Liver Cancer Drug Market Overview
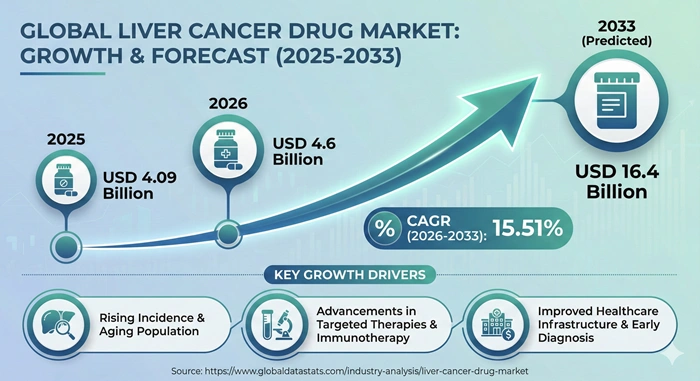
The global liver cancer drug market size is valued at USD 4.09 billion in 2025 and is predicted to increase from USD 4.6 billion in 2026 to approximately USD 16.4 billion by 2033, growing at a CAGR of 15.51% from 2026 to 2033. This remarkable expansion reflects increasing prevalence of hepatocellular carcinoma, advancements in targeted therapies and immunotherapies, and growing investments in oncology research and development.
Liver cancer, primarily hepatocellular carcinoma, represents one of the leading causes of cancer-related deaths worldwide. The rising incidence of risk factors such as hepatitis B and C infections, alcohol-related liver disease, non-alcoholic fatty liver disease, and obesity continues to drive demand for effective therapeutic interventions. The pharmaceutical industry has responded with innovative treatment options that extend survival rates and improve quality of life for patients facing this aggressive malignancy. Modern treatment approaches now combine targeted molecular therapies with immune checkpoint inhibitors to deliver superior clinical outcomes compared to traditional chemotherapy alone.
The market landscape features robust competition among multinational pharmaceutical companies developing next-generation treatments. Regulatory approvals for combination therapy regimens have reshaped standard care protocols, while precision medicine approaches tailored to genetic and molecular profiles are gaining prominence. Healthcare systems across developed and emerging economies are strengthening cancer screening infrastructure and expanding access to advanced therapeutics, further propelling market growth throughout the forecast period.
AI Impact on the Liver Cancer Drug Industry
Transforming Drug Discovery and Accelerating Clinical Development Timelines
Artificial intelligence is revolutionizing the liver cancer drug market by significantly reducing the time required for drug discovery and development. Machine learning algorithms analyze vast datasets of molecular structures, genetic profiles, and clinical trial results to identify promising therapeutic candidates with higher precision. Pharmaceutical companies are now leveraging AI-powered platforms to predict drug efficacy, optimize dosing regimens, and identify patient populations most likely to respond to specific treatments. This technology has accelerated the identification of novel biomarkers for hepatocellular carcinoma, enabling more targeted therapy development and personalized treatment strategies that improve patient outcomes while reducing development costs.
AI-driven diagnostic tools are enhancing early detection capabilities, allowing healthcare providers to identify liver cancer at stages when treatment is most effective. Advanced imaging analysis systems use deep learning to detect subtle abnormalities in medical scans that might be missed by traditional methods. Additionally, AI is optimizing clinical trial design by improving patient stratification, predicting trial outcomes, and identifying potential adverse events before they occur. The integration of artificial intelligence into drug development pipelines is expected to bring more effective liver cancer drugs to market faster while reducing the overall cost burden on healthcare systems and improving accessibility for patients globally.
Growth Factors
Rising Disease Burden and Expanding Treatment Infrastructure Drive Market Momentum
The liver cancer drug market is experiencing substantial growth due to the increasing global incidence of hepatocellular carcinoma and related liver diseases. Hepatitis B and C viral infections remain major risk factors, particularly in Asia-Pacific regions where prevalence rates are significantly higher than global averages. Lifestyle-related factors including excessive alcohol consumption, obesity, and metabolic syndrome have contributed to rising cases of non-alcoholic steatohepatitis, which often progresses to liver cancer. Healthcare systems worldwide are implementing comprehensive screening programs that facilitate earlier diagnosis, creating expanded patient populations eligible for pharmacological interventions. Government initiatives supporting cancer care infrastructure development and reimbursement policies that cover advanced therapies have improved treatment accessibility across diverse socioeconomic groups.
