High Purity Citric Acid Market Overview
Ultra-Pure Fermentation Products Transform Pharmaceutical, Biotechnology and Specialty Chemical Applications
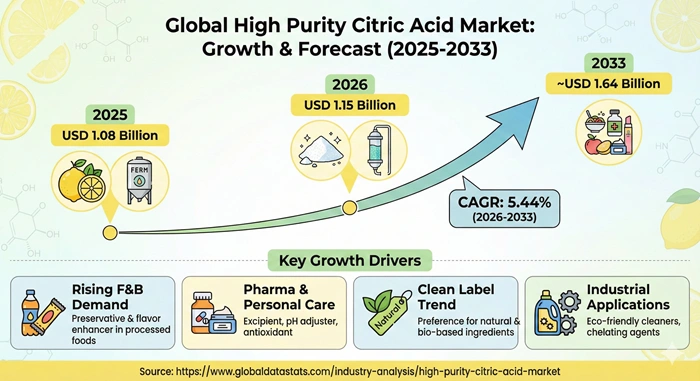
The global high purity citric acid market size is valued at USD 1.08 billion in 2025 and is predicted to increase from USD 1.15 billion in 2026 to approximately USD 1.64 billion by 2033, growing at a CAGR of 5.44% from 2026 to 2033. This specialized industry encompasses citric acid products exceeding 99.5% purity manufactured through advanced fermentation and purification processes serving pharmaceutical formulations, biotechnology research, laboratory reagents, nutraceuticals, and premium food applications. High purity citric acid demonstrates exceptional consistency, minimal impurity levels, and stringent quality control meeting regulatory requirements for drug manufacturing, cell culture media, and analytical testing where contamination risks must be eliminated.
The high purity citric acid market addresses escalating demand for pharmaceutical-grade and laboratory-grade ingredients where product safety, reproducibility, and regulatory compliance create quality requirements beyond standard food-grade citric acid specifications. These ultra-pure materials demonstrate exceptional batch-to-batch consistency, controlled trace metal content, and comprehensive documentation including certificates of analysis, regulatory filings, and traceability records essential for Good Manufacturing Practice compliance. Major application sectors including injectable drug formulations, biotechnology cell cultures, diagnostic reagents, and premium nutraceuticals require high purity citric acid delivering acidulant properties, chelation functions, buffering capacity, and antioxidant protection without introducing contaminants affecting product efficacy or patient safety throughout shelf life.
AI Impact on the High Purity Citric Acid Industry
Machine Learning and Process Analytics Revolutionize Fermentation Optimization and Quality Assurance
Artificial intelligence transforms the high purity citric acid market through advanced process control systems monitoring fermentation parameters including pH levels, dissolved oxygen, temperature profiles, and nutrient concentrations in real-time, automatically adjusting conditions maximizing citric acid yields while minimizing impurity formation. Machine learning algorithms analyze historical batch data from thousands of production runs identifying optimal substrate formulations, inoculation densities, and feeding strategies achieving 15-20% productivity improvements compared to traditional empirical methods. These AI-powered systems enable predictive quality assurance forecasting final product purity based on intermediate process measurements allowing early intervention preventing off-specification batches reaching expensive downstream purification stages. Deep learning models trained on spectroscopic data perform rapid in-line quality verification detecting trace contaminants including heavy metals, residual proteins, and byproduct acids at parts-per-million levels impossible through conventional laboratory testing timelines.
Advanced AI applications extend beyond fermentation optimization to revolutionize supply chain management and regulatory compliance within the high purity citric acid market landscape. Neural networks predict raw material quality variations analyzing supplier performance data, agricultural commodity trends, and seasonal factors enabling proactive sourcing strategies maintaining consistent feedstock specifications. Computer vision systems integrated with microscopy equipment perform automated microbial strain verification ensuring production cultures maintain genetic stability and optimal citric acid biosynthesis capabilities across serial transfers. Natural language processing mines regulatory databases, scientific literature, and customer specifications extracting critical quality attributes, testing requirements, and documentation standards guiding quality system design and continuous improvement initiatives. These AI-driven capabilities position manufacturers to achieve pharmaceutical-grade consistency while reducing production costs 10-15% through waste elimination, energy optimization, and accelerated batch cycles throughout forecast period.
