Infectious Disease Therapeutics Market Overview
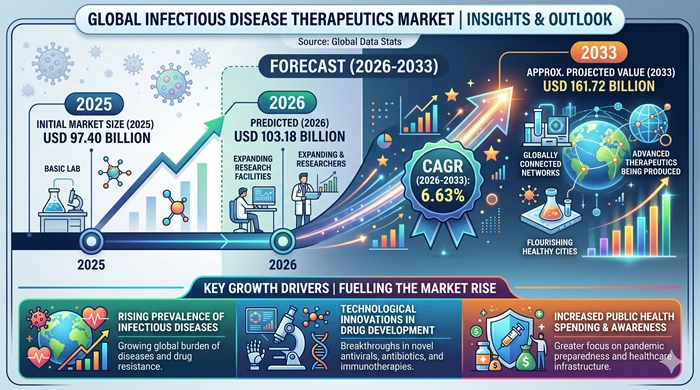
The global infectious disease therapeutics market size is valued at USD 97.40 billion in 2025 and is predicted to increase from USD 103.18 billion in 2026 to approximately USD 161.72 billion by 2033, growing at a CAGR of 6.63% from 2026 to 2033. Rising global burden of bacterial, viral, fungal, and parasitic infections, escalating antimicrobial resistance (AMR) concerns, growing investment in antiviral and antibiotic drug pipelines, and the post-pandemic prioritization of infectious disease preparedness are the core forces driving consistent and robust market expansion worldwide.
AI Impact on the Infectious Disease Therapeutics Industry
Artificial Intelligence Is Accelerating Drug Discovery, Resistance Surveillance, and Personalized Antimicrobial Therapy Across the Infectious Disease Therapeutics Landscape*
AI is fundamentally transforming how infectious disease therapeutics are discovered, developed, and deployed in clinical settings. Machine learning models trained on vast genomic, proteomic, and pharmacological datasets are enabling pharmaceutical researchers to identify novel drug targets in resistant bacterial and viral pathogens with far greater speed and precision than conventional high-throughput screening methods. AI platforms developed by companies like Pfizer, AstraZeneca, and Gilead Sciences are being used to predict drug-pathogen binding affinities, identify resistance mutation patterns, and design new molecular entities optimized for efficacy against drug-resistant strains of critical pathogens including Mycobacterium tuberculosis, MRSA, and HIV. This AI-driven drug discovery capability is significantly reducing the timeline and cost of bringing new infectious disease therapeutics from research to clinical candidate stage.
On the clinical and public health side, AI-powered surveillance platforms are reshaping how antimicrobial resistance is monitored and managed at hospital, national, and global scales. Real-time pathogen genomics analysis tools, AI-assisted clinical decision support systems for empiric antibiotic selection, and predictive outbreak models are helping clinicians and public health authorities respond to emerging infectious disease threats more rapidly and precisely. National health agencies and the WHO are increasingly deploying AI-based AMR monitoring dashboards that integrate clinical laboratory data, hospital prescribing patterns, and genomic surveillance findings to identify resistance emergence trends before they become crises. As AI integration within the infectious disease therapeutics ecosystem deepens, the pace of both drug development and clinical antimicrobial stewardship is expected to accelerate substantially.
Growth Factors
Escalating Antimicrobial Resistance, Growing Viral Infection Burden, Increasing Healthcare Infrastructure Investment, and a Robust Infectious Disease Drug Pipeline Are Powering Market Growth*
The infectious disease therapeutics market is driven at its core by the worsening global burden of antimicrobial resistance, which the WHO has declared one of the most serious global health threats of the 21st century. Drug-resistant tuberculosis, carbapenem-resistant Enterobacteriaceae, multidrug-resistant gonorrhea, and pan-resistant Acinetobacter baumannii are causing treatment failures, prolonged hospitalizations, and preventable deaths at escalating rates worldwide, creating an urgent and growing need for next-generation antibiotic and antifungal therapeutics. Government and supranational bodies including the U.S. BARDA, CARB-X, and the AMR Action Fund are channeling significant funding into antibiotic R&D programs specifically to address resistance-driven therapeutic gaps, directly stimulating investment in the infectious disease drug pipeline.