Technological advancements in drug development have yielded breakthrough therapies that demonstrate superior efficacy compared to conventional treatments. The success of tyrosine kinase inhibitors such as sorafenib and lenvatinib established targeted therapy as a cornerstone of liver cancer treatment, while immune checkpoint inhibitors like nivolumab and pembrolizumab have revolutionized outcomes for advanced-stage patients. Combination therapy regimens that pair immunotherapies with anti-angiogenic agents have shown remarkable improvements in overall survival and progression-free survival rates. Pharmaceutical companies continue investing heavily in research and development to expand their oncology portfolios, with numerous late-stage clinical trials evaluating novel mechanisms of action. Strategic collaborations between biotechnology firms and academic institutions are accelerating innovation, while regulatory agencies have established expedited approval pathways that bring effective treatments to patients more rapidly.

Market Outlook
Favorable Regulatory Environment and Innovation Pipeline Support Long-Term Growth
The liver cancer drug market outlook remains highly positive as regulatory agencies worldwide continue prioritizing oncology innovations through accelerated approval mechanisms. Recent approvals of combination immunotherapy regimens have expanded first-line treatment options beyond traditional tyrosine kinase inhibitors, providing clinicians with evidence-based alternatives that deliver meaningful survival benefits. The growing emphasis on precision medicine and biomarker-driven therapy selection is reshaping treatment paradigms, enabling more personalized approaches that optimize efficacy while minimizing adverse effects. Healthcare providers are increasingly adopting multidisciplinary treatment strategies that integrate pharmacological interventions with surgical resection, ablation techniques, and radiation therapy to maximize patient outcomes across different disease stages.
The market is witnessing a shift toward oral targeted therapies that offer improved convenience and adherence compared to intravenous chemotherapy regimens. Next-generation tyrosine kinase inhibitors with enhanced selectivity profiles and reduced toxicity are advancing through clinical development pipelines, while novel checkpoint inhibitors targeting alternative immune pathways show promise in early-stage trials. Biosimilar versions of established liver cancer drugs are beginning to enter markets in developing regions, improving affordability and expanding treatment access for underserved populations. Pharmaceutical companies are also exploring combination approaches that integrate targeted therapies with emerging modalities such as oncolytic viruses, therapeutic vaccines, and cell-based immunotherapies to overcome resistance mechanisms and achieve durable responses in difficult-to-treat patient populations.
Expert Speaks
-
Christopher Boerner, CEO of Bristol Myers Squibb, stated that the approval of combination immunotherapy for liver cancer marks a critical milestone in the company's commitment to improving outcomes for patients with hepatocellular carcinoma, emphasizing the importance of bringing innovative first-line treatment options to patients globally.
-
Bill Anderson, CEO of Bayer AG, highlighted that the company has fundamentally transformed its pharmaceutical operations and is experiencing robust sales performance of its oncology medications, with continued focus on expanding treatment options for difficult-to-treat cancers including liver malignancies.
-
Robert Davis, CEO of Merck & Co., noted that the company's leadership in immuno-oncology and strategic research collaborations position it to deliver breakthrough therapies that address unmet medical needs in cancer treatment, with significant focus on expanding indications for checkpoint inhibitors across various tumor types including hepatocellular carcinoma.
Key Report Takeaways
-
North America leads the liver cancer drug market with the largest market share due to advanced healthcare infrastructure, high R&D investments, strong presence of pharmaceutical companies, comprehensive reimbursement coverage, and early adoption of innovative therapies, accounting for approximately 42% market share with steady growth projected throughout the forecast period.
-
Asia-Pacific region is growing fastest with the highest CAGR of approximately 17.8% driven by large patient populations, increasing hepatitis B prevalence, improving healthcare infrastructure, rising healthcare expenditure, growing awareness of cancer screening, and expanding access to advanced targeted therapies and immunotherapies across developing economies.
