middle East and Africa (GCC Countries, South Africa, Rest of Middle East and Africa)
Frequently Asked Questions
Question 1: What is the expected nasal aspirate testing market size by 2033?
Answer: The global nasal aspirate testing market is expected to reach approximately USD 1.80 billion by 2033, growing from USD 1.04 billion in 2026. This substantial growth reflects increasing respiratory infection prevalence, technological advances in molecular diagnostics, and expanding point-of-care testing adoption across healthcare settings.
Question 2: Which region dominates the nasal aspirate testing market currently?
Answer: North America dominates the nasal aspirate testing market with approximately 44.10% market share in 2025, supported by advanced diagnostic infrastructure and high testing adoption rates. The region benefits from established clinical guidelines recommending testing and comprehensive insurance reimbursement coverage.
Question 3: What factors are driving growth in the nasal aspirate testing market?
Answer: The nasal aspirate testing market growth is driven by rising respiratory infection prevalence creating sustained diagnostic demand, technological advances in molecular testing improving accuracy and speed, increasing point-of-care testing adoption, and growing awareness about non-invasive sample collection benefits particularly for pediatric patients. Government investments in infectious disease surveillance further support market expansion.
Question 4: Which test type segment leads the nasal aspirate testing market?
Answer: Molecular testing leads the nasal aspirate testing market with substantial market share of approximately 58.40% in 2025, attributed to superior diagnostic accuracy and multiplex capabilities. PCR platforms enable simultaneous detection of multiple respiratory pathogens, providing comprehensive diagnostic information guiding optimal patient management.
Question 5: What role does AI play in the nasal aspirate testing market?
Answer: AI technologies are transforming the nasal aspirate testing market by enhancing diagnostic speed and improving result accuracy through machine learning algorithms analyzing test results. AI-powered automation streamlines laboratory workflows, reduces turnaround times, and supports predictive epidemiology enabling proactive public health responses to respiratory infection outbreaks.
