Medical Affairs Outsourcing Market Overview
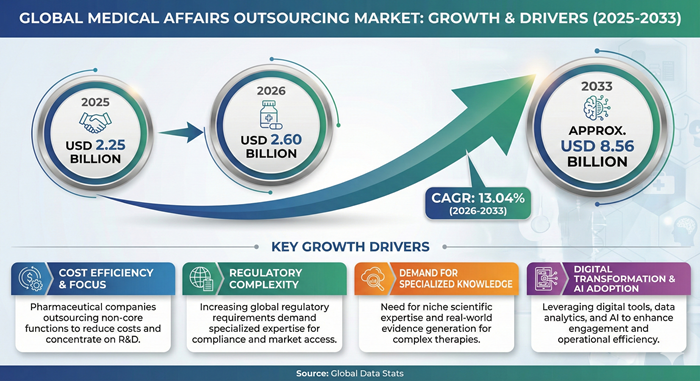
The global medical affairs outsourcing market size is valued at USD 2.25 billion in 2025 and is predicted to increase from USD 2.60 billion in 2026 to approximately USD 8.56 billion by 2033, growing at a CAGR of 13.04% from 2026 to 2033.
The industry provides specialized external support for pharmaceutical, biotechnology, and medical device companies needing expert medical affairs functions without maintaining large internal teams. These services include medical science liaison activities, scientific communications, regulatory submissions, and medical information management delivered by experienced healthcare professionals. Organizations worldwide increasingly rely on outsourcing partners to handle complex medical affairs responsibilities while focusing internal resources on core drug development activities.
The sector has become essential infrastructure supporting modern healthcare innovation as regulatory complexity and scientific specialization continue intensifying. Rising clinical trial volumes and expanding therapeutic pipelines drive steady demand for flexible, scalable medical affairs support. Companies invest strategically in outsourcing relationships to access specialized expertise, reduce operational costs, and maintain compliance across multiple global markets simultaneously.
AI Impact on the Medical Affairs Outsourcing Industry
Transforming Scientific Communication and Data Management Through Intelligent Automation
Artificial intelligence has started revolutionizing how medical affairs teams process scientific literature, analyze real-world evidence, and generate medical communications at unprecedented scale and speed. Modern AI systems scan thousands of clinical publications daily, extracting relevant safety signals, efficacy data, and competitive intelligence that would require weeks of manual review by medical science liaisons. These technologies help outsourcing providers deliver higher-quality insights faster while reducing costs for pharmaceutical clients managing extensive product portfolios. Natural language processing algorithms analyze physician inquiries, patient feedback, and healthcare provider communications to identify emerging medical information needs, enabling proactive response development before questions become widespread across medical communities.
Predictive analytics powered by AI enables medical affairs outsourcing firms to forecast regulatory submission timelines, identify potential safety concerns early, and optimize resource allocation across multiple client engagements simultaneously. Machine learning models analyze historical submission data to predict approval probabilities, helping pharmaceutical companies make informed portfolio decisions about which development programs deserve continued investment. These intelligent systems also automate routine medical writing tasks including adverse event narratives, periodic safety reports, and scientific abstracts while maintaining compliance with regulatory formatting requirements. Computer vision technologies extract data from unstructured medical records and imaging studies, accelerating real-world evidence generation supporting label expansions and new indication approvals.
Growth Factors
Rising R&D Complexity and Regulatory Burden Driving Outsourcing Adoption
The pharmaceutical industry's increasing R&D complexity creates fundamental demand for the medical affairs outsourcing market as companies develop sophisticated biologics, gene therapies, and precision medicines requiring specialized scientific expertise beyond typical internal capabilities. Modern drug development programs span multiple therapeutic areas, involve complex biomarker strategies, and navigate intricate regulatory pathways across dozens of countries simultaneously. Small and mid-sized biotech companies particularly depend on outsourced medical affairs support as they lack resources to build comprehensive internal teams covering all required specializations. Even large pharmaceutical corporations increasingly outsource specific functions like medical writing or regional medical science liaison coverage to maintain flexibility as their pipelines evolve and market priorities shift.
Regulatory requirements continue intensifying globally as health authorities demand more comprehensive safety monitoring, real-world evidence generation, and scientific communication supporting product approvals and lifecycle management. Post-marketing surveillance obligations require dedicated medical monitoring teams tracking adverse events, analyzing safety signals, and preparing regulatory submissions within strict timelines. Medical information services must respond to healthcare provider inquiries with scientifically accurate, balanced information while maintaining compliance with promotional regulations varying across jurisdictions. These expanding responsibilities strain internal medical affairs departments, pushing companies toward outsourcing arrangements providing scalable resources matching fluctuating workload demands throughout product lifecycles.