-
Targeted therapy drugs dominate usage patterns among healthcare providers and patients, representing the largest segment with approximately 58% market share due to proven efficacy, favorable safety profiles compared to traditional chemotherapy, convenient oral administration routes, and robust clinical evidence supporting use in first-line and second-line treatment settings.
-
Hospital pharmacies contribute the most to distribution channels with approximately 52% market share, serving as primary access points for specialty oncology medications, providing comprehensive patient support services, ensuring proper medication management, and facilitating coordination between oncologists, pharmacists, and patients throughout treatment journeys.
-
Tyrosine kinase inhibitors represent the most popular mechanism of action, accounting for approximately 46% market share due to well-established clinical data, multiple approved drug options including sorafenib and lenvatinib, demonstrated survival benefits in advanced hepatocellular carcinoma, and growing use in combination with immunotherapy agents for enhanced efficacy.
-
Immunotherapy segment will experience rapid future growth with projected CAGR of 16.9% and increasing market share reaching approximately 35% by 2033, driven by breakthrough clinical trial results, expanding approval indications, development of novel checkpoint inhibitors and combination regimens, and paradigm shift toward immune-based treatment strategies in oncology.
Market Scope
| Report Coverage Details | |
|---|---|
| Market Size by 2033 | USD 16.4 Billion |
| Market Size by 2025 | USD 4.09 Billion |
| Market Size by 2026 | USD 4.6 Billion |
| Market Growth Rate from 2026 to 2033 | CAGR of 15.51% |
| Dominating Region | North America |
| Fastest Growing Region | Asia-Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2033 |
| Segments Covered | Drug Class, Mechanism of Action, Stage of Cancer, End User, Distribution Channel, Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, Middle East and Africa |
Market Dynamics
Drivers Impact Analysis
Increasing Hepatitis Prevalence and Viral Infection Rates Fuel Treatment Demand
The liver cancer drug market experiences significant growth momentum from escalating hepatitis B and C infection rates globally, particularly across Asia-Pacific and African regions where these viral diseases remain endemic. Chronic hepatitis infections represent the primary risk factor for hepatocellular carcinoma development, creating sustained demand for therapeutic interventions as infected populations age and progress toward advanced liver disease. Public health initiatives focused on hepatitis screening and linkage to care have expanded the diagnosed patient population eligible for treatment, while improved survival rates among hepatitis patients receiving antiviral therapies have paradoxically increased the pool of individuals at risk for subsequent liver cancer development.
The growing recognition of non-alcoholic fatty liver disease and metabolic-associated fatty liver disease as emerging risk factors has broadened the at-risk population beyond traditional hepatitis-infected cohorts. Rising obesity rates, diabetes prevalence, and metabolic syndrome incidence across both developed and developing nations contribute to increasing liver cancer cases that require pharmacological management. Healthcare systems are responding by implementing comprehensive surveillance programs for high-risk populations, facilitating earlier detection when treatment options demonstrate greatest efficacy. Pharmaceutical companies have capitalized on this expanding patient base by developing targeted therapies specifically designed for hepatocellular carcinoma, with clinical trial programs demonstrating meaningful survival benefits that support regulatory approvals and formulary inclusion decisions across multiple jurisdictions worldwide.
| Factor | (≈) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Increasing hepatitis prevalence | 4.2% | Global, particularly Asia-Pacific and Africa | Immediate and sustained through 2033 |
| Rising metabolic disease burden | 3.1% | North America, Europe, Asia-Pacific | Progressive increase through forecast period |

Restraints Impact Analysis
High Treatment Costs and Limited Reimbursement Access Constrain Market Expansion
The liver cancer drug market faces substantial headwinds from the extremely high cost of innovative targeted therapies and immunotherapies, which often exceed USD 150,000 annually per patient for combination regimens. These prohibitive price points create significant barriers to treatment access, particularly in developing economies where healthcare budgets remain constrained and out-of-pocket expenditures represent insurmountable burdens for most patients. Even in developed markets with established insurance systems, prior authorization requirements, step therapy protocols, and coverage restrictions limit the proportion of eligible patients who can access newest therapeutic options. Reimbursement agencies increasingly demand robust health economic evidence demonstrating cost-effectiveness compared to existing standards of care, creating uncertainty around market uptake for newly approved agents.
