Fallopian Tube Cancer Therapeutics Market Overview
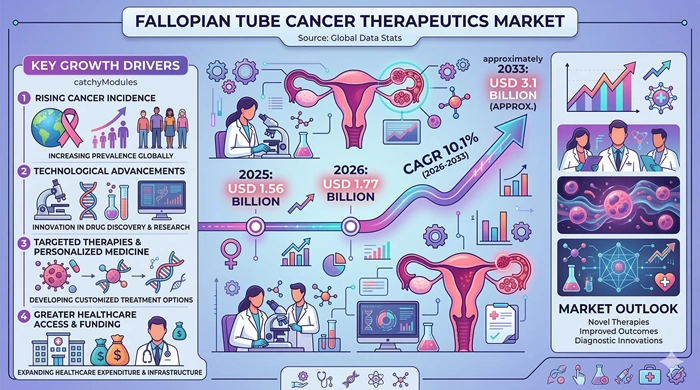
The global fallopian tube cancer therapeutics market size is valued at USD 1.56 billion in 2025 and is predicted to increase from USD 1.77 billion in 2026 to approximately USD 3.1 billion by 2033, growing at a CAGR of 10.1% from 2026 to 2033.
Fallopian tube cancer is a rare but clinically significant gynecologic malignancy that shares strong biological and therapeutic overlap with high-grade serous ovarian cancer. Long underdiagnosed due to its non-specific symptom profile and the historical tendency to misclassify it as ovarian cancer, the condition is now gaining greater clinical and scientific recognition. As molecular diagnostics improve, targeted therapy pipelines mature, and PARP inhibitors demonstrate compelling efficacy across gynecologic oncology indications, the fallopian tube cancer therapeutics market is advancing into a new phase of clinical innovation and commercial growth.
AI Impact on the Fallopian Tube Cancer Therapeutics Industry
Artificial Intelligence Is Accelerating Early Detection, Biomarker-Driven Treatment Selection, and Clinical Trial Efficiency Across the Fallopian Tube Cancer Therapeutics Landscape, Redefining Precision Gynecologic Oncology
Artificial intelligence is beginning to meaningfully transform how fallopian tube cancer is diagnosed and managed therapeutically. AI-powered genomic profiling platforms are enabling oncologists to identify actionable molecular alterations — including BRCA1/2 mutations and homologous recombination deficiency — in individual patient tumors with greater speed and accuracy than conventional testing approaches. This capability is directly informing therapy selection decisions, particularly for PARP inhibitors and other targeted agents whose efficacy is closely tied to specific molecular biomarkers. By reducing the time between diagnosis and optimal therapy initiation, AI is helping improve clinical outcomes for patients with this rare and often late-stage malignancy.
Beyond diagnostic support, AI is enhancing the efficiency of clinical trial recruitment and design for fallopian tube cancer therapeutics. Given the rarity of this cancer, identifying eligible patients for clinical trials has historically been a major logistical challenge that slows the development of new therapies. AI-driven patient matching platforms can rapidly screen large healthcare databases to identify individuals meeting specific eligibility criteria, dramatically accelerating enrollment timelines. Simultaneously, machine learning models are being applied to historical treatment outcome data to predict response patterns, informing the design of next-generation trial protocols and helping pharmaceutical companies allocate R&D resources more efficiently within this niche but growing market.
Growth Factors
Growing PARP Inhibitor Adoption, Improving Molecular Diagnostics, and Expanding Gynecologic Oncology Infrastructure Are the Primary Drivers Propelling the Fallopian Tube Cancer Therapeutics Market Forward
The most transformative growth driver in the fallopian tube cancer therapeutics market is the expanding clinical use of PARP inhibitors — a class of targeted agents that has fundamentally changed outcomes in BRCA-mutated gynecologic cancers. Drugs such as olaparib, niraparib, and rucaparib, originally developed for ovarian cancer, are now widely applied to fallopian tube cancer patients given the diseases' shared molecular biology. Their incorporation into first-line maintenance therapy following platinum-based chemotherapy has extended progression-free survival significantly, creating sustained demand for these high-value agents and substantially lifting per-patient treatment revenue across the therapeutics market.