Market Outlook
Regional Expansion and Technology Integration Reshaping Industry Dynamics
North America maintains its market leadership position as the region's dominant pharmaceutical industry, sophisticated regulatory environment, and mature outsourcing culture drive substantial demand for medical affairs services. The concentration of major pharmaceutical and biotech companies in United States and established relationships with leading contract research organizations create favorable conditions for medical affairs outsourcing growth. Regulatory complexity under FDA oversight and the need for extensive scientific communication supporting value-based pricing discussions with payers sustain robust demand for specialized medical affairs expertise.
Asia Pacific emerges as the fastest-growing regional market as pharmaceutical companies increasingly conduct clinical trials and establish commercial operations throughout the region's diverse markets. Rising healthcare expenditures, growing patient populations, and expanding regulatory frameworks across China, India, and Southeast Asia create substantial medical affairs requirements. The availability of highly educated, English-speaking medical professionals at competitive labor rates attracts global outsourcing investments establishing medical affairs hubs serving both regional and international clients. Local regulatory knowledge becomes increasingly valuable as countries implement unique requirements for clinical data, safety reporting, and medical information management differing from Western standards.
Expert Speaks
-
Giovanni Caforio, former CEO of Bristol Myers Squibb – "Strategic partnerships with medical affairs service providers enable pharmaceutical companies to access specialized expertise while maintaining operational flexibility essential for managing complex product portfolios. Outsourcing allows us to scale resources efficiently matching the dynamic needs of our development pipeline and commercial operations across global markets."
-
Vas Narasimhan, CEO of Novartis – "The evolving healthcare landscape demands that pharmaceutical companies leverage external expertise to navigate regulatory complexity and deliver scientific value to healthcare providers. Medical affairs outsourcing provides access to specialized talent and advanced technologies supporting data-driven medical strategies that improve patient outcomes."
-
Albert Bourla, CEO of Pfizer – "As we advance innovative therapies across multiple therapeutic areas, partnering with experienced medical affairs organizations helps us maintain scientific rigor while accelerating timelines. These collaborations enable us to focus internal resources on strategic priorities while ensuring comprehensive medical support throughout product lifecycles."
Key Report Takeaways
-
North America dominates the global medical affairs outsourcing market with approximately 38% market share, powered by concentrated pharmaceutical industry presence, sophisticated regulatory requirements, and mature outsourcing adoption across large pharma and emerging biotech companies driving consistent service demand
-
Asia Pacific represents the fastest-growing region with projected CAGR exceeding 14.5% through 2033, driven by expanding clinical trial activity, growing pharmaceutical manufacturing, increasing regulatory complexity, and availability of cost-competitive skilled medical professionals supporting regional market development
-
Medical science liaison services lead service categories capturing roughly 32% market share as pharmaceutical companies require extensive field-based medical teams engaging healthcare providers, conducting scientific presentations, and gathering clinical insights supporting commercial strategy development
-
Pharmaceutical companies dominate end-use segments accounting for approximately 52% of market consumption as large pharma and specialty pharmaceutical firms outsource medical affairs functions including medical writing, regulatory support, and medical information management across extensive product portfolios
-
Oncology therapeutic area shows strongest growth potential among specialty segments with highest projected CAGR approaching 14.2% as complex cancer immunotherapies, targeted therapies, and combination regimens require specialized medical affairs expertise supporting clinical development and commercialization strategies
-
Medical writing and publishing services demonstrate robust expansion with substantial market share as regulatory submission volumes increase globally and pharmaceutical companies require expert medical writers producing clinical study reports, regulatory documents, and scientific publications meeting stringent quality standards
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 8.56 Billion |
| Market Size by 2025 | USD 2.25 Billion |
| Market Size by 2026 | USD 2.60 Billion |
| Market Growth Rate from 2026 to 2033 | CAGR of 13.04% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2033 |
| Segments Covered | Service Type, End-Use Industry, Therapeutic Area, Region |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Pharmaceutical Industry Evolution and Cost Pressures Accelerating Outsourcing Adoption
| Driver | Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising R&D costs and pipeline complexity | ≈ 3.8% positive impact | Global, strongest in North America and Europe | Immediate and long-term |
| Expanding regulatory requirements globally | ≈ 3.2% positive impact | Global, particularly emerging markets | Immediate and long-term |
| Growing biopharmaceutical sector and specialty therapies | ≈ 2.9% positive impact | Global, concentrated in developed markets | Immediate |
| Increasing clinical trial outsourcing and globalization | ≈ 2.1% positive impact | Asia Pacific, Latin America, Eastern Europe | Medium to long-term |
Pharmaceutical R&D cost escalation creates compelling economic incentives for the medical affairs outsourcing market as companies seek variable cost structures replacing fixed internal departments with flexible external resources scaling up or down matching pipeline needs. Developing a single new drug now costs exceeding USD 2.6 billion including failed candidates, with medical affairs activities representing substantial portions of development and commercialization budgets. Outsourcing enables pharmaceutical companies to convert fixed personnel costs into variable service fees paid only when needed, improving financial efficiency and reducing overhead during portfolio transitions. Small biotech companies with limited capital particularly benefit from outsourcing arrangements providing enterprise-grade medical affairs capabilities without requiring substantial upfront investments in staff recruitment, training, and infrastructure.
