Medical Tubing Market Overview
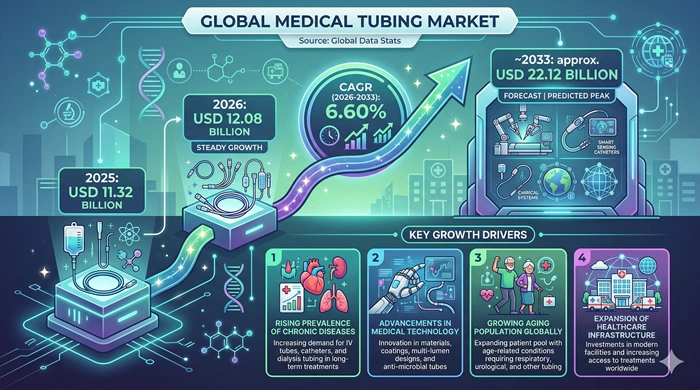
The global Medical Tubing market size is valued at USD 11.32 billion in 2025 and is predicted to increase from USD 12.08 billion in 2026 to approximately USD 22.12 billion by 2033, growing at a CAGR of 6.60% from 2026 to 2033.
Medical tubing encompasses a broad and critically important category of precision-manufactured tubular components — produced from materials including silicone, polyvinyl chloride (PVC), polyurethane, thermoplastic elastomers, fluoropolymers, and natural latex — used across an extensive range of healthcare applications including intravenous fluid delivery, drug infusion, catheterization, respiratory support, surgical drainage, endoscopic procedures, and medical device fluid management. These components form the essential circulatory infrastructure of modern clinical medicine — enabling the controlled and sterile transfer of fluids, gases, and drugs within patient care pathways across hospital critical care, surgical, diagnostic, and home healthcare settings. The Medical Tubing market is experiencing sustained growth driven by expanding global healthcare infrastructure, rising chronic disease patient populations requiring long-term device-based care, advancing minimally invasive surgical technique adoption, and continuous material science and precision manufacturing innovation.
AI Impact on the Medical Tubing Industry
Artificial Intelligence Is Transforming Medical Tubing Design Optimization, Quality Control Inspection, Predictive Failure Analysis, and Smart Healthcare System Integration in Ways That Are Meaningfully Elevating Tubing Performance, Safety, and Manufacturing Efficiency*
Artificial intelligence is making a growing and practically significant impact across the Medical Tubing market, most immediately through the application of AI-powered computer vision and machine learning quality inspection systems in tubing manufacturing environments — where automated defect detection algorithms analyze real-time production line camera feeds to identify dimensional inconsistencies, surface defects, material voids, and contamination events with speed and consistency far exceeding the capabilities of manual visual inspection. These AI-driven quality control systems are enabling medical tubing manufacturers to achieve higher product quality consistency, reduce defect escape rates, lower scrap and rework costs, and generate the comprehensive quality data documentation required by FDA and EU MDR regulatory compliance programs — collectively improving manufacturing productivity and product safety in ways that strengthen customer confidence and support premium market positioning for quality-focused tubing suppliers. Companies including Freudenberg Medical, Teleflex, and Saint-Gobain Performance Plastics are investing in AI-enhanced manufacturing quality systems as a strategic competitiveness priority in their medical tubing production operations.
AI is also accelerating innovation in medical tubing material formulation and structural design — with computational modeling and machine learning platforms being applied to simulate the mechanical performance, biocompatibility characteristics, and fluid flow dynamics of novel tubing material combinations and geometric configurations before physical prototype production. These AI-assisted design tools compress the new product development cycle for specialized medical tubing products — reducing the time and cost required to develop and validate tubing solutions for new medical device applications — and enabling closer engineering collaboration between tubing manufacturers and medical device OEM customers who require highly customized and application-specific tubing components. As the medical device industry moves toward increasingly miniaturized, precise, and multi-functional device architectures, AI-assisted tubing design optimization will become progressively more important in enabling the Medical Tubing market to deliver the specialized components that next-generation medical devices demand.