Growth Factors
Pharmaceutical Sector Expansion and Biotechnology Innovation Drive Unprecedented High Purity Citric Acid Demand
The high purity citric acid market experiences robust growth propelled by pharmaceutical industry expansion requiring ultra-pure excipients for injectable formulations, effervescent tablets, and liquid medications where citric acid functions as buffering agent, chelating compound, and stability enhancer maintaining drug efficacy throughout shelf life. Injectable drug production accounting for 30% of global pharmaceutical manufacturing consumes substantial pharmaceutical-grade citric acid volumes where trace metal specifications below 10 parts-per-million and endotoxin limits below 0.5 EU/mg ensure patient safety preventing adverse reactions. Effervescent tablet formulations incorporating citric acid with carbonate salts create carbon dioxide liberation enabling rapid disintegration and improved bioavailability for pain relievers, vitamin supplements, and electrolyte replacement products commanding premium pricing. Liquid medication development particularly pediatric syrups and geriatric solutions utilize citric acid achieving palatable taste profiles while providing preservative benefits and pH adjustment preventing microbial growth during multi-dose container use.
Biotechnology sector growth drives high purity citric acid market expansion through specialized applications in cell culture media where citric acid maintains optimal pH conditions supporting mammalian cell growth for therapeutic protein production, vaccine manufacturing, and regenerative medicine applications. Biopharmaceutical production employing Chinese Hamster Ovary cells and human embryonic kidney cells requires precisely formulated culture media containing high purity citric acid buffers preventing pH excursions that reduce cell viability and product yields worth millions per batch. Gene therapy vector production and CAR-T cell manufacturing representing cutting-edge medical treatments demand ultra-pure reagents including laboratory-grade citric acid exceeding 99.8% purity eliminating contaminants interfering with sensitive genetic engineering processes. Contract research organizations and academic laboratories consume substantial high purity citric acid volumes for analytical testing, method development, and quality control procedures where reproducibility and traceability requirements mandate premium-grade materials certified through ISO 17025 accredited suppliers throughout forecast period.

Market Outlook
Clean Label Trends and Bio-Based Production Position Industry for Sustained Long-Term Growth
The high purity citric acid market outlook remains exceptionally positive as manufacturers invest substantially in advanced fermentation technologies producing pharmaceutical-grade materials from renewable feedstocks addressing sustainability concerns while meeting stringent regulatory requirements. Bio-based production utilizing corn-derived dextrose, sugarcane molasses, or cassava starch as fermentation substrates combined with Aspergillus niger microbial strains achieves citric acid concentrations exceeding 200 grams per liter through optimized cultivation protocols. Advanced downstream purification employing membrane filtration, crystallization, and ion exchange chromatography removes residual proteins, nucleic acids, and trace metals achieving pharmaceutical-grade specifications with 99.5-99.9% purity validated through United States Pharmacopeia, European Pharmacopoeia, and Japanese Pharmacopoeia monograph testing. Continuous manufacturing technologies replacing traditional batch processing demonstrate 30-40% productivity improvements enabling cost-competitive pharmaceutical-grade production serving price-sensitive generic drug formulations and emerging market healthcare systems.
Investment trends within the high purity citric acid market reflect growing confidence in long-term demand fundamentals as leading producers expand pharmaceutical-grade capacity targeting biopharmaceutical, nutraceutical, and specialty chemical applications. Jungbunzlauer, Cargill, ADM, and COFCO Biochemical maintain robust capital expenditure programs commissioning dedicated pharmaceutical production lines incorporating advanced instrumentation, environmental control systems, and quality management infrastructure meeting FDA, EMA, and other regulatory agency requirements. Acquisition activity intensifies as specialty chemical companies pursue strategic combinations accessing established pharmaceutical customer relationships, regulatory dossiers, and quality certifications accelerating market entry timelines. Asia Pacific capacity expansions particularly China and India target domestic pharmaceutical manufacturing growth while developing export capabilities serving European and North American markets. These favorable dynamics position the market for sustained expansion exceeding general citric acid industry growth rates through 2033.
Expert Speaks
-
Brian Sikes, Chief Operating Officer of Cargill, emphasized strategic priorities: "We're investing significantly in safety, training and innovation across our operations. By advancing automation and digitalizing how we work, we're modernizing to help our people work smarter, faster and safer, all while better serving customers and reliably nourishing the world. Our nature-based, zero-calorie sweeteners and ingredient innovations are expanding access to healthy food choices".
-
Pankaj Kumar, Asia Middle East and Africa Sourcing Lead at Mondelez International, articulated supplier perspective: "Cargill is one of our supply partners who is enhancing their capabilities to develop new products that are faster to adapt and implement in markets. This collaborative approach ensures we stay ahead of changing consumer demands across diverse regional preferences and regulatory landscapes while maintaining quality standards".