The biopharmaceutical sector's explosive growth drives specialized medical affairs demand as complex biologics, cell therapies, and gene therapies require deep scientific expertise beyond traditional small molecule drug development capabilities. These innovative therapies involve novel mechanisms of action, unique safety profiles, and sophisticated administration requirements necessitating extensive healthcare provider education and medical information support. Medical affairs teams must communicate complex scientific concepts to physicians, pharmacists, and payers while maintaining regulatory compliance around balanced, evidence-based messaging. Outsourcing partners with therapeutic area specialists provide immediate access to experts who understand intricate immunology, genetics, and molecular biology underlying modern biopharmaceutical innovations.

Restraints Impact Analysis
Data Security Concerns and Quality Control Challenges Creating Market Headwinds
| Restraint | Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Data confidentiality and intellectual property risks | ≈ 1.8% negative impact | Global, particularly for novel therapies | Immediate |
| Quality consistency and regulatory compliance concerns | ≈ 1.5% negative impact | Global, strongest in highly regulated markets | Immediate and long-term |
| Internal resistance and organizational culture barriers | ≈ 1.2% negative impact | Large pharmaceutical companies | Short to medium-term |
| Vendor management complexity and coordination challenges | ≈ 0.9% negative impact | Companies with multiple outsourcing relationships | Immediate |
Data security and confidentiality concerns create significant barriers for the medical affairs outsourcing market as pharmaceutical companies hesitate sharing proprietary clinical data, competitive intelligence, and strategic information with external vendors. Medical affairs teams access sensitive patient-level data from clinical trials, post-marketing surveillance, and real-world evidence studies requiring strict privacy protections under HIPAA, GDPR, and other regulations. Companies worry about intellectual property theft, competitive intelligence leakage, and regulatory violations resulting from inadequate vendor data security controls. These concerns intensify for novel therapies representing substantial commercial value where competitive advantages depend on maintaining confidentiality around development programs, clinical strategies, and medical positioning approaches.
Quality control consistency challenges pharmaceutical companies when outsourcing critical medical affairs functions as external teams may lack deep understanding of company culture, product portfolios, and historical decision-making context. Medical information responses must maintain consistent messaging aligned with approved labeling while addressing nuanced physician questions requiring judgment calls about off-label information boundaries. Medical writing quality varies across individual writers despite standard operating procedures, potentially resulting in regulatory submissions requiring extensive revisions delaying approval timelines. Companies struggle managing multiple outsourcing vendors simultaneously, coordinating workflows, maintaining quality standards, and ensuring seamless integration with internal teams across different functions and geographic regions.
Opportunities Impact Analysis
Digital Health Technologies and Emerging Markets Creating Growth Avenues
| Opportunity | Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Digital health integration and real-world evidence generation | ≈ 2.8% positive impact | Global, led by developed markets | Medium to long-term |
| Emerging market pharmaceutical development expansion | ≈ 3.1% positive impact | Asia Pacific, Latin America, Middle East | Immediate and long-term |
| Specialty and rare disease therapeutic growth | ≈ 2.4% positive impact | Global, concentrated in developed markets | Medium to long-term |
| Strategic partnerships and full-service outsourcing models | ≈ 1.9% positive impact | Global, particularly small-mid size pharma | Medium-term |
Digital health technology integration creates substantial opportunities for the medical affairs outsourcing market as pharmaceutical companies increasingly leverage real-world data from electronic health records, wearable devices, and patient registries supporting medical strategies. Outsourcing partners with advanced analytics capabilities help pharmaceutical clients extract meaningful insights from massive unstructured datasets identifying treatment patterns, safety signals, and comparative effectiveness evidence supporting regulatory submissions and payer negotiations. These digital capabilities enable proactive medical monitoring detecting adverse events earlier, generating real-world evidence demonstrating product value, and personalizing healthcare provider engagement based on prescribing patterns and therapeutic area interests.