The post-COVID-19 pandemic landscape has permanently elevated the global profile and urgency of infectious disease preparedness, driving sustained increases in antiviral drug development, pandemic readiness stockpiling, and infectious disease diagnostics investment. mRNA vaccine technology — originally developed for COVID-19 — is being actively repurposed for other infectious disease targets including HIV, RSV, influenza, and tuberculosis, representing a technology platform expansion that could dramatically broaden the infectious disease therapeutics addressable market. Additionally, growing rates of HIV, hepatitis B and C, and opportunistic infections in immunocompromised populations — including transplant recipients, oncology patients, and individuals with HIV/AIDS — are creating consistent and expanding demand for antiviral and antifungal therapies that serve high-need patient populations with limited treatment alternatives.

Market Outlook
The Long-Term Outlook Is Strongly Positive, Anchored by a Rich Drug Pipeline, AMR Policy Momentum, and Growing Emerging Market Healthcare Access*
The long-term outlook for the infectious disease therapeutics market is exceptionally favorable, supported by one of the richest drug pipelines in the biopharmaceutical industry. Hundreds of novel antibiotic, antiviral, antifungal, and antiparasitic compounds are currently in clinical development globally, addressing both common high-volume infections and rare but high-mortality pathogen threats. Major regulatory milestones — including the U.S. PASTEUR Act framework, the EU's AMR Action Plan incentives, and Japan's new antibiotic pull-incentive measures — are improving the commercial viability of novel antibiotic development, which had suffered from inadequate financial returns relative to development costs. As these policy incentives take effect, more pharmaceutical companies are expected to re-enter the antibiotic development space, enriching the pipeline and creating commercial launch opportunities that will drive revenue growth through 2033.
Emerging markets across Asia Pacific, Latin America, and Sub-Saharan Africa represent the most dynamic long-term growth opportunity within the infectious disease therapeutics market. These regions carry the highest burden of communicable diseases globally — including tuberculosis, malaria, dengue fever, neglected tropical diseases, and HIV — and are simultaneously undergoing rapid healthcare infrastructure expansion that is improving diagnosis rates, treatment access, and therapeutic prescription volumes. As generic pharmaceutical manufacturing in India, China, and Brazil continues to scale and as multinational pharmaceutical companies increase their commercial presence in these markets through partnerships and local manufacturing, the volume of infectious disease therapeutics consumed in emerging markets is set to grow significantly through 2033.
Expert Speaks
-
"Infectious diseases remain one of the most pressing global health challenges, and Gilead's portfolio of antivirals — from HIV to hepatitis to respiratory viruses — reflects our deep commitment to addressing unmet needs in this space. The continued growth of long-acting regimens and the expanding HIV prevention market are particularly exciting areas of development for us as we look toward the next decade." — Daniel O'Day, CEO, Gilead Sciences
-
"At Pfizer, our infectious disease franchise has never been more important or more expansive. From vaccines and antivirals to novel antibiotics and antifungals, we are committed to delivering breakthrough therapies that address the world's most urgent infectious disease threats — and the pipeline we have in development today gives me great confidence in our long-term growth trajectory." — Albert Bourla, CEO, Pfizer Inc.
-
"AstraZeneca's work in respiratory infections, rare infectious diseases, and antibody-based antivirals reflects our belief that infectious disease is a scientific area where innovation can still profoundly improve patient outcomes. The integration of AI and genomics into our infectious disease drug discovery engine is accelerating our ability to identify and develop the next generation of anti-infective medicines." — Pascal Soriot, CEO, AstraZeneca
Key Report Takeaways
-
North America leads the global infectious disease therapeutics market, holding approximately 42–44% of total revenues in 2026, driven by the world's highest healthcare expenditure per capita, the largest concentration of infectious disease drug developers, robust FDA approval pathways for novel anti-infectives, and comprehensive insurance reimbursement systems that support high-volume therapeutics consumption across both hospital and community care settings.
-
Asia Pacific is the fastest-growing regional market, expanding at a CAGR of approximately 8.7% through 2033, driven by the highest global burden of tuberculosis, dengue, malaria, and hepatitis in India, China, and Southeast Asia, combined with rapidly improving healthcare infrastructure, rising generic and branded therapeutics access, and growing government infectious disease control program investment.