-
Jan Dieleman, President of Cargill's Ocean Transportation Business, highlighted sustainability commitment: "This is our moment to ensure that maritime shipping thrives responsibly, innovatively and sustainably. The seas ahead may be uncertain, but the destination is clear: a sustainable future. This vision extends across our entire supply chain including specialty ingredients and biochemicals where environmental stewardship drives innovation".
Key Report Takeaways
-
Asia Pacific dominates the high purity citric acid market with commanding share exceeding 39%, driven by concentrated pharmaceutical manufacturing across China, India, and Japan where generic drug production, biotechnology development, and nutraceutical formulation consume substantial ultra-pure citric acid volumes supported by expanding healthcare infrastructure and export-oriented manufacturing.
-
North America emerges as the fastest-growing regional market with projected CAGR approaching 5.9% through 2033, propelled by biopharmaceutical innovation, contract manufacturing expansion, advanced cell therapy development, and stringent regulatory standards requiring pharmaceutical-grade ingredients meeting United States Pharmacopeia specifications and FDA compliance.
-
Pharmaceutical grade segment representing ≥99.5% purity dominates market share accounting for approximately 61% of global revenue due to extensive injectable drug formulations, effervescent tablet production, and liquid medication applications where patient safety requirements mandate minimal trace metal content and comprehensive regulatory documentation.
-
Biotechnology and laboratory applications constitute fastest-growing end-use segment with anticipated CAGR exceeding 7.2% through 2033, attributed to cell culture media demand for biopharmaceutical production, gene therapy vector manufacturing, and analytical testing requiring ultra-pure reagents supporting reproducible research outcomes and regulatory submissions.
-
Laboratory/reagent grade segment exceeding 99.8% purity exhibits exceptional growth potential with projected CAGR of 6.8%, driven by stringent analytical testing requirements, biotechnology research expansion, and quality control procedures where contaminant sensitivity necessitates premium-grade materials certified through ISO standards and comprehensive certificates of analysis.
-
Liquid citric acid format demonstrates substantial market momentum capturing growing share through convenient handling, precise dosing capabilities, and reduced dust exposure addressing occupational health considerations while enabling automated dispensing systems in pharmaceutical manufacturing and biotechnology facilities throughout forecast period.
Market Scope
| Report Coverage Details | |
|---|---|
| Market Size by 2033 | USD 1.64 Billion |
| Market Size by 2025 | USD 1.08 Billion |
| Market Size by 2026 | USD 1.15 Billion |
| Market Growth Rate from 2026 to 2033 | CAGR of 5.44% |
| Dominating Region | Asia Pacific |
| Fastest Growing Region | North America |
| Base Year | 2025 |
| Forecast Period | 2026 to 2033 |
| Segments Covered | Purity Grade, Form, Application, End-Use, Region |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Pharmaceutical Manufacturing Growth and Quality Standards Escalation Accelerate High Purity Citric Acid Adoption
| Impact Factor | (≈) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Pharmaceutical Industry Expansion | +2.0% | Global, Led by Asia Pacific | 2026-2033 |
| Biotechnology Sector Growth | +1.6% | North America, Europe | 2026-2033 |
| Regulatory Standards Tightening | +1.2% | Developed Markets | 2026-2033 |
| Nutraceutical Market Development | +0.9% | Global Consumer Markets | 2027-2033 |
The high purity citric acid market benefits substantially from global pharmaceutical industry expansion where injectable drug production, effervescent formulations, and liquid medications require ultra-pure excipients meeting stringent regulatory specifications for trace metals, endotoxins, and microbial contamination. Injectable formulation development representing fastest-growing pharmaceutical segment consumes substantial pharmaceutical-grade citric acid as buffering agent and chelating compound preventing oxidative degradation and metal-catalyzed decomposition extending drug stability from months to years. Generic drug manufacturing particularly in India, China, and emerging markets drives cost-competitive high purity citric acid demand as producers pursue bioequivalence with branded formulations while optimizing production economics through standardized excipient specifications. Effervescent tablet technology gaining market share across pain management, vitamin supplements, and electrolyte replacement applications utilizes citric acid-carbonate reactions creating consumer-preferred dosage forms with improved bioavailability and patient compliance compared to conventional tablets.