The complexity of treatment regimens and requirement for specialized administration infrastructure pose additional constraints on market growth. Many liver cancer drugs necessitate careful patient selection based on liver function parameters, performance status, and specific biomarker profiles, limiting the addressable patient population. Adverse event profiles associated with immunotherapies and targeted agents require intensive monitoring and management by experienced oncology teams, restricting availability to specialized cancer centers rather than community practice settings where most patients receive care. Additionally, late-stage diagnosis remains problematic in many regions, with significant proportions of patients presenting with advanced disease when treatment options demonstrate limited efficacy and prognosis remains poor regardless of therapeutic intervention.
| Factor | (≈) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High treatment costs | -2.8% | Global, particularly developing markets | Sustained constraint throughout forecast period |
| Limited specialized infrastructure | -1.6% | Latin America, Middle East, Africa, rural areas | Gradual improvement as infrastructure develops |
Opportunities Impact Analysis
Emerging Combination Therapies and Biosimilar Development Create Growth Avenues
The liver cancer drug market presents substantial opportunities through development and commercialization of novel combination therapy regimens that leverage complementary mechanisms of action to overcome resistance and improve outcomes. Clinical research demonstrates that pairing immune checkpoint inhibitors with anti-angiogenic agents or tyrosine kinase inhibitors produces synergistic effects that significantly enhance overall survival compared to monotherapy approaches. Pharmaceutical companies are actively pursuing strategic combinations of their proprietary assets with complementary therapies from other manufacturers, expanding treatment options and creating new revenue streams. Regulatory agencies have shown willingness to approve combination regimens based on compelling efficacy data, accelerating time to market for innovative treatment paradigms.
The anticipated entry of biosimilar versions of established liver cancer drugs represents another significant opportunity to expand market access and penetration. As patents expire on pioneering targeted therapies and monoclonal antibodies, biosimilar manufacturers can offer clinically equivalent alternatives at substantially reduced prices, improving affordability particularly in price-sensitive markets. This competitive dynamic will likely expand the treated patient population while maintaining overall market value through volume growth. Additionally, advances in precision medicine and companion diagnostics create opportunities to identify patient subpopulations most likely to benefit from specific therapies, optimizing treatment selection and improving cost-effectiveness profiles that support broader reimbursement coverage and clinical adoption across diverse healthcare settings globally.
| Factor | (≈) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Novel combination therapies | 5.7% | Global, led by North America and Europe | Accelerating impact through 2028-2033 |
| Biosimilar market entry | 2.3% | Europe, Asia-Pacific, Latin America | Increasing impact from 2027 onwards |

Segment Analysis
Targeted Therapy Drugs
Established Efficacy Profile and Convenient Administration Drive Segment Leadership
The targeted therapy drugs segment dominates the liver cancer drug market with approximately 58% market share and projected CAGR of 14.8% throughout the forecast period, driven by robust clinical evidence, favorable safety profiles, and convenient oral administration routes that enhance patient compliance. Tyrosine kinase inhibitors including sorafenib, lenvatinib, regorafenib, and cabozantinib have demonstrated meaningful survival benefits in multiple phase 3 clinical trials, establishing these agents as standard treatment options across first-line and subsequent-line settings for advanced hepatocellular carcinoma. The segment benefits from strong physician familiarity with targeted therapy mechanisms, well-defined patient selection criteria based on liver function and performance status, and extensive real-world evidence supporting efficacy across diverse patient populations and geographic regions.