Growth Factors
Expanding Global Healthcare Infrastructure, Rising Chronic Disease Prevalence Driving Device-Based Care, Minimally Invasive Surgical Technique Adoption, and Advanced Material Innovation Are the Core Growth Drivers*
The most fundamental and durable driver of the Medical Tubing market is the continuous global expansion of healthcare infrastructure — encompassing hospital capacity additions, ambulatory surgical center development, diagnostic laboratory network growth, and home healthcare service expansion — that collectively increase the total volume of medical procedures performed and the quantity of medical tubing components consumed annually worldwide. Healthcare infrastructure investment is particularly robust in Asia Pacific and Latin America — where government health system modernization programs, private hospital group expansion, and international development funding are driving rapid growth in clinical facility numbers and procedural capacity — creating large and growing incremental demand for the diverse range of medical tubing products used in routine clinical care, surgical procedures, and patient monitoring applications. In established North American and European healthcare markets, continuous growth in procedure volumes — driven by aging populations, rising chronic disease prevalence, and expanding healthcare access coverage — maintains strong and consistent baseline demand for medical tubing products across all application categories.
The rapidly rising global prevalence of chronic diseases — including cardiovascular disease, diabetes, chronic kidney disease, cancer, and chronic respiratory conditions — is creating a large and growing patient population requiring long-term device-based medical care that generates sustained and high-volume medical tubing demand. Hemodialysis patients — of whom there are an estimated 3.5 million globally with numbers growing — require regular tubing set replacements during each dialysis session, generating enormous recurring demand for specialized dialysis tubing products. Cancer patients receiving chemotherapy infusion, diabetes patients using insulin delivery devices and continuous glucose monitoring systems, and cardiovascular patients with implanted pacemakers and cardiac catheters all represent high-volume and clinically critical tubing product consumption segments. The expanding and aging global patient population with these chronic conditions creates a structurally growing, recession-resistant demand base for medical tubing that underpins confident long-term market growth projections through the forecast period.

Market Outlook
With Global Healthcare Procedure Volumes Rising, Advanced Polymer Material Innovation Expanding Application Possibilities, Single-Use Device Adoption Driving Disposable Tubing Demand, and Emerging Market Healthcare Investment Accelerating, the Medical Tubing Market Outlook Is Strongly Positive Through 2033*
The long-term outlook for the Medical Tubing market is firmly positive and underpinned by multiple converging and structurally durable growth drivers expected to sustain healthy expansion throughout the 2026–2033 forecast period. A particularly important commercial driver shaping the market's future trajectory is the accelerating global adoption of single-use and disposable medical device policies — motivated by patient safety imperatives, infection control requirements, and the operational simplification benefits of eliminating reprocessing programs for complex fluid pathway components. As hospitals, surgical centers, and home healthcare programs progressively transition from reusable to single-use device approaches across an expanding range of clinical applications, the total volume of disposable medical tubing components consumed per procedure increases — driving unit volume growth for tubing manufacturers that exceeds underlying procedure volume growth rates and creates favorable demand dynamics for the broader industry.
The continuous advancement of specialty medical tubing material science and precision manufacturing technology — including developments in biocompatible polymer formulations, antimicrobial tubing surface treatments, drug-eluting catheter coatings, ultra-thin wall precision extrusion capabilities, and multi-lumen co-extrusion manufacturing — is progressively enabling the Medical Tubing market to serve an expanding range of sophisticated medical device applications with tubing solutions that deliver superior performance, patient safety, and clinical outcomes. These technical innovations are creating premium value segments within the medical tubing industry where specialty material expertise and precision manufacturing capability command meaningful price premiums and generate strong customer loyalty from medical device OEM partners seeking reliable, technically differentiated tubing component supply relationships. As medical device complexity, miniaturization requirements, and performance specifications continue to advance, the technical barriers to entry in high-value specialty medical tubing segments are rising — creating durable competitive advantages for established specialty tubing manufacturers with deep polymer science and precision extrusion expertise.
Expert Speaks
-
Geoff Martha, CEO, Medtronic plc — "The reliability, biocompatibility, and precision dimensional consistency of medical tubing components are absolutely critical to the clinical performance and patient safety of Medtronic's catheter-based cardiovascular and neurovascular device portfolio — making our relationships with high-quality medical tubing material and component suppliers a strategic priority. As we continue to advance minimally invasive device technology, the demands we place on medical tubing performance will only grow more sophisticated."
-
Tom Polen, CEO, Becton Dickinson — "Medical fluid pathway integrity — critically dependent on the quality and reliability of tubing and catheter components — is fundamental to patient safety in IV drug delivery, infusion therapy, and vascular access applications that represent core markets for BD. We are continuously investing in material science and tubing technology advancement to ensure our infusion system and vascular access products meet the highest standards of clinical performance and infection control."