-
Hospitals and clinical care settings are the dominant end-user segment, accounting for the largest share of infectious disease therapeutics consumption as inpatient treatment of serious bacterial, viral, and fungal infections — particularly in ICU, immunocompromised, and post-surgical patient populations — drives the highest per-patient therapeutic expenditure volumes.
-
The antiviral therapeutics segment contributes the most to total market revenues, representing approximately 43–45% of total revenues in 2025, driven by the enormous commercial scale of HIV antiretroviral therapy (ART) programs, hepatitis C curative regimens, and respiratory antiviral markets including influenza and COVID-19 treatments.
-
Oral antibiotics are the most widely consumed therapeutic format, commanding the largest prescription volume globally due to their applicability across the broadest range of bacterial infections, lower healthcare system resource requirements compared to IV antibiotics, and strong generic availability that makes them accessible in both high and low-income markets.
-
The antifungal therapeutics segment is set to grow the fastest, projected to expand at a CAGR of approximately 9.2% through 2033, driven by rapidly rising rates of drug-resistant Candida auris infections, invasive aspergillosis in immunocompromised populations, and a growing clinical pipeline of novel antifungal drugs targeting resistant fungal pathogens with mechanisms of action distinct from existing azole, echinocandin, and polyene classes.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2025 | USD 97.40 Billion |
| Market Size by 2026 | USD 103.18 Billion |
| Market Size by 2033 | USD 161.72 Billion |
| Market Growth Rate (2026–2033) | CAGR of 6.63% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2033 |
| Segments Covered | Drug Class, Indication, Mode of Administration, Distribution Channel, End User, Region |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Surging AMR Burden, Growing Viral Infection Prevalence, Expanding Biopharmaceutical R&D Investment, and Post-Pandemic Preparedness Spending Are the Core Growth Drivers*
| Driver | ≈% Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Escalating antimicrobial resistance burden | ~32% | Global, highest in Asia Pacific & Europe | Near to long-term |
| Rising prevalence of HIV, hepatitis, and respiratory viral infections | ~26% | Global | Near to long-term |
| Growing pharmaceutical R&D investment in anti-infective pipelines | ~20% | North America, Europe | Near to medium-term |
| Post-pandemic infectious disease preparedness investment | ~14% | Global | Near to medium-term |
| Expanding healthcare access and diagnostics in emerging markets | ~8% | Asia Pacific, Latin America, Africa | Medium to long-term |
The infectious disease therapeutics market is driven first and foremost by the sheer scale and growing complexity of the global infectious disease burden. Despite remarkable progress in antimicrobial drug development over the past century, infectious diseases remain the second-leading cause of mortality worldwide and the leading cause of death in low- and middle-income countries. The emergence and spread of drug-resistant pathogens — now contributing to an estimated 1.27 million deaths annually attributed directly to AMR — is creating urgent and growing demand for novel therapeutics that can overcome resistance mechanisms and restore clinical efficacy in patients failing existing treatment regimens. This clinical urgency is being matched by unprecedented policy and funding responses from governments, multilateral organizations, and the private sector.
The enormous commercial success of the antiviral sector — driven by HIV ART programs, hepatitis C cures, and COVID-19 antivirals — has demonstrated the financial viability of infectious disease drug development at scale and is encouraging continued investment in antiviral research targeting emerging viral threats including dengue, Nipah, mpox, and seasonal influenza. The growing clinical and commercial success of long-acting injectable ART formulations — particularly ViiV Healthcare's Cabenuva — is reshaping the HIV treatment market and setting a new standard for treatment convenience that is expected to drive high uptake rates and strong revenue generation over the forecast period. These commercial success stories are providing the financial fuel and strategic motivation for continued pharma investment in the broader infectious disease therapeutics market.