Improved molecular diagnostic capabilities are simultaneously expanding the diagnosed and treatable patient population for fallopian tube cancer. Next-generation sequencing panels and liquid biopsy technologies are enabling more accurate and timely identification of BRCA mutations and other targetable alterations in gynecologic cancer patients, ensuring that eligible individuals receive appropriate targeted therapy rather than empiric chemotherapy alone. Growing physician awareness of fallopian tube cancer as a distinct clinical entity — rather than a subset of ovarian cancer — is also driving more precise staging, reporting, and treatment planning, which translates directly into more patients entering therapy pathways that generate market revenue.

Market Outlook
The Fallopian Tube Cancer Therapeutics Market Is Expected to Maintain Strong Growth Momentum Through 2033, Driven by Clinical Pipeline Maturation, Combination Therapy Innovation, and Expanding Patient Access in Key Regions
The long-term outlook for the fallopian tube cancer therapeutics market is strongly positive, supported by a maturing clinical pipeline that includes novel PARP inhibitor combinations, antibody-drug conjugates, and immune checkpoint inhibitor regimens in active development. Several late-stage clinical trials are evaluating combination approaches that pair PARP inhibitors with anti-angiogenic agents such as bevacizumab or checkpoint inhibitors, with early data suggesting meaningful improvements in response depth and durability. As these combinations receive regulatory approval over the forecast period, they are expected to drive significant premium pricing and increase per-patient annual drug expenditure, lifting overall market revenue substantially.
Geographic expansion of access to advanced gynecologic oncology therapeutics in emerging markets represents another important contributor to long-term market growth. While North America and Europe currently account for the large majority of fallopian tube cancer therapeutics revenue, growing investment in oncology infrastructure in Asia Pacific — particularly in China, Japan, and South Korea — is expanding the number of patients diagnosed and treated with guideline-recommended therapy. As biosimilars of key targeted agents enter the market and improve affordability in price-sensitive regions, the global addressable market for fallopian tube cancer therapeutics is expected to broaden meaningfully through 2033.
Expert Speaks
-
AstraZeneca CEO Pascal Soriot stated that gynecologic oncology remains one of the most scientifically exciting and commercially significant areas in the company's oncology portfolio, and that the continued expansion of PARP inhibitor indications — including rare gynecologic cancers with shared molecular pathology — is a direct reflection of AstraZeneca's commitment to reaching every patient who could benefit from precision cancer medicine.
-
Pfizer CEO Albert Bourla emphasized that the convergence of molecular diagnostics and targeted oncology therapy is creating unprecedented opportunities to improve outcomes in rare gynecologic cancers, and that Pfizer's investment in antibody-drug conjugates and combination immunotherapy regimens is aimed at delivering the next generation of treatment advances for patients with limited options.
-
Johnson & Johnson CEO Joaquin Duato noted that rare gynecologic malignancies including fallopian tube cancer represent a meaningful opportunity for pharmaceutical innovation, stating that J&J's oncology pipeline is focused on delivering therapies that address the specific molecular drivers of these cancers and that improving clinical outcomes in underserved patient populations remains a core part of the company's mission.
Key Report Takeaways
-
North America leads the fallopian tube cancer therapeutics market, holding the largest revenue share globally, driven by the highest rates of molecular testing, strong reimbursement frameworks for PARP inhibitors, and a dense network of National Cancer Institute-designated gynecologic oncology centers.
-
Asia Pacific is the fastest-growing region, driven by a large and rapidly growing cancer patient population in China and India, increasing adoption of PARP inhibitors and targeted therapies, enhanced diagnostic infrastructure, and expanding reimbursement policies for oncology drugs.
-
Hospitals and specialized cancer centers are the dominant end users, managing virtually all advanced fallopian tube cancer cases due to the complexity of treatment protocols, the need for molecular testing, and the requirement for specialist gynecologic oncologist oversight.