-
Kevin Lobo, CEO, Stryker Corporation — "The miniaturization of surgical instruments and endoscopic devices is placing increasingly demanding technical requirements on the medical tubing components used within these systems — requiring precision multi-lumen architectures, enhanced kink resistance, and biocompatible surface properties that push the boundaries of conventional tubing manufacturing. Our partnership approach with specialty medical tubing suppliers is a critical enabler of Stryker's continued innovation in minimally invasive surgical device technology."
Key Report Takeaways
-
North America dominates the Medical Tubing market, holding approximately 38.40% of global market share in 2025, driven by the world's largest medical device industry generating high-volume specialty tubing demand, the most advanced healthcare infrastructure with the highest per-capita medical procedure rates, stringent FDA regulatory quality standards that favor established domestic and North American tubing suppliers, and the concentration of leading medical tubing manufacturers including Freudenberg Medical, Teleflex, and Integer Holdings in the region.
-
Asia Pacific is the fastest-growing regional market for medical tubing, projected to expand at a CAGR of approximately 8.20% from 2026 to 2033, driven by rapid healthcare infrastructure expansion across China, India, and Southeast Asia, growing domestic medical device manufacturing capability requiring local tubing component supply, rising chronic disease procedure volumes, and expanding government healthcare investment programs across the region's rapidly developing economies.
-
Silicone is the dominant material segment, accounting for approximately 28.60% of total market revenue in 2025, reflecting silicone's unparalleled combination of exceptional biocompatibility, broad chemical resistance, outstanding temperature stability, superior flexibility across a wide durometer range, and long-term implantable safety — making it the material of choice for the most demanding and highest-value medical tubing applications including implantable catheter systems, peristaltic pump tubing, and critical care fluid management devices.
-
Catheters and cannulas represent the largest application segment, contributing approximately 32.80% of total market revenue in 2025, reflecting the extraordinary volume and diversity of catheter-based clinical applications across cardiovascular, urological, neurological, respiratory, and vascular access medicine — each requiring specialized tubing material formulations, dimensional specifications, and surface treatment technologies from precision medical tubing manufacturers.
-
Single-lumen tubing remains the dominant structural segment by volume, while multi-lumen and co-extruded tubing are the fastest-growing structural segments — projected to expand at a CAGR of approximately 8.80% through 2033 — driven by the growing demand for complex multi-channel tubing architectures in sophisticated minimally invasive surgical devices, advanced catheter systems, and multi-function endoscopic instruments that require multiple independent fluid and instrumentation pathways within a single compact tubing profile.
-
Home healthcare is the fastest-growing end-user segment, expected to expand at a CAGR of approximately 9.40% through 2033, driven by the accelerating global transition of chronic disease management — including dialysis, infusion therapy, enteral nutrition, and respiratory support — from inpatient hospital settings to lower-cost home environments, creating rapidly growing demand for patient-friendly, reliable, and safe medical tubing components in home-use medical device applications.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 22.12 Billion |
| Market Size by 2025 | USD 11.32 Billion |
| Market Size by 2026 | USD 12.08 Billion |
| Market Growth Rate (2026–2033) | CAGR of 6.60% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Segments Covered | Material, Structure, Application, End User |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Expanding Healthcare Infrastructure, Rising Chronic Disease Prevalence, Single-Use Device Policy Adoption, and Minimally Invasive Surgical Growth Are the Four Pillars of Medical Tubing Market Expansion*
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Expanding global healthcare infrastructure driving procedure volume growth | ~34% | Global — especially Asia Pacific, Latin America | Short to Long-term |
| Rising chronic disease prevalence increasing device-dependent patient care volumes | ~28% | Global | Short to Long-term |
| Single-use and disposable device policy adoption increasing per-procedure tubing consumption | ~24% | North America, Europe, Asia Pacific | Short to Medium-term |
| Minimally invasive and endoscopic surgical technique adoption driving specialized tubing demand | ~14% | North America, Europe | Short to Medium-term |
The most enduring and broadly applicable driver of the Medical Tubing market is the continuous global expansion of healthcare delivery infrastructure and clinical procedure volumes — encompassing the addition of new hospital beds, surgical theaters, intensive care units, dialysis centers, catheterization laboratories, and outpatient endoscopy units that collectively increase the total number of clinical procedures performed and the quantity of medical tubing components consumed across all application categories. Healthcare infrastructure investment is accelerating particularly rapidly across Asia Pacific — where China's sustained investment in tier 2 and tier 3 city hospital modernization, India's government-sponsored Ayushman Bharat hospital capacity expansion program, and Southeast Asia's private hospital group growth are generating substantial incremental demand for the full spectrum of standard and specialty medical tubing products used in routine clinical operations. This infrastructure-driven demand growth creates a predictable and highly durable commercial foundation for Medical Tubing market expansion throughout the forecast period.