Biotechnology sector expansion drives high purity citric acid market growth through specialized cell culture applications where mammalian cell lines producing therapeutic proteins, monoclonal antibodies, and vaccines require precisely formulated growth media maintaining optimal pH conditions between 6.8-7.4. Biopharmaceutical production projected to exceed 400 billion dollars annually by 2030 incorporates citric acid buffers in culture media, purification processes, and final formulation stabilizing sensitive biological products against aggregation, denaturation, and loss of potency during manufacturing and storage. Gene therapy and cell therapy representing cutting-edge medical treatments demand ultra-pure laboratory-grade citric acid exceeding 99.8% purity eliminating contaminants including trace metals, residual solvents, and microbial byproducts interfering with viral vector production and genetic engineering protocols. Contract research organizations and academic institutions conducting drug discovery research, method development, and quality control testing consume substantial high purity citric acid volumes where reproducibility requirements mandate certified reference materials traceable to international standards throughout forecast period.

Restraints Impact Analysis
Premium Pricing and Production Complexity Challenge Market Penetration Across Price-Sensitive Applications
| Restraint Factor | (≈) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High Production Costs | -1.1% | Global Manufacturing | 2026-2033 |
| Regulatory Compliance Expenses | -0.8% | Pharmaceutical Producers | 2026-2033 |
| Competition from Food-Grade Products | -0.7% | Cost-Sensitive Segments | 2026-2031 |
| Supply Chain Complexity | -0.5% | Emerging Markets | 2026-2030 |
The high purity citric acid market faces growth constraints from substantial production cost premiums where pharmaceutical-grade materials command prices 3-5 times higher than food-grade equivalents reflecting advanced purification requirements, quality testing expenses, and regulatory compliance documentation. Multi-stage purification processes incorporating membrane filtration, crystallization cycles, activated carbon treatment, and ion exchange chromatography add 40-60% manufacturing costs compared to conventional food-grade production terminating after initial crystallization. Analytical testing requirements including heavy metals analysis by ICP-MS, residual protein quantification, endotoxin testing by LAL method, and microbial limit testing create quality control expenses exceeding 5% of production costs versus 1-2% for food-grade materials. Regulatory compliance costs including facility validation, equipment qualification, process validation, and ongoing stability studies represent substantial fixed expenses limiting participation to established pharmaceutical ingredient manufacturers with multi-million dollar quality infrastructure investments.
Competition from food-grade citric acid constrains high purity citric acid market penetration across applications where quality requirements fall between food and pharmaceutical specifications creating customer reluctance to pay premium pricing. Nutraceutical manufacturers particularly in emerging markets source lower-cost food-grade citric acid for dietary supplement formulations where regulatory oversight remains less stringent than pharmaceutical products despite consumer safety considerations. Personal care and cosmetic applications utilize food-grade citric acid as pH adjuster and chelating agent where purity specifications exceeding 99% satisfy industry standards without pharmaceutical-grade documentation requirements. Generic drug producers in price-sensitive markets pursue cost optimization sourcing borderline-specification materials meeting minimum compendial requirements rather than premium pharmaceutical-grade citric acid commanding higher prices. These competitive dynamics pressure pharmaceutical-grade producers to justify premium pricing through superior consistency, comprehensive documentation, and technical support services differentiating products beyond basic quality specifications throughout forecast period.
Opportunities Impact Analysis
Advanced Therapies and Personalized Medicine Create Substantial Growth Avenues Beyond Traditional Applications
| Opportunity Factor | (≈) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Cell and Gene Therapy Growth | +1.5% | North America, Europe | 2027-2033 |
| Continuous Manufacturing Adoption | +1.2% | Pharmaceutical Manufacturing | 2026-2033 |
| Clean Label Nutraceuticals | +0.9% | Global Consumer Markets | 2026-2033 |
| Personalized Medicine Development | +0.7% | Advanced Healthcare Markets | 2028-2033 |
The high purity citric acid market stands to capture substantial value from cell and gene therapy sector expansion requiring ultra-pure reagents for viral vector production, CAR-T cell manufacturing, and stem cell cultivation where contaminant sensitivity demands laboratory-grade materials exceeding pharmaceutical specifications. Viral vector manufacturing for gene therapy producing lentiviral, adeno-associated viral, and adenoviral vectors utilizes high purity citric acid in culture media formulations, purification buffers, and final formulation stabilization preventing aggregation and maintaining transduction efficiency. CAR-T cell therapy production requiring ex vivo T-cell expansion and genetic modification consumes specialized culture media incorporating ultra-pure citric acid buffers maintaining physiological pH during intensive cell proliferation phases generating billions of engineered cells per patient dose. These advanced therapy applications command premium pricing where batch failures costing millions create willingness to pay 2-3 times standard pharmaceutical-grade prices for materials providing additional quality assurance and comprehensive regulatory support.