-
Targeted therapy — particularly PARP inhibitors — contributes the largest share of market revenue, reflecting the high clinical adoption of olaparib and niraparib as maintenance therapies following platinum-based chemotherapy in BRCA-mutated patients.
-
Platinum-based chemotherapy remains the most widely used initial treatment modality for fallopian tube cancer, serving as the foundational first-line standard of care before maintenance targeted therapy is initiated in eligible patients.
-
Immunotherapy is the fastest-growing therapy type segment, expected to register a CAGR above the market average through 2033 with approximately 16% market share, as checkpoint inhibitor combinations with PARP inhibitors and chemotherapy show increasing clinical promise in ongoing Phase II and Phase III trials across gynecologic oncology.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 3.1 Billion |
| Market Size by 2025 | USD 1.56 Billion |
| Market Size by 2026 | USD 1.77 Billion |
| Market Growth Rate (2026–2033) | CAGR of 10.1% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2033 |
| Segments Covered | Therapy Type, Drug Type, Distribution Channel, End User |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
PARP Inhibitor Commercial Expansion, Rising BRCA Testing Rates, and Growing Gynecologic Oncology Specialist Networks Are the Core Forces Driving Revenue Growth in the Fallopian Tube Cancer Therapeutics Market
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Expanding PARP inhibitor adoption in maintenance therapy | ~33% | North America, Europe | Near to Long-term |
| Rising rates of BRCA and molecular diagnostic testing | ~26% | Global | Near to Mid-term |
| Growing pipeline of combination therapy regimens | ~21% | North America, Europe | Mid to Long-term |
| Increasing gynecologic oncology specialist access | ~13% | Global | Mid-term |
| Favorable orphan drug and rare cancer regulatory incentives | ~7% | North America, Europe | Long-term |
The commercial expansion of PARP inhibitors across gynecologic oncology indications is the single most important driver of the fallopian tube cancer therapeutics market. Following demonstrated efficacy in BRCA-mutated ovarian cancer, olaparib, niraparib, and rucaparib are now routinely used in fallopian tube cancer patients given the near-identical molecular biology of these diseases. Their use as first-line maintenance therapy following response to platinum-based chemotherapy has become standard practice in major oncology centers, generating consistent and substantial prescription revenue that anchors the overall therapeutics market. The ongoing clinical investigation of these agents in earlier treatment lines and combination settings is expected to further expand their use and associated market revenue through 2033.
Molecular diagnostic advancement is compounding this driver by identifying more patients eligible for targeted therapy. The increasing availability and clinical adoption of comprehensive genomic profiling panels and BRCA germline testing programs — including population-level genetic screening initiatives in some countries — means that a growing proportion of fallopian tube cancer patients are identified as molecular therapy candidates at the time of diagnosis rather than retrospectively. This early identification directly increases the proportion of patients who initiate PARP inhibitor therapy, driving both prescription volumes and overall market value growth in a consistent and predictable manner.

Restraints Impact Analysis
Extreme Disease Rarity, Misclassification in Cancer Registries, and High Targeted Therapy Costs in Emerging Markets Remain the Most Significant Barriers to Market Expansion
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Extremely low disease incidence limiting patient volumes | ~40% | Global | Ongoing |
| Historical misclassification as ovarian cancer | ~28% | Global | Near to Mid-term |
| High cost of PARP inhibitors and limited reimbursement | ~20% | Emerging Markets | Near-term |
| Limited awareness among general gynecologists | ~12% | Global, especially Emerging Markets | Ongoing |
The rarity of fallopian tube cancer as a distinct diagnosed entity is the most fundamental structural constraint on market size. Because the condition has historically been classified alongside ovarian and peritoneal cancers in a combined reporting category, true incidence data is difficult to quantify, and the recognized patient population remains small. Even with improving diagnostic specificity, the absolute number of new cases diagnosed annually worldwide is limited, capping the maximum achievable prescription volume for any therapeutic agent in this specific indication and constraining the overall commercial scale of the fallopian tube cancer therapeutics market relative to more common gynecologic cancers.