The globally rising burden of chronic diseases — and the progressive medical management of these conditions through device-based therapeutic and monitoring approaches that consume high volumes of medical tubing products — is simultaneously reinforcing structural demand growth across the most important application segments of the Medical Tubing market. The expanding global dialysis patient population generates exceptionally high and recurring tubing product demand — with each hemodialysis session consuming a complete set of blood tubing, arteriovenous lines, and dialyzer connections that must be replaced with new sterile components at every use. Similarly, the growing global population of cancer patients receiving IV chemotherapy infusion, patients with central venous access catheters for long-term therapy administration, and elderly patients requiring enteral nutrition tubing for chronic nutritional support collectively generate enormous and growing volumes of medical tubing product consumption that are expected to expand consistently throughout the 2026–2033 forecast period.

Restraints Impact Analysis
Stringent Regulatory Compliance Costs, Raw Material Price Volatility, Environmental Sustainability Concerns About Medical Plastics, and Supply Chain Disruption Vulnerability Restrain Faster Market Growth*
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High regulatory compliance costs for medical grade material qualification and device approval | ~33% | Global — especially North America, Europe | Short to Long-term |
| Raw material price volatility affecting polymer feedstock and specialty compound costs | ~28% | Global | Short to Medium-term |
| Environmental regulatory pressure on single-use PVC and non-recyclable medical plastics | ~26% | Europe, North America | Medium to Long-term |
| Supply chain concentration vulnerability from limited specialty polymer supplier base | ~13% | Global | Short to Medium-term |
The Medical Tubing market operates within an exceptionally demanding and costly regulatory environment — where materials used in medical tubing products must meet rigorous biocompatibility testing requirements under ISO 10993, dimensional and mechanical performance specifications defined by medical device OEM customers, sterilization compatibility validation for the intended sterilization method, and for implantable or long-term contact applications, additional safety and toxicology assessment requirements that require extensive and expensive laboratory testing programs. The regulatory qualification cost and timeline for new medical grade polymer materials and tubing formulations — particularly in markets governed by FDA 21 CFR regulations and EU Medical Device Regulation — creates meaningful barriers to product innovation and new material adoption that slow the pace of technical advancement and increase the development investment required to bring new specialty tubing products to market. These high compliance costs disproportionately burden smaller and emerging market tubing manufacturers — reinforcing the competitive advantages of established players with large regulatory affairs infrastructure and pre-qualified material libraries.
Environmental sustainability concerns about the healthcare industry's reliance on single-use plastic medical devices — including PVC and non-recyclable polymer medical tubing products — are generating growing regulatory and institutional pressure in European and North American markets to reduce medical plastic waste through material substitution, device design modification, and end-of-life recycling program development. The European Union's comprehensive Plastics Strategy and evolving medical device waste management regulations are creating compliance planning requirements for medical tubing manufacturers selling into European markets — particularly regarding the use of plasticizer additives such as DEHP in PVC tubing products that have faced scrutiny due to potential endocrine disruption concerns. While full PVC tubing replacement with alternative materials is a complex and long-term transition challenge — given the performance, cost, and regulatory qualification requirements involved — the direction of regulatory travel is clearly toward progressive material substitution, creating product development investment obligations for manufacturers and moderate near-term commercial disruption in the PVC-heavy commodity tubing segment.
Opportunities Impact Analysis
Advanced Antimicrobial and Drug-Eluting Tubing Development, Home Healthcare Expansion Driving New Product Categories, Emerging Market Local Manufacturing Growth, and Biodegradable Medical Polymer Innovation Create Compelling Opportunities*
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Antimicrobial and infection-resistant specialty tubing development for catheter applications | ~33% | North America, Europe | Short to Medium-term |
| Home healthcare expansion creating new medical tubing product category demand | ~28% | North America, Europe, Asia Pacific | Short to Long-term |
| Emerging market local medical device manufacturing growth driving regional tubing supply demand | ~26% | Asia Pacific, Latin America | Medium to Long-term |
| Sustainable and biodegradable medical polymer tubing development | ~13% | Europe, North America | Medium to Long-term |
The development and commercialization of antimicrobial and infection-resistant specialty medical tubing — incorporating silver ion technology, antimicrobial polymer additives, or surface-active coatings that inhibit bacterial biofilm formation on catheter and tubing surfaces — represents one of the most commercially promising and clinically important innovation opportunities in the Medical Tubing market. Catheter-associated bloodstream infections, urinary tract infections, and ventilator-associated pneumonia collectively represent a major category of preventable healthcare-associated infections that impose enormous patient safety and economic costs on healthcare systems — with catheter and tubing surface contamination a primary contributing pathway. Specialty tubing manufacturers including Freudenberg Medical and Saint-Gobain are investing in antimicrobial tubing platform development as a premium value-added product category that commands significant price premiums, builds strong clinical customer loyalty, and addresses one of the most practically important patient safety challenges in device-based clinical care.