Restraints Impact Analysis
High Drug Development Costs, Inadequate Commercial Returns for Novel Antibiotics, Generic Competition Pressure, and AMR-Driven Treatment Complexity Are Key Market Restraints*
| Restraint | ≈% Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Inadequate commercial returns for novel antibiotic development | ~34% | Global, primarily North America & Europe | Near to medium-term |
| Strong generic competition eroding branded drug revenues | ~27% | Global | Near-term |
| High drug development costs and clinical trial complexity for resistant pathogens | ~23% | Global | Near to medium-term |
| Access and affordability barriers in low-income markets | ~16% | Africa, South Asia, Latin America | Near to long-term |
One of the most structurally significant challenges facing the infectious disease therapeutics market — particularly in the antibiotic segment — is the inadequate financial return on investment that has historically discouraged pharmaceutical companies from committing to novel antibiotic development. Unlike chronic disease medications that are taken daily for years or decades, antibiotics are typically used for short treatment courses of days to weeks, and clinical stewardship programs intentionally restrict the use of novel antibiotics to preserve their efficacy — further limiting commercial prescribing volumes. Several small biotech companies that successfully developed and launched novel antibiotics in the 2010s subsequently filed for bankruptcy due to insufficient commercial revenues, creating a chilling effect on new market entrants that government pull-incentive programs are now working to counteract.
The rapid erosion of branded drug revenues by generic competition — particularly in mature antibiotic and antiviral classes — creates ongoing revenue pressure for major infectious disease therapeutic manufacturers. As HIV antiretroviral regimens, hepatitis C direct-acting antivirals (DAAs), and broad-spectrum antibiotics lose patent protection and face generic entry, innovator companies must continuously innovate and launch new branded products to offset revenue declines. This innovation treadmill creates sustained R&D cost pressure and makes financial forecasting for infectious disease therapeutics portfolios particularly complex, especially for smaller biotech companies that lack the diversified revenue base of large pharmaceutical multinationals.
Opportunities Impact Analysis
Novel Antibiotic Classes, Long-Acting Antiviral Formulations, mRNA Infectious Disease Vaccines, and Emerging Market Expansion Represent the Most Compelling Growth Opportunities*
| Opportunity | ≈% Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Development of novel antibiotic classes targeting AMR pathogens | ~31% | North America, Europe | Near to medium-term |
| Long-acting injectable and implantable antiviral formulations | ~27% | North America, Europe, Asia Pacific | Near to medium-term |
| mRNA-based infectious disease vaccine and therapeutic development | ~24% | North America, Europe | Medium to long-term |
| Expanding access programs in high-burden emerging markets | ~18% | Asia Pacific, Africa, Latin America | Medium to long-term |
The development of novel antibiotic classes with mechanisms of action distinct from existing drug families represents one of the most critical and commercially promising opportunities in the infectious disease therapeutics market. Several promising compounds — including teixobactin analogs, new beta-lactamase inhibitor combinations, and antibiotics targeting novel bacterial targets such as LpxC and MurA — are advancing through clinical development, with the potential to restore clinical activity against carbapenem-resistant and pan-resistant Gram-negative pathogens that currently have very limited treatment options. The market exclusivity periods and protected commercial positions these novel antibiotics will hold — particularly if paired with government pull-incentive mechanisms — offer attractive long-term revenue potential for developing companies.
The rapid expansion of long-acting injectable (LAI) and extended-release formulations in the infectious disease therapeutics market — pioneered by the HIV ART sector and now being explored for hepatitis B, tuberculosis, and malaria — represents a transformative commercial opportunity that improves patient adherence, reduces treatment burden, and justifies premium pricing. Long-acting formulations that convert previously daily oral regimens into monthly or bimonthly injectable treatments dramatically improve patient quality of life and medication adherence rates, particularly in resource-limited settings where consistent daily oral dosing is challenging to maintain. As more infectious disease therapeutic classes adopt long-acting formulation strategies, the addressable premium market segment within the broader infectious disease therapeutics space is expected to expand meaningfully.

