Endoscopy Devices Market Overview
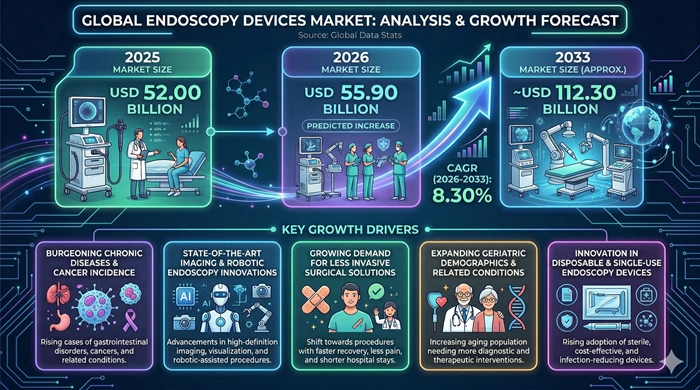
The global Endoscopy Devices market size is valued at USD 52.00 billion in 2025 and is predicted to increase from USD 55.90 billion in 2026 to approximately USD 112.30 billion by 2033, growing at a CAGR of 8.30% from 2026 to 2033.
Endoscopy devices represent a broad and strategically vital category of minimally invasive medical instruments and systems — encompassing endoscopes, visualization systems, operative accessories, fluid management platforms, and imaging enhancement tools — that are collectively used by clinicians to visually examine, diagnose, and treat conditions within the body's internal structures through natural body openings or small surgical incisions without requiring large open surgical access. These instruments have become indispensable across virtually every major clinical specialty — including gastroenterology, pulmonology, urology, orthopedics, gynecology, and otolaryngology — and the Endoscopy Devices market is experiencing strong and sustained growth driven by the globally rising prevalence of gastrointestinal cancers and chronic diseases, the accelerating adoption of minimally invasive surgical approaches, rapid technological innovation in AI-assisted imaging and single-use disposable devices, and expanding endoscopic screening program investments worldwide.
AI Impact on the Endoscopy Devices Industry
Artificial Intelligence Is Transforming Real-Time Polyp Detection, Automated Lesion Characterization, Robotic Endoscope Navigation, and Intelligent Image Enhancement in Ways That Are Meaningfully Improving Diagnostic Accuracy and Procedural Safety Across All Major Endoscopy Device Application Categories*
Artificial intelligence is delivering its most commercially impactful contribution to the Endoscopy Devices market through the integration of deep learning computer vision systems directly into colonoscopy and upper gastrointestinal endoscope platforms — where AI-powered computer-aided detection algorithms analyze real-time video streams from high-definition endoscope cameras to identify polyps, adenomas, and early-stage malignant lesions with sensitivity levels meeting or exceeding experienced endoscopist performance. Multiple commercially cleared AI colonoscopy detection systems — including Medtronic's GI Genius, Fujifilm's CAD EYE, and Olympus' EndoBRAIN — have now accumulated substantial real-world deployment evidence demonstrating statistically significant improvements in adenoma detection rates compared to standard colonoscopy practice, building the clinical quality imperative that is accelerating gastroenterology program adoption of AI-assisted colonoscopy as the evolving standard of care. These AI polyp detection platforms represent a new and fast-growing premium technology layer within the endoscopy devices ecosystem — generating ongoing software subscription revenue streams alongside traditional endoscope hardware and accessory sales — and their commercial adoption is progressively expanding from leading academic medical centers into community gastroenterology practices and hospital endoscopy programs globally.
AI is simultaneously advancing robotic endoscopy guidance applications — where computer vision and real-time anatomical mapping algorithms are integrated into bronchoscope and flexible robotic endoscope platforms to improve biopsy targeting accuracy for peripheral lung lesions that are difficult to access with conventional manual bronchoscopy. Robotic flexible endoscopy systems including Intuitive Surgical's Ion and Johnson & Johnson's Monarch platforms combine advanced AI navigation software with precision-engineered robotic bronchoscope hardware to achieve superior peripheral lung lesion biopsy success rates — creating a transformative new early-stage lung cancer diagnostic pathway that is generating strong clinical adoption momentum in thoracic medicine programs across North America and Europe. The progressive integration of AI diagnostic intelligence and robotic procedural precision across the Endoscopy Devices market is creating important technology differentiation dynamics that are raising innovation investment requirements, accelerating product upgrade cycles, and driving consolidation around vendors with the most comprehensive and clinically validated AI endoscopy capability portfolios.