The rapid global expansion of home healthcare — driven by the progressive shift of chronic disease management including dialysis, infusion therapy, respiratory support, and enteral nutrition from institutional to home settings — is creating a large and growing new product category opportunity for medical tubing manufacturers to develop patient-friendly, simplified, and home-use optimized tubing solutions for devices designed for self-administration by non-clinical users. Home healthcare tubing products must balance the same rigorous safety and performance requirements of institutional medical tubing with additional design considerations including patient ease of use, simplified connection systems, intuitive color coding, robust kink resistance for daily handling by untrained users, and cost structures compatible with home healthcare reimbursement frameworks. Manufacturers that successfully develop differentiated home healthcare tubing product portfolios — optimized for the specific requirements of this rapidly growing care setting — will capture significant incremental revenue growth opportunities as the home healthcare market expands substantially during the forecast period.

Segment Analysis
By Material
Silicone Dominates the Medical Tubing Market Material Segment While Thermoplastic Elastomers Emerge as the Fastest-Growing Material Alternative*
Silicone holds the dominant position within the material segment of the Medical Tubing market, accounting for approximately 28.60% of total global market revenue in 2025, driven by its exceptional combination of physical properties that make it uniquely suitable for the most demanding and highest-value medical tubing applications. Silicone's outstanding biocompatibility — validated across decades of clinical use in implantable and body-contact applications — combined with its broad temperature stability, superior flexibility, chemical inertness, and resistance to biological degradation makes it the preferred material for critical care IV tubing, peristaltic pump tubing, implantable catheter systems, and long-term indwelling medical device applications where material integrity and patient safety are paramount. North America is the largest regional market for silicone medical tubing — driven by the concentration of high-value specialty medical device manufacturing and the premium clinical application focus of U.S. healthcare — while Asia Pacific is the fastest-growing region for silicone tubing adoption as local medical device manufacturing capability advances. Freudenberg Medical, Saint-Gobain Performance Plastics, Trelleborg, and NewAge Industries are among the leading silicone medical tubing manufacturers with strong global supply capabilities for demanding OEM applications.
Thermoplastic elastomers (TPE) represent the fastest-growing material segment in the Medical Tubing market, projected to expand at a CAGR of approximately 8.40% from 2026 to 2033, driven by their attractive combination of silicone-like flexibility and biocompatibility with thermoplastic processing characteristics that enable higher-volume, lower-cost manufacturing compared to silicone extrusion — and their freedom from the plasticizer and regulatory concerns associated with conventional PVC tubing. TPE medical tubing is gaining rapid commercial adoption as a technically credible and cost-competitive alternative material in applications where PVC tubing is facing environmental regulatory pressure — including IV administration sets, blood pressure monitoring lines, and enteral feeding tubing — with leading polymer companies including Kraton, Teknor Apex, and PolyOne (now Avient) developing medical-grade TPE compounds specifically optimized for healthcare tubing applications. Europe is the fastest-growing region for TPE medical tubing adoption, driven by the EU's environmental regulatory pressure on conventional medical plastics and the progressive reformulation of medical device fluid pathway components to eliminate DEHP-plasticized PVC.
By Application
Catheters and Cannulas Lead the Medical Tubing Market Application Revenue While Drug Delivery System Tubing Records the Fastest Growth*
Catheters and cannulas represent the largest application segment in the Medical Tubing market, accounting for approximately 32.80% of total global market revenue in 2025, reflecting the extraordinary clinical breadth and commercial depth of catheter-based medical procedures across cardiovascular, urological, neurological, vascular access, and respiratory medicine. The catheter and cannula application segment encompasses an enormous diversity of specialized tubing product requirements — from ultra-soft and radiopaque cardiac catheter shafts to rigid urological catheter materials, from micro-diameter neurological catheter tubes to large-bore thoracic drainage systems — each requiring carefully formulated material specifications, precision-controlled dimensional tolerances, and application-specific surface or mechanical properties that drive premium product value. North America dominates catheter tubing revenue globally — driven by the world's highest interventional cardiology, electrophysiology, and vascular procedure volumes — while Asia Pacific is the fastest-growing region for catheter tubing demand as cardiac catheterization laboratory networks expand rapidly across China, India, and Southeast Asia. Leading suppliers of catheter tubing components include Freudenberg Medical, Putnam Plastics, Resonetics, and Microspec — all of which specialize in precision-engineered specialty catheter tubing for medical device OEM customers globally.