Continuous manufacturing technology adoption presents exceptional growth opportunities for the high purity citric acid market as pharmaceutical producers transition from batch processing to integrated continuous systems requiring consistent raw material quality and real-time release testing capabilities. Continuous direct compression tablet manufacturing, continuous granulation systems, and continuous crystallization processes demand ultra-pure excipients including citric acid demonstrating minimal batch-to-batch variability and real-time analytical verification enabling process analytical technology implementation. These advanced manufacturing systems achieving 30-50% productivity improvements create demand for premium-grade citric acid suppliers offering comprehensive technical support, process development assistance, and regulatory collaboration accelerating FDA approval timelines. Clean label nutraceutical segment expansion driven by consumer preferences for natural ingredients, transparent sourcing, and sustainable production creates opportunities for bio-based high purity citric acid positioning products as naturally-derived alternatives to synthetic acidulants while delivering pharmaceutical-grade consistency supporting health claims and regulatory compliance throughout forecast period.

Segment Analysis
By Purity Grade: Pharmaceutical Grade (≥99.5% Purity)
Injectable Drug Formulations and Regulatory Requirements Sustain Pharmaceutical Grade Segment Market Leadership
Pharmaceutical grade citric acid representing ≥99.5% purity commands the largest share within the high purity citric acid market, accounting for approximately 61% of global revenue with continued expansion projected at steady CAGR of 5.3% through 2033. This segment encompasses ultra-pure materials meeting United States Pharmacopeia, European Pharmacopoeia, and Japanese Pharmacopoeia monograph specifications including stringent limits for heavy metals, residual solvents, sulfate ash, and readily carbonizable substances ensuring patient safety across injectable drugs, oral medications, and topical formulations. The high purity citric acid market benefits from pharmaceutical-grade materials' extensive regulatory documentation including Drug Master Files, certificates of analysis, stability data, and manufacturing process descriptions enabling streamlined drug approval processes. Injectable formulation applications particularly parenteral nutrition solutions, antibiotic reconstitution systems, and chelation therapy products require pharmaceutical-grade citric acid preventing adverse reactions from trace metal contaminants while providing essential buffering and antioxidant functions.
Asia Pacific leads pharmaceutical-grade consumption within the market, driven by concentrated generic drug manufacturing across India and China where cost-competitive production combined with international quality certifications enables global export growth. Leading pharmaceutical ingredient suppliers including Jungbunzlauer, COFCO Biochemical, Gadot Biochemical, and Weifang Ensign Industry maintain dedicated pharmaceutical production facilities incorporating cleanroom environments, validated analytical laboratories, and comprehensive quality management systems meeting FDA, EMA, and WHO Good Manufacturing Practice requirements. Indian pharmaceutical manufacturers particularly in Hyderabad, Ahmedabad, and Bangalore pharmaceutical clusters consume substantial pharmaceutical-grade citric acid volumes producing injectable antibiotics, effervescent tablets, and liquid formulations serving domestic healthcare needs while exporting to developed markets. Chinese biotechnology companies in Shanghai, Beijing, and Suzhou life science parks specify pharmaceutical-grade citric acid for cell culture media, diagnostic reagents, and biopharmaceutical purification buffers supporting domestic innovation initiatives. North American and European pharmaceutical-grade applications emphasize premium-priced specialty products including orphan drugs, biologics, and advanced therapy medicinal products where quality assurance and supply chain security justify pharmaceutical-grade materials from established Western suppliers throughout forecast period.
By Application: Biotechnology and Laboratory Use
Cell Culture Media and Analytical Testing Position Biotechnology as Fastest-Growing Application Segment
Biotechnology and laboratory applications represent the fastest-growing segment within the high purity citric acid market, projected to expand at exceptional CAGR of 7.2% through 2033 driven by biopharmaceutical production, genomic research, and analytical chemistry applications requiring ultra-pure reagents. This sector encompasses cell culture media formulations where citric acid maintains optimal pH conditions supporting mammalian cell growth for therapeutic protein production including monoclonal antibodies, recombinant enzymes, and fusion proteins achieving expression titers exceeding 5 grams per liter. The high purity citric acid market benefits from biotechnology segment's willingness to pay premium pricing for laboratory-grade materials exceeding 99.8% purity where trace metal specifications below 5 parts-per-million and comprehensive certificates of analysis ensure reproducible research outcomes and regulatory compliance. Viral vector production for gene therapy and vaccine manufacturing utilizes high purity citric acid in perfusion culture systems, purification buffers, and final formulation achieving yields supporting commercial-scale manufacturing ranging thousands to millions of doses annually.