Growth Factors
Rising Global GI Cancer Burden and Screening Program Expansion, Accelerating Minimally Invasive Surgery Adoption, Single-Use Endoscopy Device Innovation, and Growing Healthcare Infrastructure Investment in Emerging Economies Are the Foundational Growth Drivers*
The most structurally powerful and commercially durable growth driver of the Endoscopy Devices market is the continuously rising global burden of gastrointestinal cancers and chronic diseases — including colorectal cancer, gastric cancer, esophageal cancer, inflammatory bowel disease, peptic ulcers, and chronic liver disease — that collectively generate enormous and growing procedural demand for endoscopic diagnosis, surveillance, and therapeutic intervention worldwide. Colorectal cancer remains the second most commonly diagnosed cancer globally, and the progressive expansion of organized endoscopic screening programs — most prominently colonoscopy-based colorectal cancer screening in North America, Europe, Australia, Japan, and South Korea — is systematically translating rising disease awareness and clinical guideline support into high-volume, recurring endoscopy device utilization demand that underpins confident long-term revenue growth projections for endoscopy equipment and consumable manufacturers. The concurrent expansion of gastric cancer endoscopic screening programs across high-incidence East Asian populations — combined with the growing global adoption of upper gastrointestinal endoscopy for Barrett's esophagus surveillance and GERD evaluation — is generating additional procedural volume streams that further reinforce the broad and structurally durable demand foundation supporting endoscopy devices market growth throughout the forecast period.
The global surgical community's decisive and accelerating transition toward minimally invasive laparoscopic, arthroscopic, and endoscopic surgical approaches — driven by compelling clinical evidence of superior patient outcomes, reduced postoperative pain, shorter hospital stays, and faster recovery compared to traditional open surgery — is simultaneously generating strong and growing capital equipment demand for premium endoscopy visualization systems, operative endoscopy accessories, and fluid management platforms across hospital operating rooms and ambulatory surgical centers worldwide. Continuous technological innovation across the endoscopy devices category — including the commercialization of 4K ultra-high-definition and 3D stereoscopic visualization systems, fluorescence imaging platforms for intraoperative tissue perfusion assessment, narrow band imaging for improved lesion characterization, and progressively more capable single-use disposable endoscope systems — is creating compelling product upgrade cycle dynamics that drive recurring capital equipment replacement investment from healthcare organizations seeking to maintain clinical technology currency. The rapid expansion of ambulatory surgical center infrastructure globally — where endoscopic procedures including colonoscopy, upper endoscopy, laparoscopy, and arthroscopy are progressively shifting from hospital inpatient to outpatient settings — is simultaneously expanding the addressable capital equipment and consumable customer base for endoscopy devices manufacturers.

Market Outlook
With AI-Powered Diagnostic Tools Achieving Mainstream Adoption, Disposable Endoscopy Platform Investments Accelerating, Robotic Endoscopy Systems Expanding Clinically, and Emerging Market Healthcare Infrastructure Growing Rapidly, the Endoscopy Devices Market Outlook Is Exceptionally Strong Through 2033*
The long-term outlook for the Endoscopy Devices market is firmly positive and supported by a compelling combination of clinical, technological, regulatory, and demographic growth catalysts expected to sustain robust revenue expansion throughout the 2026–2033 forecast period. The most commercially transformative structural shift currently reshaping the endoscopy device product landscape is the accelerating adoption of single-use disposable endoscopes — driven by compelling infection control and patient safety advantages over reusable endoscopes following high-profile duodenoscope-associated infection outbreaks and supportive FDA regulatory guidance that have collectively created strong institutional momentum for disposable endoscopy device adoption across bronchoscopy, duodenoscopy, urological endoscopy, and increasingly flexible gastrointestinal endoscopy categories. As leading manufacturers including Ambu, Boston Scientific, and Olympus scale their single-use endoscopy product portfolios and manufacturing capacity — progressively reducing per-unit product costs through manufacturing scale economies — the total cost of care economics of disposable versus reusable endoscopy approaches are becoming more favorable in an expanding range of clinical settings, accelerating the commercial adoption trajectory of single-use platforms throughout the forecast period.
Emerging market healthcare infrastructure development represents one of the most commercially exciting long-term growth opportunities within the Endoscopy Devices market — with rapidly growing middle-class populations in China, India, Southeast Asia, Latin America, and Africa combining rising healthcare expenditure, increasing gastrointestinal disease awareness, and government hospital quality improvement investment to create large cohorts of first-time endoscopy program adopters entering the market during the forecast period. The substantial unmet clinical need for endoscopic cancer screening and gastrointestinal disease diagnosis in these large population markets — combined with the improving accessibility of cost-competitive endoscopy equipment from domestic manufacturers and the growing clinical training programs developing endoscopist workforce capacity in emerging economies — is enabling healthcare organizations in these regions to establish meaningful endoscopy program infrastructure for the first time. This emerging market adoption wave is expected to become progressively more significant as a global endoscopy devices market growth contributor through 2033 — adding substantial new revenue from geographies that are currently underrepresented in global endoscopy device sales but carry large and rapidly growing unmet clinical demand for endoscopic diagnosis and treatment.
Expert Speaks
-
Gary Guthart, CEO, Intuitive Surgical — "The convergence of robotic systems, AI-guided navigation, and advanced imaging within endoscopy and minimally invasive surgery platforms is creating a new era of procedural precision and diagnostic accuracy that is transforming patient outcomes across oncological, gastrointestinal, and thoracic medicine applications. We believe the integration of intelligent software and flexible robotic endoscopy systems represents one of the most commercially significant and clinically meaningful technology development trajectories in the endoscopy devices industry over the coming decade, and we are investing aggressively to lead this transformation."