Drug delivery system tubing represents the fastest-growing application segment in the Medical Tubing market, projected to expand at a CAGR of approximately 9.20% from 2026 to 2033, driven by the rapidly expanding global market for biologic drug infusion therapy, targeted oncology drug delivery, wearable insulin patch delivery systems, and implantable drug pump applications — all of which require highly specialized tubing components with exceptional drug compatibility, low drug adsorption characteristics, and precise flow path geometry. The extraordinary commercial growth of biologic and specialty pharmaceutical therapies — which often require IV infusion administration and place stringent requirements on tubing material compatibility to prevent drug degradation, adsorption, or contamination — is generating strong and growing demand for premium drug delivery tubing products. Europe and North America are the primary markets for premium drug delivery tubing — driven by the concentration of biopharmaceutical manufacturing and clinical drug administration infrastructure — while global growth in oncology treatment programs is expanding drug delivery tubing demand across all major regional markets simultaneously.

Regional Insights
North America
North America Leads the Global Medical Tubing Market With the World's Largest Medical Device Industry, Most Advanced Clinical Infrastructure, and Highest Specialty Tubing Component Demand Per Procedure*
North America holds the dominant position in the global Medical Tubing market, accounting for approximately 38.40% of total global market revenue in 2025, and is projected to maintain steady leadership at a CAGR of approximately 6.20% from 2026 to 2033. The United States is overwhelmingly the primary regional revenue driver — home to the world's largest and most technically advanced medical device manufacturing industry, the highest global per-capita medical procedure rates, the most demanding regulatory quality standards that consistently favor premium specialty tubing suppliers, and the highest concentration of catheter-based cardiovascular, interventional, and endoscopic procedure volume that generates the most valuable specialty tubing demand. Leading companies dominating the North American Medical Tubing market include Freudenberg Medical, Integer Holdings, Teleflex, Putnam Plastics, and Saint-Gobain Performance Plastics — all of which maintain major U.S. manufacturing and product development operations that serve the region's sophisticated medical device OEM customer base.
Canada contributes meaningfully to regional medical tubing revenue through its well-developed hospital system and medical device manufacturing sector — particularly in the cardiovascular and orthopedic device segments where Canadian manufacturers maintain global competitiveness. The North American Medical Tubing market outlook for 2026–2033 is firmly positive — reinforced by the ongoing medical device innovation pipeline, the accelerating home healthcare market transition creating new tubing product requirements, and the continued expansion of minimally invasive and catheter-based procedural medicine that demands increasingly sophisticated specialty tubing components from precision-manufacturing expert suppliers.
Asia Pacific
Asia Pacific Is the Fastest-Growing Medical Tubing Market, Driven by Rapid Healthcare Infrastructure Expansion, Growing Domestic Medical Device Manufacturing, and Rising Clinical Procedure Volumes Across China, India, and Southeast Asia*
Asia Pacific is the fastest-growing regional segment in the Medical Tubing market, projected to expand at a CAGR of approximately 8.20% from 2026 to 2033 — the highest of any region globally. China is the most commercially important growth driver within the region — where the government's sustained investment in hospital capacity expansion, medical device industry development, and domestic healthcare manufacturing self-sufficiency is simultaneously creating demand for imported premium specialty tubing products and developing local tubing manufacturing capability that is progressively competing for standard and mid-range tubing supply within the Chinese medical device supply chain. India represents a rapidly growing second-largest Asia Pacific market — where the combination of Ayushman Bharat healthcare access expansion, private hospital group growth, and India's development as a global medical device manufacturing hub is generating strong incremental demand for both domestic and imported medical tubing products. Key international tubing companies with significant Asia Pacific market presence include Freudenberg Medical, Saint-Gobain, and Trelleborg — alongside growing domestic manufacturers in China including Kangda Intercontinental Medical Device Corporation.