Emerging market expansion presents enormous growth potential as pharmaceutical companies establish operations throughout Asia, Latin America, and Middle Eastern regions with expanding healthcare systems and growing patient access to innovative therapies. These markets require localized medical affairs support understanding regional regulatory frameworks, cultural contexts, and healthcare delivery systems differing substantially from Western markets. Outsourcing provides cost-effective solutions for pharmaceutical companies lacking established regional presence, enabling rapid market entry without substantial fixed investments in local staff and infrastructure. The availability of well-educated, multilingual medical professionals at competitive compensation rates in countries including India, Philippines, and Eastern Europe attracts global medical affairs outsourcing investments establishing regional hubs serving international clients.

Segment Analysis
Medical Science Liaison Services
Field-Based Medical Expertise Driving Segment Leadership and Strategic Value Creation
Medical science liaison services dominate the medical affairs outsourcing market with approximately 32% share in 2025, expanding at 13.5% CAGR through the forecast period. This segment encompasses field-based medical professionals engaging key opinion leaders, conducting scientific presentations at medical meetings, gathering clinical insights from healthcare providers, and supporting investigator-initiated research programs. North America represents the largest regional market as pharmaceutical companies maintain extensive MSL teams covering major academic medical centers, community oncology practices, and specialty clinics where thought leaders influence prescribing patterns and treatment guidelines. The complexity of modern therapies particularly in oncology, immunology, and rare diseases requires highly specialized MSLs with advanced degrees and deep therapeutic area expertise that many pharmaceutical companies prefer accessing through flexible outsourcing arrangements.
Leading MSL service providers including IQVIA, Syneos Health, ICON plc, and specialized firms like Amplity Health serve global pharmaceutical clients through geographically distributed teams covering diverse therapeutic areas. These organizations recruit, train, and manage MSL professionals maintaining consistent quality standards while providing pharmaceutical clients flexibility to expand or contract field teams matching product lifecycle stages and commercial priorities. Outsourced MSLs increasingly leverage digital tools including virtual meeting platforms, AI-powered scientific literature databases, and customer relationship management systems tracking healthcare provider interactions and gathering competitive intelligence. The biopharmaceutical sector shows particularly strong MSL outsourcing adoption as small biotech companies launching first products lack internal infrastructure supporting field medical teams but require expert scientific engagement with key opinion leaders influencing early adoption.
Pharmaceutical End-Use Industry
Dominant Client Segment Spanning Drug Development Through Commercialization Lifecycle
Pharmaceutical companies represent the largest end-use category capturing approximately 52% market share in 2025, growing at 12.8% CAGR as both large multinational corporations and emerging specialty pharma firms outsource diverse medical affairs functions. This segment includes branded pharmaceutical manufacturers developing and marketing innovative therapies across multiple therapeutic areas requiring comprehensive medical affairs support including medical information services, regulatory medical writing, safety surveillance, and scientific communications. Europe shows particularly strong pharmaceutical outsourcing adoption as regional pharmaceutical companies navigate complex multi-country regulatory frameworks across EU member states while managing medical affairs operations in diverse languages and healthcare systems. Cost pressures from payer pricing negotiations and generic competition drive pharmaceutical companies toward outsourcing models reducing fixed costs while maintaining high-quality medical affairs capabilities.
Major pharmaceutical clients including both large corporations and mid-sized specialty companies work with established outsourcing partners like Parexel, Covance, PPD, and specialized medical affairs providers delivering customized service packages matching specific client needs. These relationships often evolve from transactional project-based engagements toward strategic partnerships where outsourcing providers embed teams within pharmaceutical organizations functioning as extensions of internal medical affairs departments. Generic pharmaceutical companies increasingly adopt outsourcing for medical information services and pharmacovigilance functions required by regulatory authorities despite not conducting extensive clinical development programs. The pharmaceutical industry's shift toward rare diseases and orphan drugs accelerates outsourcing as companies require specialized medical affairs expertise for narrow therapeutic areas where maintaining full-time internal staff proves economically inefficient.