North America and Europe represent the largest markets for targeted therapy drugs, accounting for approximately 65% of segment revenue, with major pharmaceutical companies including Bayer AG, Eisai Co. Ltd., and Exelixis Inc. maintaining dominant positions through their marketed products and robust clinical development pipelines. Asia-Pacific markets are experiencing rapid growth driven by increasing diagnosis rates, improving reimbursement access, and introduction of locally manufactured generic versions that enhance affordability. The segment continues evolving through development of next-generation tyrosine kinase inhibitors with enhanced selectivity profiles that minimize off-target toxicities while maintaining or improving efficacy. Pharmaceutical companies are also exploring combination approaches that integrate targeted therapies with immunotherapy agents to achieve synergistic effects, overcome resistance mechanisms, and extend progression-free survival and overall survival compared to monotherapy approaches in treatment-naive and previously treated patient populations.
Immunotherapy Drugs
Breakthrough Clinical Results and Paradigm-Shifting Combinations Accelerate Growth Trajectory
The immunotherapy drugs segment represents the fastest-growing category within the liver cancer drug market with projected CAGR of 16.9% and expanding market share reaching approximately 35% by 2033, fueled by revolutionary clinical trial outcomes demonstrating superior survival benefits compared to traditional targeted therapy monotherapy. Immune checkpoint inhibitors targeting PD-1, PD-L1, and CTLA-4 pathways have transformed treatment approaches for hepatocellular carcinoma, with combination regimens such as atezolizumab plus bevacizumab and nivolumab plus ipilimumab receiving regulatory approvals as first-line treatment options. These immunotherapy combinations have shown remarkable improvements in overall survival, objective response rates, and complete response rates, establishing new benchmarks for efficacy in this difficult-to-treat malignancy.
The immunotherapy segment experiences strongest growth in North America and Europe where regulatory agencies have approved multiple checkpoint inhibitor combinations and monotherapies for various treatment lines, with Bristol Myers Squibb, Merck & Co. Inc., Roche, and AstraZeneca leading market development through their established immuno-oncology portfolios. Asia-Pacific markets are rapidly adopting immunotherapy approaches as local regulatory authorities approve international trial data and healthcare systems expand reimbursement coverage for these premium-priced agents. Regional pharmaceutical companies in China and South Korea are developing domestic checkpoint inhibitors that offer more affordable alternatives while maintaining comparable efficacy profiles, democratizing access to cutting-edge immunotherapy treatments. The segment benefits from ongoing clinical research evaluating immunotherapy combinations with targeted therapies, conventional chemotherapy, and locoregional treatments such as ablation and transarterial chemoembolization, creating multiple pathways for market expansion as positive trial results translate into regulatory approvals and guideline recommendations throughout the forecast period.

Regional Insights
North America
Advanced Infrastructure and Innovation Leadership Sustain Market Dominance
North America maintains its position as the dominant region in the liver cancer drug market with approximately 42% market share and steady CAGR of 14.2% throughout the forecast period, supported by world-class healthcare infrastructure, substantial research and development investments, comprehensive insurance coverage, and early adoption of innovative therapeutic approaches. The United States represents the primary contributor to regional revenue, driven by high drug pricing that supports premium reimbursement for novel targeted therapies and immunotherapy combinations. The region benefits from concentration of leading pharmaceutical companies including Bristol Myers Squibb, Merck & Co. Inc., Pfizer Inc., and Eli Lilly and Company that maintain extensive clinical trial networks, regulatory expertise, and commercial capabilities enabling rapid market penetration following regulatory approvals.
The North American market experiences favorable regulatory dynamics through FDA programs such as breakthrough therapy designation, accelerated approval pathways, and priority review mechanisms that reduce development timelines for promising liver cancer drugs demonstrating substantial improvements over existing standards of care. Academic medical centers and comprehensive cancer centers across the region serve as innovation hubs where cutting-edge combination therapy regimens are developed, tested, and refined before broader dissemination to community oncology practices. The region also benefits from sophisticated companion diagnostic infrastructure that enables biomarker-driven patient selection, optimizing treatment outcomes and supporting health economic value propositions that justify premium pricing and broad insurance coverage for advanced liver cancer therapeutics throughout both commercial and government-funded payer systems.