North America and Europe lead biotechnology citric acid adoption within the market, driven by concentrated biopharmaceutical manufacturing across Massachusetts, California, New Jersey, Switzerland, and Ireland biotech hubs where established contract development and manufacturing organizations serve global pharmaceutical companies. United States biotechnology sector consuming over 40% of global high purity citric acid laboratory-grade volumes supports diverse applications including antibody production by companies like Regeneron, Amgen, and Genentech, cell therapy manufacturing by Novartis and Gilead, and vaccine production by Pfizer and Moderna utilizing specialized culture media and buffers. European biopharmaceutical producers including Roche, Sanofi, and Lonza specify ultra-pure citric acid for Good Manufacturing Practice production facilities where regulatory compliance and supply chain security considerations favor established pharmaceutical ingredient suppliers offering comprehensive technical support and regulatory documentation. Leading laboratory reagent suppliers including Merck KGaA, Thermo Fisher Scientific, and VWR International distribute high purity citric acid to academic research institutions, contract research organizations, and quality control laboratories requiring certified reference materials traceable to international standards supporting method validation and regulatory submissions throughout forecast period.

Regional Insights
Asia Pacific
Manufacturing Scale and Pharmaceutical Sector Growth Establish Regional Dominance in High Purity Citric Acid Consumption
Asia Pacific commands the high purity citric acid market with dominant share exceeding 39%, driven by concentrated pharmaceutical manufacturing across China, India, and Japan where generic drug production, biotechnology development, and nutraceutical formulation consume substantial ultra-pure citric acid volumes supporting domestic healthcare needs and global exports. China alone accounts for over 40% of global citric acid production capacity with pharmaceutical-grade facilities operated by COFCO Biochemical, RZBC Group, and Weifang Ensign Industry incorporating advanced purification technologies and quality systems meeting international regulatory standards. The region benefits from vertical integration spanning fermentation substrate production through final pharmaceutical-grade material distribution with corn-based dextrose, cassava starch, and molasses feedstocks manufactured locally reducing supply chain costs and ensuring consistent raw material quality. India's pharmaceutical industry particularly Hyderabad and Ahmedabad clusters consumes growing high purity citric acid volumes producing injectable drugs, effervescent formulations, and active pharmaceutical ingredients serving domestic market exceeding 1.4 billion population while exporting to over 200 countries worldwide.
Japan represents technology leadership within Asia Pacific region through advanced biotechnology applications and stringent quality standards where pharmaceutical and laboratory-grade citric acid supports therapeutic protein production, regenerative medicine research, and precision analytical testing. Leading regional producers including Jungbunzlauer Asia Pacific, Gadot Biochemical, and Tate & Lyle maintain technical centers and customer support teams serving Japanese pharmaceutical companies, biotechnology startups, and research institutions requiring comprehensive documentation and application expertise. Chinese biotechnology sector expansion particularly Shanghai, Beijing, and Shenzhen innovation hubs drives specialized laboratory-grade citric acid demand for cell culture media, gene therapy vector production, and diagnostic reagent manufacturing where government initiatives support domestic biopharmaceutical capabilities. Southeast Asian nations including Thailand, Vietnam, and Indonesia develop pharmaceutical manufacturing capacity creating diversified demand opportunities across generic drugs, nutraceuticals, and personal care applications. The region's market dominance continues strengthening through 2033 as domestic consumption growth, manufacturing capacity expansion, and quality certification achievement create self-reinforcing dynamics supporting continued leadership position.
North America
Biopharmaceutical Innovation and Regulatory Leadership Position Region as Fastest-Growing Geographic Market
North America emerges as the fastest-growing regional high purity citric acid market with projected CAGR of 5.9% through 2033, propelled by biopharmaceutical innovation, advanced cell therapy development, contract manufacturing expansion, and stringent regulatory standards requiring pharmaceutical-grade ingredients meeting United States Pharmacopeia specifications. The United States dominates regional market activity through established biotechnology clusters across Massachusetts, California, New Jersey, and North Carolina where therapeutic antibody production, gene therapy development, and cell therapy manufacturing create specialized demand for ultra-pure laboratory-grade citric acid. FDA regulatory framework establishing comprehensive quality requirements for pharmaceutical excipients including extensive documentation, stability data, and supplier qualification creates barriers favoring established high purity citric acid suppliers with proven regulatory track records and comprehensive technical support capabilities.