-
Michael Mahoney, CEO, Boston Scientific — "Endoscopy and minimally invasive devices remain among the most clinically important and commercially dynamic segments of Boston Scientific's global portfolio — with continuous innovation in therapeutic endoscopy tools, advanced imaging platforms, single-use flexible endoscope technology, and AI-assisted diagnostic systems creating compelling new growth opportunities across gastrointestinal, pulmonary, and urological procedural applications. The global expansion of endoscopic screening programs and growing clinical adoption of endoscopic interventional therapies as alternatives to open surgery are creating a sustainably growing endoscopy market that we are actively investing in to capture."
-
Curt Hartman, CEO, Stryker Corporation — "The continued global transition toward minimally invasive and endoscopic surgical approaches in orthopedics, ENT, neurosurgery, and general surgery generates strong and growing demand for high-quality visualization systems, operative endoscopy instruments, and fluid management platforms that enable surgeons to perform complex procedures with superior outcomes and smaller patient footprints. Stryker is investing in next-generation 4K and 3D imaging platforms, AI-assisted surgical guidance tools, and single-use endoscopy technologies to address the most important clinical and operational priorities of our global endoscopy device customer base."
Key Report Takeaways
-
North America dominates the Endoscopy Devices market, holding approximately 38.60% of global market share in 2025, driven by the world's highest endoscopic procedure volumes supported by the most comprehensive colorectal cancer screening program infrastructure, the highest per-capita healthcare capital equipment investment levels, the most progressive AI-assisted endoscopy regulatory clearance environment, global leadership in robotic endoscopy system commercialization, and the concentration of leading endoscopy device companies including Olympus, Boston Scientific, Medtronic, Stryker, and Intuitive Surgical with dominant North American commercial positions.
-
Asia Pacific is the fastest-growing regional market for endoscopy devices, projected to expand at a CAGR of approximately 9.80% from 2026 to 2033, driven by the world's largest populations with high gastrointestinal disease prevalence in China, India, Japan, and South Korea, rapidly expanding hospital endoscopy unit infrastructure investment, progressive government cancer screening program adoption, rising per-capita healthcare expenditure, and strong domestic endoscopy device manufacturing capability in Japan and China generating competitive premium equipment for regional health systems.
-
Endoscopes are the dominant product segment, accounting for approximately 47.20% of total market revenue in 2025, reflecting the enormous procedural volume and capital equipment value of flexible gastrointestinal endoscopes, rigid laparoscopes and arthroscopes, bronchoscopes, and urological endoscopes — which collectively form the foundational clinical instrument category that drives the majority of endoscopy device capital equipment investment and associated accessories and consumables revenue globally.
-
Gastrointestinal endoscopy is the largest application segment, contributing approximately 43.60% of total market revenue in 2025, driven by the globally rising colorectal cancer, gastric cancer, and gastrointestinal disease burden combined with expanding organized endoscopic screening program coverage that collectively generate the highest-volume clinical endoscopy procedure category and the most commercially significant endoscopy device capital equipment and consumable demand globally.
-
Hospitals represent the largest end-user segment, contributing approximately 48.40% of total revenue in 2025, driven by the most complex and highest-acuity endoscopic procedure requirements — including therapeutic ERCP, endoscopic submucosal dissection, robotic bronchoscopy, and minimally invasive oncological surgery — that demand the most comprehensive capital equipment investments in premium visualization systems, operative endoscopy platforms, and fluid management systems available in the market.
-
Ambulatory surgical centers are the fastest-growing end-user segment, expected to expand at a CAGR of approximately 10.40% through 2033, driven by the global shift of colonoscopy, upper endoscopy, laparoscopy, and arthroscopy from hospital inpatient to outpatient ambulatory settings enabled by single-use endoscopy device adoption, improving procedure efficiency, and favorable reimbursement models that make ambulatory endoscopy economically attractive for providers, payers, and patients alike.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 112.30 Billion |
| Market Size by 2025 | USD 52.00 Billion |
| Market Size by 2026 | USD 55.90 Billion |
| Market Growth Rate (2026–2033) | CAGR of 8.30% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Segments Covered | Product, Application, End User |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Rising GI Cancer Screening Volumes, Minimally Invasive Surgery Growth, Disposable Endoscopy Device Infection Safety Demand, and AI Integration Are the Four Pillars Powering Endoscopy Devices Market Growth*
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising global GI cancer burden and expanding endoscopic screening program coverage | ~34% | Global | Short to Long-term |
| Accelerating minimally invasive laparoscopic and endoscopic surgical adoption | ~28% | North America, Europe, Asia Pacific | Short to Long-term |
| Single-use disposable endoscopy device adoption driven by infection control demand | ~25% | North America, Europe | Short to Medium-term |
| AI-powered endoscopy diagnostic tool integration improving detection and outcomes | ~13% | North America, Europe, Japan | Short to Medium-term |
The most enduring and commercially significant growth driver of the Endoscopy Devices market is the continuously expanding global burden of gastrointestinal cancers and chronic diseases — generating rising procedural demand that creates a large and structurally growing base of recurring endoscopy device capital equipment and consumable revenue. The progressive implementation of colorectal cancer screening colonoscopy programs across additional national health systems and insurance coverage frameworks — combined with the ongoing lowering of recommended screening initiation age to 45 in the United States and equivalent policy shifts in other markets — is systematically growing colonoscopy procedure volumes at predictable and durable rates. Upper gastrointestinal endoscopy access for Barrett's esophagus surveillance, GERD evaluation, and upper GI cancer screening is simultaneously expanding — with reimbursement coverage improvements and growing clinical guideline support for upper GI endoscopy in additional patient populations further broadening the procedural volume base that drives endoscopy device market revenue growth.