Segment Analysis
By Drug Class
Antiviral Therapeutics Lead Market Revenue While Antifungal Drugs Emerge as the Fastest-Growing Drug Class Amid Rising Resistant Fungal Infection Rates*
The antiviral therapeutics segment holds the largest revenue share in the infectious disease therapeutics market, accounting for approximately 43–45% of total revenues in 2025 at a CAGR of approximately 6.8% through 2033. The HIV antiretroviral therapy market alone — encompassing single-tablet regimens, integrase inhibitor-based combinations, and the rapidly growing long-acting injectable segment — generates tens of billions of dollars annually and is expanding as testing programs improve diagnosis rates in underserved populations globally. North America is the dominant geography for antiviral revenues, with companies including Gilead Sciences (Biktarvy, Sunlenca), ViiV Healthcare (Cabenuva, Cabotegravir), Merck (Lagevrio), and AbbVie (Mavyret) generating the majority of global antiviral prescription revenues across HIV, hepatitis C, and respiratory virus indications. The commercial success of COVID-19 antivirals — particularly Paxlovid (Pfizer) — further reinforced the antiviral segment's dominant position and demonstrated the enormous potential revenue scale achievable from antiviral drugs addressing widespread viral infections.
The antifungal therapeutics segment is the fastest-growing drug class within the infectious disease therapeutics market, projected to expand at a CAGR of approximately 9.2% through 2033, driven by the dramatic rise in drug-resistant fungal infections — particularly Candida auris, azole-resistant Aspergillus fumigatus, and Cryptococcus neoformans — in immunocompromised patient populations globally. Asia Pacific is both a dominant fungal infection burden region and the fastest-growing geographic market for antifungal therapeutics adoption, where rising rates of uncontrolled diabetes, expanding immunosuppressed patient populations, and improving hospital diagnostics are increasing the clinical recognition and treatment of serious fungal infections. Companies including Pfizer, Merck & Co. (Cancidas), Astellas Pharma (Mycamine), and innovative biotechs including Scynexis (Brexafemme) and Amplyx Pharmaceuticals are at the forefront of the antifungal drug development and commercialization landscape.
By Mode of Administration
Oral Therapeutics Lead Volume-Based Revenue While Intravenous and Injectable Formats Generate the Highest Per-Course Value in the Infectious Disease Therapeutics Market*
The oral drug administration segment is the largest revenue contributor by mode of administration in the infectious disease therapeutics market, accounting for approximately 55–57% of total revenues in 2025 at a CAGR of approximately 6.4% through 2033. Oral antibiotics, oral antivirals — including HIV ART single-tablet regimens and hepatitis C DAAs — and oral antifungal agents are prescribed at enormous global volume across both inpatient and outpatient settings, benefiting from patient preference for self-administered therapy, lower healthcare system cost compared to IV administration, and broad generic availability that ensures affordability across income levels. North America and Europe lead oral infectious disease therapeutic revenues, where outpatient prescribing of broad-spectrum oral antibiotics, antiviral prophylaxis programs, and HIV treatment regimens generate high sustained volume. Leading companies including Gilead Sciences, AbbVie, Pfizer, and GSK (GlaxoSmithKline) derive major revenue streams from oral anti-infective formulations that are prescribed and refilled at scale across global pharmacy and hospital dispensing networks.
The injectable and intravenous administration segment is the second-largest mode of administration in the infectious disease therapeutics market, generating the highest per-course revenue values as IV antibiotics, IV antifungals, and parenteral antivirals are used in the most clinically severe and high-acuity infection settings — including ICU, hematology, oncology, and transplant wards where oral dosing is not feasible or pharmacokinetically sufficient. Asia Pacific is registering the fastest growth in injectable infectious disease therapeutics adoption, driven by rapidly expanding tertiary hospital capacity, growing ICU infrastructure, and improving capabilities for managing complex opportunistic infections in immunocompromised inpatients at major hospital centers in China, India, and Southeast Asia.

Regional Insights
North America
North America Dominates the Infectious Disease Therapeutics Market, Driven by the World's Highest Healthcare Spending, Leading Drug Development Ecosystem, and Broad Therapeutic Access*
North America leads the global infectious disease therapeutics market with approximately 42–44% of total revenues in 2026 and a CAGR of approximately 6.2% through 2033. The United States is the undisputed largest country market, where the combination of the world's highest per-capita pharmaceutical spending, the FDA's efficient drug approval pathways, comprehensive insurance coverage for branded and generic anti-infectives, and the largest concentration of infectious disease pharmaceutical companies creates a uniquely favorable commercial environment. Major companies including Gilead Sciences, Pfizer, Merck & Co., AbbVie, and Bristol-Myers Squibb generate a large proportion of their global infectious disease revenues in the United States, where premium branded drug pricing, high treatment initiation rates, and extensive clinical trial infrastructure reinforce market leadership. Canada also contributes meaningfully to the regional market, with publicly funded healthcare systems that provide broad antiviral and antibiotic access and a growing domestic biotech sector with active infectious disease R&D programs.