Regional Insights
North America
Market Leadership Through Pharmaceutical Industry Concentration and Mature Outsourcing Adoption
North America dominates with approximately 38% of the global medical affairs outsourcing market share, valued at over USD 855 million in 2025 and growing at 12.1% CAGR through 2033. The region benefits from concentrated pharmaceutical and biotechnology industry presence with major companies headquartered in United States alongside extensive biotech clusters in Boston, San Francisco, and Research Triangle regions. Sophisticated regulatory environment under FDA oversight creates substantial medical affairs workload including extensive safety reporting, medical information management, and scientific communication supporting regulatory submissions and commercial activities. The mature outsourcing culture across American pharmaceutical industry normalizes external partnerships for specialized functions, reducing organizational resistance compared to regions where outsourcing faces cultural barriers.
Leading regional service providers including IQVIA, Syneos Health, Parexel International, ICON plc, and Medpace serve domestic pharmaceutical clients through extensive North American operations while also supporting global programs requiring regional medical affairs expertise. These companies maintain large teams of medical writers, medical science liaisons, and medical information specialists based throughout United States and Canada covering diverse therapeutic areas and regulatory specializations. The region's competitive advantages include access to top-tier medical talent from prestigious universities, proximity to pharmaceutical decision-makers enabling collaborative partnerships, and established infrastructure supporting complex clinical development and commercialization programs. Specialty pharmaceutical companies and emerging biotech firms particularly drive regional outsourcing growth as these organizations pursue asset-light business models focusing internal resources on core R&D capabilities while outsourcing non-core medical affairs functions.
Asia Pacific
Fastest Regional Growth Through Pharmaceutical Expansion and Cost-Competitive Talent Access
Asia Pacific emerges as the fastest-growing regional market expanding at approximately 14.5% CAGR as pharmaceutical companies increasingly conduct clinical development and establish commercial operations throughout the region's diverse markets. China and India lead regional growth driven by substantial investments in pharmaceutical manufacturing, expanding clinical trial activity, and growing domestic biotech sectors requiring comprehensive medical affairs support. The availability of highly educated medical professionals including physicians, pharmacists, and PhD scientists at competitive compensation rates attracts global pharmaceutical companies establishing regional medical affairs centers serving both local markets and international operations. Government initiatives supporting pharmaceutical innovation in countries including Singapore, South Korea, and Malaysia create favorable environments for medical affairs outsourcing hub development.
Major global outsourcing providers including IQVIA, Parexel, ICON, and regional specialists like Novotech establish significant Asian operations recruiting local medical talent understanding regional regulatory frameworks, healthcare systems, and cultural contexts. These regional centers provide cost-competitive services including medical writing, medical information, and pharmacovigilance supporting global pharmaceutical clients while also serving growing numbers of Asian pharmaceutical and biotech companies entering international markets. Japan represents a mature market with sophisticated regulatory requirements and strong preference for local language medical affairs support, while Southeast Asian countries show rapid growth as pharmaceutical companies expand clinical trials leveraging diverse patient populations and experienced clinical research infrastructure. The region's linguistic diversity creates opportunities for multilingual medical affairs services supporting regulatory submissions and medical communications across dozens of languages and dialects.