Japan, South Korea, and Australia represent mature and technically sophisticated Asia Pacific medical tubing markets — characterized by advanced medical device manufacturing sectors, high clinical procedure quality standards, and strong demand for premium specialty tubing components. Southeast Asian markets — including Thailand, Malaysia, Indonesia, and Vietnam — are experiencing rapid growth in healthcare infrastructure investment and medical device manufacturing development that is progressively building substantial regional medical tubing demand. The breadth and demographic scale of Asia Pacific's healthcare expansion opportunity — spanning from Japan's sophisticated specialty tubing market through India's rapidly growing generic device manufacturing sector to Southeast Asia's first-time infrastructure build-out — positions the region as the most commercially dynamic growth frontier in global medical tubing through 2033.
Top Key Players
-
Freudenberg Medical LLC (United States)
-
Integer Holdings Corporation (United States)
-
Teleflex Incorporated (United States)
-
Saint-Gobain Performance Plastics (France)
-
Trelleborg AB — Trelleborg Healthcare and Medical (Sweden)
-
Putnam Plastics Corporation (United States)
-
Resonetics LLC (United States)
-
NewAge Industries Inc. (United States)
-
Nordson Corporation — Nordson Medical (United States)
-
Raumedic AG (Germany)
-
W. L. Gore & Associates Inc. (United States)
-
Tekni-Plex Inc. (United States)
Recent Developments
-
Freudenberg Medical (2025) — Expanded its precision medical tubing manufacturing capabilities through a strategic capital investment in advanced multi-lumen co-extrusion production lines at its facility in Carpinteria, California — significantly increasing its capacity for complex multi-lumen catheter shaft and specialty endoscope channel tubing production — enabling the company to meet growing OEM customer demand for technically sophisticated multi-function tubing architectures used in next-generation minimally invasive surgical and interventional device systems.
-
Integer Holdings (2024) — Completed the acquisition of a specialized medical cable and tubing assembly manufacturer — adding precision catheter component assembly capabilities to Integer's existing medical tubing manufacturing platform and strengthening the company's ability to supply fully integrated catheter and electrophysiology device components to cardiac rhythm management and vascular intervention device OEM customers who increasingly prefer consolidated component supply relationships from technically capable manufacturing partners.
-
Saint-Gobain Performance Plastics (2025) — Launched a new range of silicone medical tubing products incorporating proprietary antimicrobial surface treatment technology — specifically developed to reduce bacterial biofilm formation risk in catheter and fluid management tubing applications — providing healthcare customers with a premium infection-control enhanced tubing option that directly addresses the critical patient safety concern of catheter-associated infection, reinforcing Saint-Gobain's position as a technical innovator in high-value specialty medical tubing materials.
-
Trelleborg Healthcare and Medical (2024) — Announced the opening of a new medical tubing manufacturing and product development center in Malaysia — strategically positioned to serve the rapidly growing Asia Pacific medical device manufacturing market with locally produced specialty silicone and thermoplastic elastomer tubing components — reducing supply chain lead times for regional OEM customers and positioning Trelleborg to capture a larger share of the fast-growing Asia Pacific medical tubing demand during the forecast period.
-
Raumedic AG (2025) — Received FDA clearance for a new generation of neurovascular catheter tubing products featuring an innovative multi-layer co-extrusion architecture combining ultra-flexible inner layer materials with reinforced outer layer structures — delivering exceptional pushability, trackability, and kink resistance performance in complex neurovascular navigation procedures — representing a significant product development milestone that strengthens Raumedic's competitive positioning in the technically demanding and premium-value neurovascular catheter tubing segment globally.
Market Trends
The Progressive Transition Away From PVC Toward Advanced Alternative Polymer Materials and the Accelerating Demand for Precision Multi-Lumen and Specialty Coated Tubing in Next-Generation Medical Device Applications Are the Two Most Defining Trends Reshaping the Medical Tubing Market*
The most structurally significant material trend reshaping the Medical Tubing market is the progressive and accelerating transition away from conventional plasticized PVC tubing — particularly DEHP-plasticized formulations — toward alternative polymer materials including thermoplastic elastomers, polyurethane, and advanced silicone compounds that deliver equivalent or superior performance without the environmental and health concern profile associated with PVC plasticizers. This material transition is being driven by a combination of European regulatory pressure through the EU's SVHC (Substances of Very High Concern) framework, progressive hospital purchasing policy commitments to reduce PVC medical plastic usage, and the growing technical maturity of TPE and polyurethane tubing alternatives that can meet the performance requirements of IV administration, blood contact, and fluid management applications previously dominated by PVC. Medical tubing manufacturers are investing significantly in TPE and polyurethane product line development to position for this structural material transition — which is expected to progressively reshape the commodity tubing segment's material composition throughout the 2026–2033 forecast period.