United States high purity citric acid market specifically benefits from biopharmaceutical sector leadership where companies including Regeneron, Amgen, Gilead, and Genentech consume substantial pharmaceutical-grade citric acid volumes producing blockbuster antibody drugs, CAR-T cell therapies, and gene therapy products achieving billions in annual revenues. Contract development and manufacturing organizations including Lonza Biologics, WuXi Biologics, and Samsung Biologics operate large-scale bioreactors requiring consistent high purity citric acid supplies for culture media formulations where batch failures costing millions create willingness to pay premium pricing for materials providing additional quality assurance. Canada contributes to North American growth through pharmaceutical manufacturing concentrated in Quebec and Ontario, biotechnology research at universities and institutes, and nutraceutical production serving health-conscious consumer segments. Leading pharmaceutical ingredient suppliers including Jungbunzlauer, Cargill specialty ingredients division, and ADM pharmaceutical solutions maintain North American distribution centers, technical support teams, and regulatory affairs specialists enabling close customer collaboration supporting formulation development, regulatory submissions, and supply chain optimization throughout forecast period.
Top Key Players
-
Jungbunzlauer Suisse AG (Switzerland)
-
Cargill Incorporated (United States)
-
Archer Daniels Midland Company (United States)
-
COFCO Biochemical Co. Ltd. (China)
-
Weifang Ensign Industry Co. Ltd. (China)
-
RZBC Group Co. Ltd. (China)
-
Gadot Biochemical Industries Ltd. (Israel)
-
Tate & Lyle PLC (United Kingdom)
-
Citrique Belge N.V. (Belgium)
-
TTCA Co. Ltd. (China)
-
Huangshi Xinghua Biochemical Co. Ltd. (China)
-
Merck KGaA (Germany)
-
Foodchem International Corporation (China)
-
Jiangsu Guoxin Union Energy Co. Ltd. (China)
-
Laiwu Taihe Biochemistry Co. Ltd. (China)
Recent Developments
-
Cargill Incorporated (2025): Announced 100 million dollars investment in new acidulant manufacturing facility in Brazil during Q1 2025, expanding production capacity serving Latin American pharmaceutical and food industries with advanced fermentation technologies and quality systems meeting international regulatory standards while supporting regional supply chain development and sustainability initiatives.
-
Cargill Incorporated (2025): Released 2025 Annual Report highlighting 154 billion dollars revenue and strategic reorganization into three business enterprises including Food, Agriculture & Trading, and Specialized Portfolio, with significant investments in nature-based ingredients innovation, automation, and digitalization supporting pharmaceutical-grade ingredient production capabilities and customer technical support services.
-
Pharmaceutical Industry (2025): Witnessed accelerating adoption of bio-based production methods utilizing renewable feedstocks and fermentation technologies achieving pharmaceutical-grade purity specifications while meeting environmental sustainability standards and regulatory requirements, with leading producers investing in continuous manufacturing capabilities and real-time analytical testing supporting process analytical technology implementation.
-
Biotechnology Sector (2025): Experienced robust expansion driven by increasing demand across healthcare, nutraceutical, and cosmetic sectors requiring high purity citric acid for formulation stability, pH regulation, and preservative functions, with innovations in fermentation technologies utilizing renewable feedstocks enhancing product purity and sustainability credentials aligning with global environmental standards.
-
Global Market (2025): Demonstrated strategic collaborations and technological licensing agreements among established players and new entrants aimed at expanding geographic reach, product portfolio diversification, and scalable high-efficiency production capacities, reflecting confidence in long-term growth prospects supported by increasing regulatory acceptance of bio-based ingredients and rising health-conscious consumer preferences.
Market Trends
Clean Label Formulations and Fermentation Technology Advancement Reshape Industry Competitive Dynamics
The high purity citric acid market experiences transformative trends centered on clean label positioning as pharmaceutical companies, nutraceutical brands, and cosmetic manufacturers promote naturally-derived ingredients addressing consumer preferences for recognizable components over synthetic alternatives. Bio-based production utilizing corn-derived dextrose, sugarcane molasses, or cassava starch as fermentation substrates combined with Aspergillus niger microbial strains creates marketing narratives emphasizing natural origins despite identical chemical structures to synthetic citric acid. Organic certification programs including USDA Organic, EU Organic, and various national standards create premium product tiers where high purity citric acid derived from certified organic feedstocks commands 20-30% price premiums serving natural products manufacturers pursuing comprehensive organic ingredient declarations. Non-GMO verification particularly important in North American and European markets drives fermentation substrate sourcing from non-genetically modified corn and cane sugar varieties despite higher costs and limited availability creating supply chain complexities for pharmaceutical-grade producers throughout forecast period.