Top Key Players
-
IQVIA Holdings Inc. (United States)
-
Syneos Health Inc. (United States)
-
ICON plc (Ireland)
-
Parexel International Corporation (United States)
-
Thermo Fisher Scientific Inc. (United States)
-
Medpace Holdings Inc. (United States)
-
PPD Inc. (United States)
-
Covance Inc. (United States)
-
Charles River Laboratories (United States)
-
Pharmaceutical Product Development LLC (United States)
-
PROMETRIKA LLC (United States)
-
Amplity Health (United States)
-
Novotech (Australia)
-
Accell Clinical (United States)
Recent Developments
-
IQVIA Holdings (2024) – Expanded medical affairs service capabilities through strategic acquisition of specialized medical communications agency strengthening scientific writing, publication planning, and congress management offerings serving pharmaceutical and biotech clients globally with integrated medical affairs solutions
-
Syneos Health (2025) – Launched AI-powered medical information platform automating routine inquiry responses and enabling predictive analytics identifying emerging safety signals and medical information trends supporting proactive pharmaceutical client communications and regulatory compliance
-
ICON plc (2024) – Completed integration of PRA Health Sciences acquisition creating world's largest dedicated contract research organization with comprehensive medical affairs capabilities spanning early development through post-marketing surveillance across all therapeutic areas
-
Parexel International (2025) – Announced significant investment expanding Asian medical affairs operations establishing regional centers in Singapore, Shanghai, and Bangalore providing localized medical writing, MSL services, and regulatory support serving growing pharmaceutical activity throughout Asia Pacific markets
-
Thermo Fisher Scientific (2024) – Enhanced PPD clinical research business with advanced real-world evidence capabilities integrating electronic health record data analytics, patient registry management, and outcomes research supporting pharmaceutical medical affairs strategies and regulatory submissions
Market Trends
Digital Transformation and Strategic Partnership Models Reshaping Service Delivery
The medical affairs outsourcing market experiences fundamental transformation toward digital-first service delivery as providers invest heavily in artificial intelligence, machine learning, and advanced analytics capabilities augmenting traditional human expertise. Outsourcing firms develop proprietary technology platforms automating routine medical information responses, accelerating literature surveillance, and generating real-world evidence from diverse data sources including electronic health records, claims databases, and patient registries. These digital tools enable pharmaceutical clients to scale medical affairs operations efficiently while maintaining quality standards and regulatory compliance across global markets. Virtual medical science liaison engagement through online platforms complements traditional field-based activities, expanding reach to community healthcare providers and enabling more frequent scientific exchanges without travel costs and logistical constraints.
Strategic partnership models increasingly replace transactional project-based relationships as pharmaceutical companies seek deeper collaborations with outsourcing providers functioning as extensions of internal medical affairs departments. These partnerships involve co-location of vendor staff within pharmaceutical facilities, integrated technology platforms sharing data seamlessly, and aligned incentive structures rewarding quality and strategic value beyond simple cost reduction. Full-service outsourcing arrangements consolidate multiple medical affairs functions with single providers simplifying vendor management, improving operational coordination, and enabling more sophisticated analytics across integrated datasets. Pharmaceutical companies particularly value outsourcing partners bringing therapeutic area expertise, regulatory intelligence, and strategic insights informing medical strategies rather than simply providing staff augmentation executing predefined tasks.
Segments Covered in the Report
By Service Type
-
Medical Science Liaison Services
-
Medical Writing & Publishing
-
Medical Information Services
-
Medical Monitoring
-
Regulatory Affairs Support
-
Pharmacovigilance & Safety Services
By End-Use Industry
-
Pharmaceutical
-
Biopharmaceutical
-
Medical Devices
By Therapeutic Area
-
Oncology
-
Cardiovascular
-
Neurology
-
Immunology
-
Infectious Diseases
-
Rare Diseases
-
Others
By Region
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What drives growth in the medical affairs outsourcing market globally?
Answer: The medical affairs outsourcing market experiences robust growth from rising pharmaceutical R&D costs and increasing regulatory complexity requiring specialized expertise. Small biotech companies and large pharmaceutical firms increasingly outsource medical affairs functions to access scalable resources and reduce fixed costs while maintaining quality standards.
Question 2: Which region leads the medical affairs outsourcing market currently?
Answer: North America dominates the medical affairs outsourcing market with approximately 38% share backed by concentrated pharmaceutical industry presence and mature outsourcing adoption. The region benefits from sophisticated regulatory environment and established relationships between pharmaceutical companies and leading contract research organizations.
Question 3: How does digital transformation impact the medical affairs outsourcing market?
Answer: Digital transformation significantly influences the medical affairs outsourcing market through AI-powered platforms automating routine tasks and generating real-world evidence. These technologies enable outsourcing providers to deliver higher-quality insights faster while reducing costs for pharmaceutical clients managing extensive product portfolios.
Question 4: What services lead the medical affairs outsourcing market consumption?
Answer: Medical science liaison services lead the medical affairs outsourcing market representing approximately 32% of market share. Field-based medical teams engaging healthcare providers and gathering clinical insights remain essential for pharmaceutical companies supporting product launches and lifecycle management strategies.
Question 5: Which therapeutic area shows fastest growth in the medical affairs outsourcing market?
Answer: Oncology demonstrates the strongest growth trajectory in the medical affairs outsourcing market with projected CAGR approaching 14.2%. Complex cancer immunotherapies and targeted therapies require specialized medical affairs expertise supporting clinical development and commercialization activities across global markets.