The global surgical transition toward minimally invasive endoscopic approaches is generating equally powerful capital equipment demand dynamics within the endoscopy devices ecosystem — with the combination of compelling patient outcome evidence, growing surgical specialty training program emphasis on laparoscopic and endoscopic technique, and continuous visualization system technology innovation creating a self-reinforcing cycle of procedural volume growth and capital equipment investment that sustains the market's strong revenue expansion. The innovation-driven product upgrade cycle is an important and often underappreciated commercial growth driver within the Endoscopy Devices market — as hospitals and ambulatory surgical centers systematically refresh their endoscopy capital equipment to access the imaging quality, procedural capability, and clinical safety improvements that new-generation endoscopy visualization platforms and operative device systems offer. This recurring capital equipment replacement demand — layered on top of the procedural volume-driven consumable and accessories revenue stream — creates a robust and multi-layered commercial revenue foundation that supports consistent market growth regardless of short-term healthcare spending fluctuations.

Restraints Impact Analysis
High Capital Equipment Costs, Complex Reprocessing Requirements, Skilled Workforce Shortages, and Reimbursement Constraints Restrain Faster Global Endoscopy Devices Market Expansion*
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High capital equipment cost limiting adoption in budget-constrained healthcare settings | ~33% | Asia Pacific, Latin America, MEA | Short to Long-term |
| Complex endoscope reprocessing requirements creating operational burden and infection risk | ~27% | Global | Short to Long-term |
| Shortage of trained endoscopists limiting procedure capacity and device utilization | ~28% | Global — especially developing markets | Short to Medium-term |
| Reimbursement rate constraints reducing economic viability of endoscopic procedures | ~12% | Europe, Asia Pacific | Short to Medium-term |
The most broadly experienced commercial constraint on the Endoscopy Devices market is the high capital equipment cost of premium endoscopy systems — particularly the flagship flexible videoendoscope platforms from Olympus, Fujifilm, and Pentax Medical and the advanced visualization system towers from Karl Storz and Stryker — that represent significant capital investment requirements for hospitals and ambulatory surgical centers in cost-sensitive healthcare markets across Asia Pacific, Latin America, Africa, and the Middle East. The total capital investment required to establish a fully equipped endoscopy program — including endoscopes, visualization systems, reprocessing infrastructure, fluid management equipment, and operative accessory inventory — can represent millions of dollars in initial outlay that poses real affordability barriers for healthcare organizations in developing economies seeking to establish or expand their endoscopy program capacity. Domestic manufacturers in China and South Korea are progressively addressing this cost barrier with competitively priced endoscopy systems — but achieving the imaging performance, instrument durability, and clinical outcome equivalence of leading Japanese and U.S. manufacturer platforms at substantially lower price points remains a technical and commercial challenge.
The persistent global shortage of qualified endoscopists capable of performing high-quality diagnostic and therapeutic endoscopic procedures creates a fundamental constraint on endoscopy device utilization rates and procedure capacity expansion — particularly in developing healthcare markets where endoscopist training infrastructure is still being established and the physician-to-population ratio for gastrointestinal medicine specialists is substantially below the levels needed to serve current unmet clinical demand. Even in developed healthcare markets, the growing procedural demand driven by aging demographics and expanding screening program coverage is progressively straining endoscopist workforce capacity — with colonoscopy wait times extending in many healthcare systems and outpatient endoscopy scheduling backlogs limiting the procedural volume growth that would otherwise drive even stronger endoscopy device capital equipment and consumable revenue growth. Training program capacity expansion, simulation-based endoscopy education advancement, and the progressive development of AI-assisted and robotic endoscopy systems that reduce the skill threshold for certain endoscopic procedures are the primary avenues being pursued to address this workforce constraint.