Continuous manufacturing technology adoption trends reshape the high purity citric acid market as producers implement integrated fermentation-purification systems achieving consistent product quality, reduced environmental footprints, and improved economic efficiency compared to traditional batch processing. Continuous fermentation platforms maintaining steady-state microbial populations produce citric acid at consistent rates eliminating productivity fluctuations associated with batch cycle variations including lag phases, growth phases, and decline phases inherent to conventional processing. Membrane separation technologies replacing energy-intensive evaporation and crystallization steps reduce processing costs 15-25% while enabling modular capacity expansion responding to demand growth without large-scale facility investments. Real-time analytical monitoring incorporating spectroscopic sensors, chromatographic detectors, and automated sampling systems enables process analytical technology implementation supporting continuous quality verification and real-time release testing accelerating product availability eliminating traditional hold times awaiting laboratory analysis results throughout forecast period.
Segments Covered in the Report
By Purity Grade
-
Pharmaceutical Grade (≥99.5% Purity)
-
Laboratory/Reagent Grade (≥99.8% Purity)
-
Food Grade (99.0-99.5% Purity)
By Form
-
Anhydrous Powder
-
Monohydrate Powder
-
Liquid Solution
By Application
-
Pharmaceutical Formulations (Injectable Drugs, Oral Medications, Topical Products)
-
Biotechnology & Laboratory Use (Cell Culture Media, Analytical Reagents, Buffers)
-
Nutraceuticals & Functional Foods
-
Personal Care & Cosmetics
-
Industrial Applications
By End-Use
-
Pharmaceutical Manufacturing
-
Biotechnology Companies
-
Contract Research Organizations
-
Academic & Research Institutions
-
Nutraceutical Manufacturers
-
Cosmetic & Personal Care Producers
By Region
-
North America (United States, Canada, Mexico)
-
Europe (Germany, United Kingdom, France, Switzerland, Italy, Rest of Europe)
-
Asia Pacific (China, India, Japan, South Korea, Southeast Asia, Rest of Asia Pacific)
-
Latin America (Brazil, Argentina, Rest of Latin America)
-
Middle East & Africa (Israel, Saudi Arabia, South Africa, Rest of MEA)
Frequently Asked Questions
Question 1: What is the projected size of the high purity citric acid market by 2033?
Answer: The global high purity citric acid market is expected to reach approximately USD 1.64 billion by 2033, growing from USD 1.15 billion in 2026. This substantial expansion reflects increasing pharmaceutical manufacturing, biotechnology applications, and regulatory standards requiring ultra-pure ingredients for patient safety and product efficacy.
Question 2: Which region dominates the high purity citric acid market currently?
Answer: Asia Pacific leads the high purity citric acid market with over 39% share, driven by concentrated pharmaceutical manufacturing in China, India, and Japan. Leading producers including COFCO Biochemical, Weifang Ensign Industry, and Jungbunzlauer maintain dominant regional positions serving domestic and export markets.
Question 3: What are the primary drivers of high purity citric acid market growth?
Answer: Key growth drivers include pharmaceutical industry expansion requiring ultra-pure excipients, biotechnology sector development demanding specialized cell culture reagents, regulatory standards tightening for ingredient quality, and nutraceutical market growth emphasizing natural clean-label products. These factors create sustained demand across high-value applications.
Question 4: Which purity grade holds the largest high purity citric acid market share?
Answer: Pharmaceutical grade citric acid representing ≥99.5% purity dominates with approximately 61% market share due to extensive use in injectable drug formulations, effervescent tablets, and liquid medications. Patient safety requirements mandate minimal trace metal content and comprehensive regulatory documentation supporting this segment's leadership position.
Question 5: How does biotechnology sector growth impact the high purity citric acid market?
Answer: Biotechnology applications drive fastest segment growth at 7.2% CAGR through 2033, requiring ultra-pure laboratory-grade citric acid for cell culture media, gene therapy vector production, and analytical testing. Biopharmaceutical manufacturing expansion and advanced therapy development create premium-priced opportunities where quality assurance justifies substantial cost premiums.