North America's dominance in the infectious disease therapeutics market is also reinforced by the U.S. government's substantial investment in pandemic preparedness, BARDA-funded anti-infective development programs, and AMR research initiatives that continuously stimulate innovation within the commercial drug pipeline. The region's mature HIV treatment market — with one of the world's highest ART initiation rates and a growing long-acting injectable segment — provides a stable and high-value revenue base that supports consistent regional market leadership through 2033 and beyond.
Asia Pacific
Asia Pacific Is the Fastest-Growing Market for Infectious Disease Therapeutics, Driven by the Highest Global Communicable Disease Burden and Rapidly Expanding Healthcare Systems*
Asia Pacific is the fastest-growing regional market for infectious disease therapeutics, projected to expand at a CAGR of approximately 8.7% through 2033, with a current market share of approximately 21–23% in 2026. India is the world's highest-burden country for tuberculosis and carries a massive load of antibiotic-resistant infections, viral hepatitis, dengue, and HIV — creating an enormous clinical demand for infectious disease therapeutics that is supported by both the world's largest generic pharmaceutical manufacturing industry and growing branded drug market development. China's infectious disease therapeutics market is expanding rapidly through a combination of national healthcare reform, improved hospital diagnostic capabilities, and government-funded disease control programs for hepatitis B, HIV, and tuberculosis that are systematically improving treatment initiation and adherence rates. Companies including Cipla, Sun Pharmaceutical, and Chinese domestic pharma players are significant suppliers in the regional generic anti-infective market, while multinationals including Gilead, GSK, and AbbVie are expanding their branded therapeutic presence through local partnerships and access pricing strategies.
Japan and South Korea are mature, high-value markets within the Asia Pacific infectious disease therapeutics space, with well-funded national health insurance systems that support broad access to both branded and generic anti-infective drugs, and advanced domestic pharmaceutical industries that contribute to regional market development. Southeast Asia — particularly Indonesia, Vietnam, Thailand, and the Philippines — represents a high-growth frontier for the infectious disease therapeutics market, as improving healthcare infrastructure, growing middle-class insurance enrollment, and expanding hospital networks are increasing the volume of diagnosed and treated infectious disease cases at a rate that significantly outpaces mature market growth. The infectious disease therapeutics market across Asia Pacific is expected to sustain above-average growth through the entire forecast period, supported by persistent high infection burden, continued healthcare investment, and expanding therapeutic access.
Top Key Players
-
Gilead Sciences Inc. (United States)
-
Pfizer Inc. (United States)
-
Merck & Co. Inc. (United States)
-
AbbVie Inc. (United States)
-
GlaxoSmithKline plc (GSK) (United Kingdom)
-
Johnson & Johnson (Janssen Pharmaceuticals) (United States)
-
AstraZeneca plc (United Kingdom / Sweden)
-
Bristol-Myers Squibb Company (United States)
-
ViiV Healthcare Ltd. (United Kingdom)
-
Roche Holding AG (Switzerland)
-
Novartis AG (Switzerland)
-
Sanofi S.A. (France)
-
Cipla Limited (India)
-
Sun Pharmaceutical Industries Ltd. (India)
-
Shionogi & Co. Ltd. (Japan)
Recent Developments
-
2024 – Gilead Sciences received FDA approval for Sunlenca (lenacapavir), a twice-yearly injectable HIV capsid inhibitor, marking a landmark advance in long-acting HIV therapy for treatment-experienced patients with multidrug-resistant HIV, and initiated the PURPOSE clinical trial program evaluating lenacapavir for HIV pre-exposure prophylaxis in key populations globally.