Opportunities Impact Analysis
Single-Use Disposable Endoscopy Platform Expansion, AI Endoscopy Tool Mainstream Adoption, Emerging Market Program Build-Out, and Robotic Flexible Endoscopy Commercialization Create Substantial Endoscopy Devices Market Opportunities*
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Single-use disposable endoscopy device platform expansion across new categories | ~34% | North America, Europe | Short to Medium-term |
| AI computer-aided detection endoscopy tool mainstream clinical program adoption | ~27% | North America, Europe, Japan | Short to Medium-term |
| Emerging market first-time endoscopy program establishment and infrastructure investment | ~26% | Asia Pacific, Latin America, MEA | Medium to Long-term |
| Robotic flexible endoscopy system commercial expansion and indication broadening | ~13% | North America, Europe | Medium to Long-term |
The most immediately commercially transformative opportunity reshaping the Endoscopy Devices market is the rapid expansion of single-use disposable endoscopy platforms — driven by compelling infection control and patient safety advantages that are progressively converting hospital endoscopy programs from reusable to disposable instruments across bronchoscopy, duodenoscopy, urological endoscopy, and increasingly flexible gastrointestinal endoscopy categories. Companies including Ambu, Boston Scientific, Olympus, and Vatom Medical are investing substantially in single-use endoscopy product portfolio development — recognizing that the disposable transition represents one of the most significant and commercially valuable business model shifts in endoscopy device industry history, transforming the revenue model from cyclical capital equipment replacement cycles toward predictable, recurring per-procedure disposable product revenue streams. As manufacturing scale increases and per-unit product costs progressively decline, the total cost of care economics of disposable endoscopy are becoming favorable in an expanding range of clinical applications — accelerating the conversion of reusable endoscopy programs toward mixed or fully disposable models.
The mainstream clinical adoption of AI-powered computer-aided detection systems for colonoscopy and upper gastrointestinal endoscopy represents a major near-term commercial opportunity within the endoscopy devices ecosystem — as the growing clinical quality evidence base and progressively clearer return-on-investment case for AI endoscopy tools are translating into gastroenterology program procurement decisions at an accelerating pace across North American, European, and Japanese endoscopy markets. As AI endoscopy tool applications broaden beyond colonoscopy polyp detection into capsule endoscopy automated reading, bronchoscopy navigation guidance, and upper GI lesion characterization — the AI platform becomes a comprehensive clinical intelligence layer integrated across the full endoscopy device product stack, generating ongoing software subscription revenue and creating meaningful technology differentiation that drives premium endoscopy system adoption among healthcare organizations seeking to deliver best-in-class endoscopy diagnostic performance.

Segment Analysis
By Product
Endoscopes Lead the Endoscopy Devices Market Revenue While Visualization Systems and Single-Use Accessories Register the Fastest Investment Growth*
Endoscopes represent the dominant product segment in the Endoscopy Devices market, accounting for approximately 47.20% of total global market revenue in 2025. The category encompasses the broad range of flexible gastrointestinal videoendoscopes, bronchoscopes, urological endoscopes, rigid laparoscopes and arthroscopes, wireless capsule endoscopes, and emerging robot-assisted flexible endoscopes that collectively generate the largest share of capital equipment revenue in the endoscopy industry — reflecting both the high unit value of premium endoscope systems and the enormous procedural volume that drives recurring endoscope replacement and accessories demand. Flexible gastrointestinal endoscopes from leading manufacturers Olympus, Fujifilm, and Hoya (Pentax Medical) dominate the endoscope product segment — supported by the massive procedural volume of colonoscopy and upper endoscopy that creates strong and highly predictable recurring capital equipment and accessories revenue. North America leads global endoscope product segment revenue — driven by the highest colonoscopy and upper GI endoscopy procedure volumes, the most developed therapeutic endoscopy program infrastructure, and the strongest AI-enhanced endoscope adoption — while Asia Pacific is the fastest-growing product segment region at approximately 9.80% CAGR through 2033.
Endoscopy visualization systems — encompassing the HD, 4K ultra-high-definition, 3D stereoscopic, and fluorescence imaging tower systems that process and display endoscope camera video in surgical and endoscopy suite environments — represent the second-largest and fastest-innovating product segment in the Endoscopy Devices market, accounting for approximately 22.80% of total market revenue in 2025. The ongoing commercial transition from HD to 4K imaging systems across hospital endoscopy suites and operating rooms is generating substantial capital equipment upgrade investment cycles — as surgeons and endoscopists demand the superior image resolution, color accuracy, and anatomical detail that 4K endoscopy visualization delivers for complex therapeutic endoscopy and minimally invasive surgery applications. Karl Storz, Stryker, Smith & Nephew, and Olympus are among the leading visualization system competitors — each investing in next-generation 4K and AI-enhanced imaging platform development and offering comprehensive visualization system ecosystems that integrate their endoscope product lines with compatible tower systems, image management software, and operative accessory portfolios. Europe is the fastest-growing visualization system market — driven by strong hospital capital equipment upgrade investment and growing laparoscopic and endoscopic surgical procedure volumes across European healthcare systems prioritizing minimally invasive surgery program development.