Asia-Pacific
Large Patient Populations and Improving Access Drive Fastest Regional Growth
Asia-Pacific emerges as the fastest-growing region in the liver cancer drug market with remarkable CAGR of 17.8% driven by massive patient populations, high hepatitis B prevalence, rapidly improving healthcare infrastructure, rising disposable incomes, and expanding government initiatives supporting cancer care access and affordability. China, Japan, and South Korea represent the largest markets within the region, collectively accounting for approximately 75% of Asia-Pacific revenue, with major international pharmaceutical companies including Bayer AG, Eisai Co. Ltd., Bristol Myers Squibb, and Roche establishing strong commercial presences through local subsidiaries and strategic partnerships with regional distributors.
The Asia-Pacific liver cancer drug market benefits from growing awareness of screening importance, implementation of national cancer control programs, and increasing availability of advanced diagnostic technologies that facilitate earlier detection when treatment options demonstrate greatest efficacy. Regional pharmaceutical companies such as BeiGene Ltd., Innovent Biologics, Hengrui Medicine Co. Ltd., and Shanghai Pharmaceuticals are developing domestic checkpoint inhibitors and targeted therapies that offer comparable efficacy to international products at substantially reduced prices, improving affordability and accelerating market penetration across price-sensitive populations. Government initiatives including volume-based procurement programs, national reimbursement list expansions, and regulatory reforms streamlining approval processes for innovative oncology drugs are creating favorable market dynamics that support sustained high growth rates throughout the forecast period across both developed markets like Japan and Australia as well as rapidly emerging economies including India, Indonesia, and Southeast Asian nations.
Top Key Players
-
Bayer AG (Germany)
-
Bristol Myers Squibb (United States)
-
Merck & Co. Inc. (United States)
-
Pfizer Inc. (United States)
-
Exelixis Inc. (United States)
-
Eisai Co. Ltd. (Japan)
-
Eli Lilly and Company (United States)
-
F. Hoffmann-La Roche Ltd. (Switzerland)
-
Novartis AG (Switzerland)
-
AstraZeneca (United Kingdom)
-
Ipsen (France)
-
Gilead Sciences (United States)
-
BeiGene Ltd. (China)
-
Innovent Biologics (China)
-
Sanofi (France)
Recent Developments
-
Bristol Myers Squibb received European Commission approval in 2025 for Opdivo (nivolumab) plus Yervoy (ipilimumab) combination therapy as first-line treatment for adult patients with unresectable or advanced hepatocellular carcinoma, based on positive results from the CheckMate-9DW Phase 3 study demonstrating significant improvements in overall survival and objective response rates compared to lenvatinib monotherapy.
-
Junshi Biosciences obtained NMPA approval in March 2025 for TUOYI (toripalimab) in combination with bevacizumab for first-line treatment of unresectable or metastatic hepatocellular carcinoma in China, marking a significant advancement for liver cancer treatment options available to Chinese patients in early-stage disease management.
-
Akeso Inc. initiated Phase 2 registrational trial in September 2025 evaluating cadonilimab, a first-in-class PD-1/CTLA-4 bispecific antibody, in combination with lenvatinib for treating advanced hepatocellular carcinoma in patients previously treated with atezolizumab and bevacizumab, addressing unmet needs in second-line treatment settings.
-
AstraZeneca completed acquisition in December 2025 of an immuno-oncology developer in Europe to strengthen its combination therapy portfolio for hepatocellular carcinoma and other solid tumors, expanding research capabilities in novel checkpoint inhibitor mechanisms and biomarker-driven patient selection strategies.
-
Roche Holding AG acquired clinical-stage oncology company in January 2026 in the United States focused on targeted therapy platforms to expand its liver cancer drug pipeline, demonstrating continued industry consolidation and strategic investments in innovative treatment modalities for difficult-to-treat malignancies.
Market Trends
Precision Medicine and Combination Therapy Approaches Reshape Treatment Paradigms
The liver cancer drug market is experiencing transformative shifts toward precision medicine strategies that leverage biomarker testing and molecular profiling to guide treatment selection and optimize patient outcomes. Pharmaceutical companies are investing substantially in companion diagnostic development to identify patient subpopulations most likely to benefit from specific targeted therapies or immunotherapy regimens, enabling more personalized treatment approaches that improve efficacy while reducing unnecessary exposure to ineffective or toxic therapies. Advanced genomic sequencing technologies are revealing actionable mutations and pathway alterations that can be targeted with existing or novel therapeutic agents, expanding the addressable patient population for precision oncology approaches. Healthcare systems are implementing tumor board processes that integrate molecular data with clinical characteristics to inform multidisciplinary treatment planning, representing a fundamental departure from traditional one-size-fits-all approaches to liver cancer management.