-
2024 – Pfizer expanded its infectious disease portfolio through continued investment in novel antibiotic development via its collaboration with Re-Pharma, targeting pan-resistant Gram-negative pathogens, while also pursuing commercial growth of Paxlovid (nirmatrelvir/ritonavir) across additional high-burden COVID-19 markets in Asia Pacific and Latin America.
-
2024 – AbbVie reported strong global commercial growth for Mavyret (glecaprevir/pibrentasvir) in the hepatitis C market, with particularly strong performance in pan-genotypic treatment-naive patient populations in Asia Pacific and Latin America markets where hepatitis C prevalence and diagnosis rates are improving through government screening programs.
-
2025 – ViiV Healthcare announced positive Phase III results from the LATITUDE study evaluating long-acting injectable cabotegravir and rilpivirine in HIV-positive individuals with high social barriers to oral medication adherence, demonstrating the regimen's superiority over oral ART in real-world adherence-challenged populations and supporting potential label expansion for this patient segment.
-
2025 – Shionogi & Co. received approval in Japan for cefiderocol (Fetroja) for use in additional carbapenem-resistant Gram-negative infections, reinforcing Shionogi's position as a leading innovator in the novel antibiotic space and supporting broader commercial launch activities for this critically important anti-infective in Asia Pacific hospital markets.
Market Trends
Long-Acting Anti-Infective Formulations, Phage Therapy, Microbiome-Based Therapeutics, and AI-Driven Drug Discovery Are the Defining Innovation Trends in the Infectious Disease Therapeutics Market*
The most transformative trend reshaping the infectious disease therapeutics market is the rapid commercial and clinical expansion of long-acting anti-infective formulations. Beginning with the HIV sector — where monthly and twice-yearly injectable ART regimens are now commercially available and rapidly growing — the long-acting formulation paradigm is being actively explored for other high-burden infections including tuberculosis, malaria, and hepatitis B. Long-acting formulations fundamentally change the patient-treatment relationship by eliminating the daily pill burden, reducing adherence failures, and enabling clinicians to confirm treatment administration at each clinical contact. This formulation innovation is expected to command significant pricing premiums, drive high market uptake among adherence-challenged patient populations, and generate strong revenue growth within the broader infectious disease drug market through 2033.
A second major trend is the growing interest in bacteriophage therapy and microbiome-based therapeutics as novel approaches to treating drug-resistant bacterial infections. Bacteriophages — viruses that infect and kill specific bacterial pathogens — are being clinically evaluated as precision therapies for patients with life-threatening MRSA, Mycobacterium abscessus, and pan-resistant Gram-negative infections that have exhausted all antibiotic options. While still largely in clinical trial and compassionate use settings, early case series and small clinical studies have demonstrated remarkable responses in some patients, generating significant scientific and commercial interest in phage-based infectious disease therapeutics. Several biotech companies and academic medical centers are actively developing phage banks, phage engineering platforms, and standardized manufacturing processes that could make phage therapy a commercially available infectious disease treatment modality within the forecast period.
Segments Covered in the Report
By Drug Class:
-
Antiviral Therapeutics
-
HIV Antiretrovirals (ART)
-
Hepatitis B and C Antivirals
-
Respiratory Antivirals (Influenza, COVID-19, RSV)
-
Other Antiviral Drugs
-
-
Antibiotic Therapeutics
-
Broad-Spectrum Antibiotics
-
Narrow-Spectrum / Targeted Antibiotics
-
Novel Antibiotic Classes
-
-
Antifungal Therapeutics
-
Azoles
-
Echinocandins
-
Polyenes
-
Novel Antifungal Agents
-
-
Antiparasitic Therapeutics
-
Antimalarials
-
Antiprotozoals
-
Antihelminthics
-
-
Other Drug Classes
By Indication:
-
HIV/AIDS
-
Hepatitis B and C
-
Tuberculosis
-
Respiratory Infections (Including COVID-19, Influenza, RSV)
-
Sexually Transmitted Infections (STIs)
-
Fungal Infections
-
Parasitic Infections
-
Other Indications
By Mode of Administration:
-
Oral
-
Injectable / Intravenous
-
Topical
-
Other Modes
By Distribution Channel:
-
Hospital Pharmacies
-
Retail and Community Pharmacies
-
Government and Public Health Programs
-
Online Pharmacies
-
Other Channels