By Application
Gastrointestinal Endoscopy Leads the Endoscopy Devices Market While Bronchoscopy Registers the Fastest Technology Investment and Procedural Growth*
Gastrointestinal endoscopy represents the dominant application segment in the Endoscopy Devices market, accounting for approximately 43.60% of total global market revenue in 2025, reflecting the overwhelming procedural volume dominance of colonoscopy, upper endoscopy, ERCP, endoscopic ultrasound, and the rapidly growing range of therapeutic endoscopic interventions that collectively generate the largest single application category of endoscopy device capital equipment and accessory demand globally. The progressive expansion of colorectal cancer screening colonoscopy programs — with both the United States and multiple European health systems recommending colonoscopy initiation at age 45 and organized screening program participation rates improving — is generating structurally growing colonoscopy procedure volume growth that sustains strong recurring demand for gastrointestinal endoscopy devices. The therapeutic endoscopy application range within gastrointestinal endoscopy is simultaneously expanding — with endoscopic mucosal resection, endoscopic submucosal dissection, POEM, endoscopic bariatric procedures, and per-oral endoscopic myotomy generating growing demand for specialized therapeutic endoscopy operative devices and accessories in academic and high-volume community hospital programs. Olympus, Fujifilm, Boston Scientific, and Medtronic are the leading companies serving the gastrointestinal endoscopy application segment globally — competing on flexible endoscope imaging quality, therapeutic instrument capability breadth, and AI-assisted polyp detection platform performance.
Bronchoscopy represents the fastest-growing application segment in the Endoscopy Devices market, projected to expand at a CAGR of approximately 12.20% from 2026 to 2033, driven by the extraordinary commercial momentum of robotic bronchoscopy platforms and the large unmet clinical need for accessible, minimally invasive peripheral lung lesion biopsy in the context of rapidly growing lung cancer screening CT scan adoption. The expanding implementation of low-dose CT lung cancer screening programs — now recommended for high-risk current and former smokers by leading clinical societies in the United States and progressively adopted in European and Asian health systems — is generating a rapidly growing cohort of patients with screen-detected peripheral lung nodules requiring biopsy evaluation, creating a large and commercially compelling procedure volume opportunity for robotic bronchoscopy systems that can access these lesions with higher diagnostic yield than conventional bronchoscopy. Intuitive Surgical's Ion and Johnson & Johnson's Monarch robotic bronchoscopy platforms are the leading commercial systems serving this application — with both platforms generating strong procedure volume growth as thoracic medicine programs accumulate experience with robot-assisted peripheral lung lesion biopsy and integrate these systems into their standard diagnostic pathway for screen-detected lung nodule management.

Regional Insights
North America
North America Commands the Global Endoscopy Devices Market With the World's Highest Procedural Volumes, Most Advanced Cancer Screening Infrastructure, AI Endoscopy Tool Leadership, and Global Robotic Endoscopy Technology Commercialization Strength*
North America holds the dominant position in the global Endoscopy Devices market, accounting for approximately 38.60% of total global market revenue in 2025, and is projected to maintain steady leadership at a CAGR of approximately 7.90% from 2026 to 2033. The United States drives the overwhelming majority of North American revenue — where the combination of the world's highest colonoscopy screening procedure volumes, the most comprehensive insurance-reimbursed upper gastrointestinal endoscopy access, the leading robotic bronchoscopy and flexible endoscopy technology commercialization ecosystem, the most progressive AI-assisted endoscopy regulatory clearance environment, and the highest per-capita healthcare capital equipment investment collectively create the world's most commercially developed and technically advanced endoscopy device market. Leading companies commanding the North American Endoscopy Devices market include Olympus Medical Systems, Boston Scientific, Medtronic, Karl Storz, Stryker, Intuitive Surgical, and Ambu — each maintaining strong commercial positions across their respective product categories and clinical application segments.
Canada contributes meaningfully to North American market revenue through its well-developed colorectal cancer and upper gastrointestinal screening program infrastructure and consistently growing hospital endoscopy unit capital investment — with Canadian provincial health systems progressively expanding endoscopy access capacity to address growing procedural demand from aging demographics, improving screening participation rates, and expanding therapeutic endoscopy program development. The North American Endoscopy Devices market trajectory through 2033 is exceptionally positive — reinforced by the simultaneous growth catalysts of expanding cancer screening procedural volumes, single-use disposable endoscopy adoption acceleration, AI colonoscopy tool mainstream deployment, and robotic bronchoscopy platform commercial ramp-up that collectively create multiple high-value and concurrent growth vectors within the region's large, sophisticated, and highly competitive endoscopy device market environment.