The high cost of PARP inhibitors and other targeted agents presents a significant access barrier in markets outside North America and Western Europe. Annual treatment costs for approved PARP inhibitors can reach tens of thousands of dollars, and many national healthcare systems in Asia Pacific, Latin America, and the Middle East have yet to establish reimbursement coverage for these agents in gynecologic cancer indications. This pricing barrier forces a significant proportion of potentially eligible patients in emerging markets to fall back on conventional chemotherapy-only treatment protocols, reducing both clinical outcomes and market revenue generation in these geographies.
Opportunities Impact Analysis
Antibody-Drug Conjugates, Combination Immunotherapy Regimens, and Biosimilar Entry in Emerging Markets Present the Most Compelling Near and Long-Term Opportunities in This Market
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Antibody-drug conjugate development for gynecologic cancers | ~34% | North America, Europe | Mid to Long-term |
| PARP inhibitor and checkpoint inhibitor combinations | ~28% | Global | Mid to Long-term |
| Biosimilar PARP inhibitor entry in emerging markets | ~23% | Asia Pacific, Latin America | Mid-term |
| Expanding molecular testing programs globally | ~15% | Global | Near to Mid-term |
Antibody-drug conjugates represent one of the most exciting therapeutic frontiers in gynecologic oncology and a significant commercial opportunity for the fallopian tube cancer therapeutics market. Agents such as mirvetuximab soravtansine — targeting folate receptor alpha, which is highly expressed in a significant proportion of gynecologic cancers — are demonstrating compelling efficacy in platinum-resistant settings and are being explored in combination with other targeted agents. As additional ADC candidates enter clinical development with relevance to fallopian tube cancer's molecular profile, this emerging drug class is expected to create a new premium treatment tier that meaningfully lifts market revenue in the mid to long term.
The potential entry of biosimilar versions of key PARP inhibitors and other branded targeted agents offers a compelling opportunity to expand market access in price-sensitive emerging markets. As intellectual property protection for first-generation PARP inhibitors begins to expire and biosimilar manufacturers — particularly in India and China — develop lower-cost alternatives, the cost barrier for targeted therapy in gynecologic cancers including fallopian tube cancer is expected to decline significantly. This affordability improvement is anticipated to bring a meaningful volume of previously untreated or undertreated patients in Asia Pacific and Latin America into therapeutic pathways, contributing to incremental market growth while expanding the overall global treated patient population.

Segment Analysis
By Therapy Type
Targeted Therapy Leads the Fallopian Tube Cancer Therapeutics Market Revenue, Driven by the Strong Clinical and Commercial Success of PARP Inhibitors in BRCA-Mutated Gynecologic Oncology Patients
Targeted therapy accounts for approximately 44% of the total fallopian tube cancer therapeutics market revenue and represents the highest-value and fastest-growing therapy type segment through 2033. PARP inhibitors — particularly olaparib marketed by AstraZeneca and niraparib by GlaxoSmithKline — dominate this category due to their established efficacy as first-line maintenance therapy in patients with BRCA mutations or homologous recombination deficiency. This segment is expected to grow at a CAGR of approximately 11.2% through 2033, driven by expanding label indications, combination therapy development, and increasing molecular testing identifying more eligible patients. North America leads targeted therapy adoption within the fallopian tube cancer therapeutics market, supported by comprehensive genomic profiling programs in major cancer centers and robust insurance reimbursement for PARP inhibitors in gynecologic oncology indications. AstraZeneca, GlaxoSmithKline, and Clovis Oncology are the key commercial players generating the majority of targeted therapy revenue in this region.
Europe is the second-largest and fast-growing geography for targeted therapy within the fallopian tube cancer therapeutics space, driven by EMA approvals, national health technology assessments incorporating PARP inhibitors into formularies, and active clinical research through the European Network of Gynaecological Oncological Trial groups. Germany, France, the United Kingdom, and the Netherlands are leading European markets where gynecologic oncology centers routinely use molecular profiling to guide therapy selection and are actively participating in trials evaluating next-generation targeted combinations. Regional academic institutions are generating real-world evidence supporting the clinical value of PARP inhibitors in fallopian tube cancer patients, strengthening the reimbursement case with payers and driving broader adoption of targeted therapy outside the largest academic centers.