By End User:
-
Hospitals and Tertiary Care Centers
-
Specialty Infectious Disease Clinics
-
Community Clinics and Primary Care
-
Home Care Settings
-
Government Health Programs
-
Other End Users
By Region:
-
North America
-
United States
-
Canada
-
-
Europe
-
Germany
-
United Kingdom
-
France
-
Italy
-
Spain
-
Rest of Europe
-
-
Asia Pacific
-
China
-
India
-
Japan
-
South Korea
-
Australia
-
Rest of Asia Pacific
-
-
Latin America
-
Brazil
-
Mexico
-
Rest of Latin America
-
-
Middle East & Africa
-
GCC Countries
-
South Africa
-
Rest of Middle East & Africa
-
"Built for Every Level — From Startups to Industry Giants"
Here Is Exactly How This Report Works for You
-
For Tier 1 global pharmaceutical corporations, large hospital systems, and institutional investors, this report delivers granular competitor revenue analysis, branded versus generic drug revenue source mapping, and pipeline benchmarking across all major infectious disease drug classes and geographies — enabling precise portfolio investment decisions, licensing and acquisition target identification, and strategic commercial planning in one of pharma's largest and most dynamically evolving therapeutic categories.
-
For Tier 2 and Tier 3 biotech companies, emerging market generic manufacturers, and specialty infectious disease therapy distributors, this report provides detailed supply-demand dynamics, drug patent cliff analysis, pricing trend benchmarks, and white-space indication mapping — giving growing organizations the market intelligence needed to prioritize drug development programs, target high-growth geographies, and build commercially competitive positioning in the infectious disease therapeutics landscape.
-
For all decision-makers navigating today's complex geopolitical and regulatory environment, this report delivers clear analysis of how international trade policies, API supply chain vulnerabilities concentrated in India and China, cross-border regulatory divergences, and global health emergency frameworks are impacting drug manufacturing costs, supply security, and market access — equipping pharmaceutical executives and health system leaders with the situational intelligence to make confident, proactive decisions in a rapidly changing global health landscape.
Frequently Asked Questions
Question 1: What is the current market size of the infectious disease therapeutics market and what is the growth forecast through 2033?
Answer: The infectious disease therapeutics market is valued at USD 97.40 billion in 2025 and is projected to reach USD 161.72 billion by 2033. The market is expected to grow at a CAGR of 6.63% from 2026 to 2033.
Question 2: What are the primary growth drivers in the infectious disease therapeutics market?
Answer: The infectious disease therapeutics market is driven primarily by the escalating global burden of antimicrobial resistance, rising prevalence of viral infections including HIV and hepatitis, and growing pharmaceutical R&D investment in novel anti-infective drug pipelines. Post-pandemic infectious disease preparedness investment and expanding healthcare access in emerging markets are further accelerating market growth.
Question 3: Which drug class holds the largest share in the infectious disease therapeutics market?
Answer: The antiviral therapeutics segment holds the largest revenue share, accounting for approximately 43–45% of total market revenues in 2025, driven by the enormous scale of global HIV antiretroviral therapy programs, hepatitis C cures, and COVID-19 antiviral markets. The antifungal therapeutics segment is the fastest-growing drug class, expanding at approximately 9.2% CAGR through 2033.
Question 4: Which region leads the infectious disease therapeutics market and which is growing fastest?
Answer: North America leads the infectious disease therapeutics market with approximately 42–44% of global revenues in 2026, supported by the world's highest healthcare spending and leading pharmaceutical development ecosystem. Asia Pacific is the fastest-growing region, expanding at approximately 8.7% CAGR through 2033 driven by the world's highest communicable disease burden and rapidly expanding healthcare access.
Question 5: Who are the top companies in the infectious disease therapeutics market?
Answer: Leading companies in the infectious disease therapeutics market include Gilead Sciences, Pfizer, Merck & Co., AbbVie, GSK, Johnson & Johnson (Janssen), AstraZeneca, ViiV Healthcare, Roche, Shionogi, and Cipla, among others. These companies compete through innovative drug development, long-acting formulation strategies, clinical evidence generation, and global market access programs.