Asia Pacific
Asia Pacific Is the Fastest-Growing Endoscopy Devices Market, Powered by the World's Largest GI Disease Patient Populations, Rapid Healthcare Infrastructure Expansion, Government Cancer Screening Investment, and Strong Domestic Manufacturing in Japan, China, and South Korea*
Asia Pacific is the fastest-growing regional segment in the Endoscopy Devices market, projected to expand at a CAGR of approximately 9.80% from 2026 to 2033 — the highest regional growth rate globally. Japan represents the most technically sophisticated and commercially mature Asia Pacific market — home to global endoscopy industry leaders Olympus and Fujifilm — and simultaneously the country with the world's most advanced gastric cancer and colorectal cancer screening endoscopy infrastructure, the highest endoscopic procedure quality standards, and the strongest clinical adoption of AI-assisted endoscopy diagnostic tools including Fujifilm's CAD EYE and Olympus' EndoBRAIN systems. China and India are the most commercially dynamic high-growth endoscopy device markets in the region — with China's large and rapidly expanding hospital network capital equipment investment combined with India's growing private hospital endoscopy program establishment generating exceptional first-time and upgrade endoscopy device capital equipment demand. Key companies serving Asia Pacific Endoscopy Devices market growth include Olympus, Fujifilm, Karl Storz, Boston Scientific, and rapidly growing domestic Chinese manufacturers including Sonoscape Medical Corporation and SonoScape.
South Korea contributes meaningfully to Asia Pacific market growth through its highly developed gastric cancer and colorectal cancer screening program infrastructure — with South Korea operating one of the world's most comprehensive national cancer endoscopic screening programs — and its strong domestic medical device manufacturing ecosystem that generates competitive endoscopy equipment options for regional healthcare markets. Southeast Asian markets — including Thailand, Malaysia, Singapore, Indonesia, and Vietnam — are experiencing accelerating first-time endoscopy device adoption driven by government hospital quality improvement programs, expanding medical tourism infrastructure investment, and the growing availability of cost-competitive endoscopy systems that make premium endoscopy program establishment accessible within regional healthcare budgets. The demographic scale, economic growth momentum, and rapidly expanding healthcare infrastructure investment across Asia Pacific collectively position the region as the most commercially exciting and fastest-expanding regional market in global endoscopy devices through 2033.
Top Key Players
-
Olympus Corporation — Olympus Medical Systems (Japan)
-
Fujifilm Holdings Corporation (Japan)
-
Hoya Corporation — Pentax Medical (Japan)
-
Boston Scientific Corporation (United States)
-
Medtronic plc (Ireland / United States)
-
Karl Storz SE & Co. KG (Germany)
-
Stryker Corporation (United States)
-
Ambu A/S (Denmark)
-
Intuitive Surgical Inc. (United States)
-
Smith & Nephew plc (United Kingdom)
-
CONMED Corporation (United States)
-
Johnson & Johnson MedTech — Auris Health (United States)
Recent Developments
-
Olympus Corporation (2025) — Launched its next-generation EVIS X1 endoscopy system featuring RealLumi simultaneous multi-light imaging technology that enables real-time white light, narrow band imaging, and texture and color enhancement imaging display — combined with integrated AI-powered intelligent endoscope assistance — representing Olympus's most significant flagship gastrointestinal endoscopy system advancement in over a decade and reinforcing its global market leadership position in premium flexible videoendoscopy across gastrointestinal, respiratory, and urological application categories.
-
Ambu A/S (2024) — Reported strong commercial revenue growth for its single-use endoscopy device portfolio including the aScope 5 Broncho range — driven by accelerating hospital adoption of disposable bronchoscopy as standard clinical practice — while simultaneously announcing expansion of its single-use endoscopy product pipeline into urological and flexible gastrointestinal endoscope categories, reinforcing Ambu's position as the global category leader in single-use endoscopy devices and demonstrating first-mover commercial advantage as hospital infection control programs progressively mandate disposable endoscopy adoption.
-
Boston Scientific (2025) — Achieved strong commercial momentum for its Exalt Model D single-use duodenoscope following FDA guidance recommending transition from reusable duodenoscopes — while simultaneously advancing AI-assisted endoscopy capability through the integration of its LumenVision imaging enhancement platform across its therapeutic gastrointestinal endoscopy device portfolio — creating a differentiated premium endoscopy offering combining infection safety, advanced imaging, and AI-assisted lesion detection that is gaining strong traction among hospital therapeutic endoscopy programs.
-
Intuitive Surgical (2025) — Reported continued strong commercial expansion of its Ion endoluminal bronchoscopy system — achieving growing robotic bronchoscopy program establishment across community and academic hospital thoracic medicine programs in the United States and initiating international market expansion in Europe and Japan — with the Ion system generating expanding procedure volume growth as thoracic medicine specialists gain experience with robot-assisted peripheral lung lesion biopsy and integrate the platform into standard screen-detected lung nodule diagnostic pathways.
-
Karl Storz (2024) — Introduced its IMAGE1 S Rubina 4K fluorescence imaging system for laparoscopic and endoscopic surgery — enabling surgeons to perform real-time near-infrared fluorescence imaging during minimally invasive procedures for intraoperative tissue perfusion assessment, sentinel lymph node mapping, and bile duct visualization — representing a significant advancement in rigid endoscopy visualization capability that expands the clinical utility of laparoscopy visualization systems beyond standard white light imaging and addresses growing oncological and reconstructive surgery quality requirements.