By End User
Hospitals and Specialized Cancer Centers Dominate the Fallopian Tube Cancer Therapeutics Market End-User Landscape, Concentrating the Vast Majority of Diagnosed Cases and Advanced Treatment Delivery
Hospitals and specialized cancer centers account for approximately 68% of the total fallopian tube cancer therapeutics market revenue across all end-user categories. Given the complexity and rarity of this malignancy, virtually all diagnosed cases are managed within tertiary care or specialized gynecologic oncology settings equipped with molecular diagnostic laboratories, multidisciplinary tumor boards, and access to clinical trial protocols. This segment is expected to maintain a steady CAGR of approximately 9.6% through 2033, supported by expanding investment in gynecologic oncology infrastructure and the growing concentration of advanced cancer care within comprehensive cancer center networks. National Cancer Institute-designated cancer centers in the United States, and their European equivalents within ESMO-accredited oncology centers, represent the primary treatment hubs generating the majority of therapeutics revenue in this segment. Leading pharmaceutical companies including AstraZeneca and GlaxoSmithKline maintain dedicated oncology sales forces targeting these high-value institutional accounts.
Academic and research institutes represent the fastest-growing end-user segment within the fallopian tube cancer therapeutics market, fueled by increasing clinical trial activity and the growing role of academic medical centers in generating the evidence base for new therapeutic approaches in rare gynecologic cancers. These institutions are actively enrolling patients in trials evaluating PARP inhibitor combinations with immunotherapy, ADCs, and novel targeted agents, creating a clinical research ecosystem that both informs future treatment practice and generates near-term therapeutics consumption within the trial setting. Asia Pacific academic centers — particularly in Japan, South Korea, and China — are increasingly participating in multinational gynecologic oncology trials, expanding regional contributions to the overall therapeutics market and building local clinical expertise that will support commercial adoption of advanced therapies in these markets over the forecast period.

Regional Insights
North America
North America Dominates the Fallopian Tube Cancer Therapeutics Market, Anchored by Comprehensive Molecular Testing Programs, Strong PARP Inhibitor Reimbursement, and the World's Most Concentrated Network of Gynecologic Oncology Specialists
North America holds approximately 43% of the global fallopian tube cancer therapeutics market share and is expected to maintain its leading position through 2033, growing at a CAGR of approximately 9.7%. The United States drives the vast majority of regional revenue, underpinned by mandatory BRCA testing recommendations for all epithelial ovarian, fallopian tube, and primary peritoneal cancer patients — a practice guideline that has substantially increased identification of molecularly eligible patients for PARP inhibitor therapy. The strong commercial infrastructure of major oncology pharmaceutical companies, combined with broad private and public insurance reimbursement for targeted gynecologic cancer therapies, creates a favorable environment for high per-patient treatment revenue. Key companies with dominant North American commercial presence include AstraZeneca (U.K./U.S.), GlaxoSmithKline (U.K.), and Pfizer (U.S.).
Canada contributes meaningfully to North America's regional leadership through its publicly funded provincial cancer programs that have progressively incorporated PARP inhibitors into their formularies for eligible gynecologic cancer patients. Canadian academic cancer centers are active participants in international gynecologic oncology clinical trials, and the increasing standardization of molecular testing within Canadian oncology programs is expanding the identification of fallopian tube cancer patients eligible for targeted therapy. Together, the United States and Canada form a mature, well-supported regional market that will continue to set the clinical and commercial benchmark for the global fallopian tube cancer therapeutics industry through 2033.