The accelerating demand for precision-engineered multi-lumen, co-extruded, and specialty coated medical tubing — driven by the next generation of minimally invasive surgical instruments, complex catheter-based intervention systems, advanced endoscopic devices, and sophisticated implantable drug delivery systems — is simultaneously elevating the technical requirements and competitive positioning dynamics of the Medical Tubing market's highest-value segments. Medical device innovation is continuously pushing tubing component requirements toward smaller outer diameters, tighter dimensional tolerances, more complex multi-lumen architectures, enhanced column strength with maintained flexibility, improved radiopacity for fluoroscopic visualization, and specialized lubricious or hydrophilic surface coatings — creating a growing demand for the precision polymer extrusion expertise, advanced material science knowledge, and sophisticated quality management capabilities that characterize leading specialty medical tubing manufacturers. This technical complexity escalation is reinforcing the competitive advantages of expert specialty tubing manufacturers and creating durable barriers to entry for less technically capable competitors in the premium medical tubing segments.
Segments Covered in the Report
By Material:
-
Silicone
-
Polyvinyl Chloride (PVC)
-
Polyurethane
-
Thermoplastic Elastomers
-
Fluoropolymers
-
Latex
-
Other Materials
By Structure:
-
Single-Lumen Tubing
-
Multi-Lumen Tubing
-
Co-Extruded Tubing
-
Braided Tubing
-
Heat-Shrink Tubing
By Application:
-
Bulk Disposable Tubing
-
Catheters and Cannulas
-
Drug Delivery Systems
-
Intravenous and Peristaltic Pump Tubing
-
Endoscopy Tubing
-
Suction and Drainage Tubing
By End User:
-
Hospitals and Clinics
-
Ambulatory Surgical Centers
-
Diagnostic Laboratories
-
Home Healthcare
-
Pharmaceutical and Biotechnology Companies
By Region:
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the Medical Tubing market and what is its projected value by 2033?
Answer: The Medical Tubing market was valued at USD 11.32 billion in 2025 and is projected to reach USD 22.12 billion by 2033. It is expected to grow at a CAGR of 6.60% from 2026 to 2033, driven by expanding global healthcare infrastructure, rising chronic disease patient populations, single-use device adoption, and continuous medical tubing material and manufacturing innovation.
Question 2: What materials are most commonly used in the Medical Tubing market?
Answer: Silicone dominates the premium Medical Tubing market with approximately 28.60% of total revenue in 2025, prized for its exceptional biocompatibility, temperature stability, and flexibility across demanding clinical applications including catheters, peristaltic pump systems, and implantable devices. PVC, polyurethane, thermoplastic elastomers, and fluoropolymers are also widely used across different application segments — with thermoplastic elastomers emerging as the fastest-growing material alternative driven by their performance advantages over PVC and the growing regulatory pressure on conventional plasticized PVC formulations.
Question 3: Which region dominates the Medical Tubing market and which is the fastest growing?
Answer: North America dominates the Medical Tubing market with approximately 38.40% of global revenue in 2025, driven by the world's largest medical device industry, highest per-capita procedure rates, and most demanding specialty tubing technical requirements. Asia Pacific is the fastest-growing region at approximately 8.20% CAGR through 2033, driven by rapid healthcare infrastructure expansion, growing domestic medical device manufacturing capability, and rising clinical procedure volumes across China, India, and Southeast Asia.
Question 4: What are the key applications driving demand in the Medical Tubing market?
Answer: Catheters and cannulas represent the largest application segment in the Medical Tubing market at approximately 32.80% of total revenue — encompassing the extensive range of catheter-based procedures in cardiovascular, urological, neurological, and vascular access medicine. Drug delivery system tubing is the fastest-growing application at approximately 9.20% CAGR — driven by the expanding biologic infusion therapy market, wearable drug delivery device growth, and the stringent tubing material compatibility requirements of specialty pharmaceutical applications.
Question 5: How is the transition away from PVC affecting the Medical Tubing market?
Answer: The progressive regulatory and institutional pressure to reduce DEHP-plasticized PVC usage in medical devices is driving significant product development investment across the Medical Tubing market — as manufacturers develop and qualify thermoplastic elastomer, polyurethane, and advanced silicone alternatives that can replace conventional PVC in IV tubing, blood contact, and fluid management applications. This material transition is reshaping the commodity tubing segment's composition throughout the forecast period — creating both product development challenges and commercial opportunities for manufacturers with strong alternative polymer material expertise and established regulatory qualification capabilities.