Combination therapy regimens pairing immunotherapies with targeted agents or anti-angiogenic drugs have emerged as the new standard of care in first-line treatment settings, demonstrating superior survival outcomes compared to monotherapy approaches that dominated the market in previous years. Clinical research continues exploring novel combination strategies that integrate checkpoint inhibitors with different classes of targeted therapies, oncolytic viruses, therapeutic vaccines, and locoregional treatments to achieve synergistic effects and overcome resistance mechanisms that limit durability of single-agent responses. Pharmaceutical companies are pursuing strategic collaborations to evaluate complementary mechanisms of action, with regulatory agencies demonstrating willingness to approve combinations based on compelling efficacy data even when components come from different manufacturers. This trend toward rational combinations based on mechanistic understanding rather than empiric testing represents a maturation of the oncology drug development paradigm that promises to deliver continued improvements in patient outcomes throughout the forecast period.
Segments Covered in the Report
By Drug Class
-
Targeted Therapy Drugs
-
Tyrosine Kinase Inhibitors
-
Multi-Kinase Inhibitors
-
Monoclonal Antibodies
-
-
Immunotherapy Drugs
-
Checkpoint Inhibitors
-
Combination Immunotherapies
-
-
Chemotherapy Drugs
-
Systemic Chemotherapy
-
Regional Chemotherapy
-
By Mechanism of Action
-
Tyrosine Kinase Inhibitors
-
Checkpoint Inhibitors (PD-1/PD-L1, CTLA-4)
-
Monoclonal Antibodies
-
Multi-Kinase Inhibitors
-
VEGF Inhibitors
By Stage of Cancer
-
Early-Stage Liver Cancer
-
Advanced-Stage Liver Cancer
-
Metastatic Liver Cancer
By End User
-
Hospitals
-
Cancer Centers
-
Specialty Clinics
-
Ambulatory Surgical Centers
By Distribution Channel
-
Hospital Pharmacies
-
Retail Pharmacies
-
Online Pharmacies
-
Specialty Pharmacies
By Region
-
North America
-
United States
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
United Kingdom
-
France
-
Italy
-
Spain
-
Rest of Europe
-
-
Asia-Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
Rest of Asia-Pacific
-
-
Latin America
-
Brazil
-
Argentina
-
Rest of Latin America
-
-
Middle East and Africa
-
GCC Countries
-
South Africa
-
Rest of Middle East and Africa
-
Frequently Asked Questions
Question 1: What is the expected liver cancer drug market size by 2033?
Answer: The global liver cancer drug market is projected to reach approximately USD 16.4 billion by 2033. This growth is driven by increasing disease prevalence and advancing treatment options.
Question 2: Which region dominates the liver cancer drug market currently?
Answer: North America dominates the liver cancer drug market with approximately 42% market share. The region benefits from advanced healthcare infrastructure and early adoption of innovative therapies.
Question 3: What are the key drivers of liver cancer drug market growth?
Answer: Primary drivers include rising hepatitis prevalence, increasing metabolic disease burden, breakthrough immunotherapy approvals, and expanding combination therapy adoption. Growing investment in oncology research also supports market expansion.
Question 4: Which segment is growing fastest in the liver cancer drug market?
Answer: The immunotherapy drugs segment is experiencing the fastest growth with projected CAGR of 16.9%. This acceleration reflects revolutionary clinical outcomes from checkpoint inhibitor combinations.
Question 5: What is the liver cancer drug market CAGR from 2026 to 2033?
Answer: The liver cancer drug market is expected to grow at a CAGR of 15.51% from 2026 to 2033. This robust growth reflects strong demand for advanced targeted and immunotherapy treatments.