Asia Pacific
Asia Pacific Is the Fastest-Growing Region in the Fallopian Tube Cancer Therapeutics Market, Driven by a Large and Growing Cancer Patient Population, Increasing PARP Inhibitor Adoption, and Rapidly Expanding Oncology Infrastructure
Asia Pacific is the fastest-growing region in the fallopian tube cancer therapeutics market, expected to register a CAGR of approximately 12.4% through 2033, significantly exceeding the global market average. China is the primary growth engine, with a rapidly expanding gynecologic oncology specialist workforce, significant government investment in cancer care infrastructure, and an increasingly sophisticated biopharmaceutical sector introducing both innovative and biosimilar oncology products. Japan maintains a long-established and advanced oncology care system, with high molecular testing rates and growing adoption of international gynecologic oncology treatment guidelines driving PARP inhibitor utilization in eligible patients. Key companies active in the Asia Pacific region include Takeda Pharmaceutical (Japan), Eisai Co. Ltd. (Japan), and global players including AstraZeneca and Roche that have established strong regional oncology commercial operations.
India and South Korea represent compelling growth opportunities within the Asia Pacific fallopian tube cancer therapeutics market. South Korea's highly advanced medical infrastructure, high healthcare expenditure per capita, and active clinical trial participation are supporting rapid adoption of targeted gynecologic cancer therapies. India, while at an earlier stage of market development, is investing meaningfully in oncology infrastructure expansion and is home to a large and growing population of gynecologic cancer patients who are beginning to access PARP inhibitor therapy through national cancer programs and private hospital networks. The combination of large patient volumes, expanding diagnostic capabilities, and improving drug reimbursement frameworks makes Asia Pacific the most strategically important long-term growth region for the fallopian tube cancer therapeutics market globally.
Top Key Players
-
AstraZeneca plc (United Kingdom)
-
GlaxoSmithKline plc (United Kingdom)
-
Pfizer Inc. (United States)
-
Roche Holding AG (Switzerland)
-
Bristol Myers Squibb Company (United States)
-
Merck & Co. Inc. (United States)
-
Clovis Oncology Inc. (United States)
-
Novartis AG (Switzerland)
-
Johnson & Johnson (United States)
-
Eisai Co. Ltd. (Japan)
-
Immunomedics (Gilead Sciences) (United States)
-
Regeneron Pharmaceuticals Inc. (United States)
Recent Developments
-
AstraZeneca (2025): AstraZeneca announced updated long-term overall survival data from the SOLO-1 trial evaluating olaparib maintenance therapy in BRCA-mutated advanced gynecologic cancer patients including fallopian tube cancer, confirming a sustained and clinically meaningful survival benefit that is expected to further strengthen the drug's positioning as the preferred first-line maintenance therapy in eligible patients globally.
-
GlaxoSmithKline (2024): GlaxoSmithKline reported positive results from the PRIME trial evaluating niraparib plus dostarlimab combination therapy in newly diagnosed advanced ovarian and fallopian tube cancer patients, demonstrating improved progression-free survival in the all-comers population and supporting a potential supplemental regulatory submission for this combination regimen.
-
Merck & Co. (2024–2025): Merck & Co. continued to advance pembrolizumab-based combination regimens in gynecologic oncology, with ongoing Phase III trials evaluating pembrolizumab in combination with chemotherapy and PARP inhibitors in first-line advanced fallopian tube and ovarian cancer settings, reinforcing its commitment to expanding checkpoint inhibitor utility across gynecologic malignancies.
-
Roche (2024): Roche reported positive data supporting the continued use of bevacizumab in combination with chemotherapy and PARP inhibitor maintenance in gynecologic cancer patients with homologous recombination deficiency, strengthening the clinical rationale for triplet combination regimens that are expected to become increasingly important in advanced fallopian tube cancer management over the coming years.
-
Eisai Co. Ltd. (2025): Eisai advanced its pipeline antibody-drug conjugate program targeting folate receptor alpha into expanded clinical evaluation in gynecologic cancers including fallopian tube cancer, with Phase II data demonstrating promising response rates in platinum-resistant patients, positioning the company as an emerging competitor in the premium targeted therapy segment of this market.
Market Trends
Combination Therapy Innovation, Antibody-Drug Conjugate Entry, and the Shift Toward Biomarker-Stratified Treatment Protocols Are the Defining Trends Reshaping the Fallopian Tube Cancer Therapeutics Market Through 2033
The most clinically consequential trend in the fallopian tube cancer therapeutics market is the rapid advancement of combination therapy regimens pairing PARP inhibitors with checkpoint inhibitors or anti-angiogenic agents. Early clinical evidence from multiple ongoing trials suggests that combining PARP inhibitor-mediated DNA damage with immune checkpoint blockade can generate synergistic antitumor activity, potentially converting immunologically "cold" gynecologic tumors into more immunologically responsive targets. This combination approach is being actively pursued by AstraZeneca, GlaxoSmithKline, Merck, and Bristol Myers Squibb, with multiple regulatory submissions anticipated over the 2026–2033 forecast period, each representing a meaningful premium pricing opportunity that will lift average revenue per treated patient in this market.
Concurrently, the field is witnessing growing momentum around biomarker-stratified treatment decision-making that goes beyond BRCA mutation status alone. Homologous recombination deficiency scoring, tumor mutational burden, and folate receptor alpha expression are emerging as clinically relevant biomarkers that can identify optimal candidates for specific therapeutic approaches, including antibody-drug conjugates. As companion diagnostic tools for these biomarkers gain regulatory approval and clinical validation, the fallopian tube cancer therapeutics market is expected to evolve toward increasingly precise, multi-biomarker-guided treatment algorithms that improve patient outcomes while creating differentiated commercial opportunities for pharmaceutical companies with molecularly targeted pipeline assets.
Segments Covered in the Report
By Therapy Type
-
Chemotherapy
-
Platinum-Based Regimens
-
Taxane-Based Regimens
-
Others
-
Targeted Therapy
-
PARP Inhibitors
-
Bevacizumab
-
Others
-
Immunotherapy
-
Checkpoint Inhibitors
-
Others
-
Hormonal Therapy
-
Surgery-Assisted Therapeutics
-
Others
By Drug Type
-
Branded Drugs
-
Generic Drugs
By Distribution Channel
-
Hospital Pharmacies
-
Retail Pharmacies
-
Online Pharmacies
By End User
-
Hospitals & Cancer Centers
-
Specialty Gynecologic Oncology Clinics
-
Academic & Research Institutes
-
Others
By Region
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the global fallopian tube cancer therapeutics market and what is the projected value by 2033?
Answer: The global fallopian tube cancer therapeutics market is valued at USD 1.56 billion in 2025 and is projected to reach USD 3.1 billion by 2033. It is expected to grow at a CAGR of 10.1% from 2026 to 2033.
Question 2: What are the primary drivers of growth in the fallopian tube cancer therapeutics market?
Answer: The key growth drivers include the expanding clinical adoption of PARP inhibitors as maintenance therapy in BRCA-mutated patients and the rising rates of molecular diagnostic testing that identify more therapy-eligible patients. Growing investment in gynecologic oncology infrastructure and a maturing clinical pipeline of combination regimens are also fueling sustained market growth.
Question 3: Which therapy type dominates the fallopian tube cancer therapeutics market?
Answer: Targeted therapy — particularly PARP inhibitors — holds the largest revenue share in the fallopian tube cancer therapeutics market at approximately 44% of total revenue. Agents such as olaparib and niraparib have become the standard of care for maintenance therapy in eligible patients following platinum-based chemotherapy.
Question 4: Which region leads the fallopian tube cancer therapeutics market and which is growing the fastest?
Answer: North America leads the fallopian tube cancer therapeutics market with approximately 43% of global revenue share, driven by high BRCA testing rates and strong reimbursement frameworks. Asia Pacific is the fastest-growing region, projected to register a CAGR of approximately 12.4% through 2033, supported by a large and rapidly growing cancer patient population and expanding oncology infrastructure.
Question 5: How is the fallopian tube cancer therapeutics market expected to evolve with combination therapy development?
Answer: The fallopian tube cancer therapeutics market is expected to shift significantly toward combination regimens pairing PARP inhibitors with checkpoint inhibitors and anti-angiogenic agents, which are demonstrating synergistic clinical activity in ongoing trials. These premium combination therapies are anticipated to drive meaningful increases in per-patient annual treatment costs and lift overall market revenue substantially through 2033.