Market Trends
The Accelerating Transition to Single-Use Disposable Endoscopy Device Platforms and the Mainstream Clinical Integration of AI-Powered Diagnostic Intelligence Into Endoscopy Systems Are the Two Most Defining Trends Reshaping the Endoscopy Devices Market*
The most commercially transformative structural trend in the Endoscopy Devices market is the accelerating institutional adoption of single-use disposable endoscopy instruments — progressively expanding from bronchoscopes and duodenoscopes into urological and flexible gastrointestinal endoscopy categories. The high-profile duodenoscope-associated infection outbreaks and subsequent FDA regulatory guidance recommending disposable alternatives have established a clear infection safety case for single-use endoscopy that hospital infection control committees and risk management teams are progressively translating into institutional product conversion decisions. As leading manufacturers scale single-use endoscopy manufacturing capacity — progressively reducing per-unit costs and improving product performance equivalence to reusable alternatives — the economic barriers to disposable endoscopy adoption are declining and the total cost of care advantages of eliminating reprocessing labor, equipment, and quality assurance costs are becoming more broadly recognized, accelerating the commercial conversion trajectory across multiple endoscopy device categories simultaneously.
The progressive integration of AI-powered computer-aided detection and characterization tools into clinical endoscopy practice is the second major trend transforming performance standards and technology differentiation dynamics in the endoscopy devices ecosystem. Multiple commercially cleared and clinically validated AI colonoscopy detection systems are achieving growing adoption in gastroenterology practices and hospital endoscopy programs — driven by clinical quality imperative evidence demonstrating significant adenoma detection rate improvements and by payer and health system quality program incentives that reward measurable colonoscopy quality metric improvements. As AI endoscopy tool applications expand beyond colonoscopy into upper GI lesion characterization, Barrett's surveillance, bronchoscopy navigation, and capsule endoscopy automated reading — AI is becoming an expected standard capability of premium endoscopy platforms rather than a premium feature add-on — creating important technology differentiation pressure on endoscopy device manufacturers to deliver comprehensive and clinically validated AI platform capabilities across their full endoscopy device product portfolios.
Segments Covered in the Report
By Product:
-
Endoscopes
-
Flexible Endoscopes
-
Rigid Endoscopes
-
Capsule Endoscopes
-
Robot-Assisted Endoscopes
-
Endoscopy Visualization Systems
-
HD Imaging Systems
-
4K Imaging Systems
-
3D Imaging Systems
-
Endoscopy Operative Devices
-
Biopsy Forceps
-
Graspers
-
Scissors
-
Clip Appliers
-
Endoscopy Fluid Management Systems
-
Endoscopy Irrigation Pumps
By Application:
-
Gastrointestinal Endoscopy
-
Laparoscopy
-
Arthroscopy
-
Bronchoscopy
-
Urology
-
Gynecology
-
Otolaryngology
By End User:
-
Hospitals
-
Ambulatory Surgical Centers
-
Specialty Clinics
-
Diagnostic Imaging Centers
By Region:
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the Endoscopy Devices market and what is its projected value by 2033?
Answer: The Endoscopy Devices market was valued at USD 52.00 billion in 2025 and is projected to reach USD 112.30 billion by 2033, growing at a CAGR of 8.30% from 2026 to 2033. This growth is driven by rising gastrointestinal cancer screening program expansion, accelerating minimally invasive surgery adoption, single-use disposable endoscopy platform growth, and AI-assisted diagnostic tool integration across clinical endoscopy specialties globally.
Question 2: Which product segment dominates the Endoscopy Devices market?
Answer: Endoscopes represent the dominant product segment in the Endoscopy Devices market with approximately 47.20% of total global market revenue in 2025 — driven by the enormous procedural volume of flexible gastrointestinal endoscopy, minimally invasive laparoscopic and arthroscopic surgery, and bronchoscopy that collectively generate the largest capital equipment and accessories revenue in the endoscopy industry. Visualization systems are the fastest-innovating product segment — with the commercial transition from HD to 4K imaging generating substantial capital equipment upgrade cycles across hospital endoscopy suites and surgical operating rooms globally.
Question 3: Which region leads the Endoscopy Devices market and which is growing the fastest?
Answer: North America leads the Endoscopy Devices market with approximately 38.60% of global revenue in 2025 — driven by the world's highest endoscopic procedure volumes, the most advanced cancer screening infrastructure, AI endoscopy tool leadership, and global robotic endoscopy technology commercialization strength. Asia Pacific is the fastest-growing region at approximately 9.80% CAGR through 2033 — driven by the world's largest GI disease patient populations, rapidly expanding hospital endoscopy infrastructure, government cancer screening program investment, and strong domestic manufacturing capability in Japan, China, and South Korea.
Question 4: Why are single-use disposable endoscopy devices growing so rapidly in the Endoscopy Devices market?
Answer: Single-use disposable endoscopy devices are growing exceptionally fast within the Endoscopy Devices market — driven primarily by compelling infection control and patient safety advantages that completely eliminate cross-contamination risk from reprocessing failures, following high-profile infection outbreaks associated with reusable duodenoscopes and subsequent FDA regulatory guidance recommending disposable alternatives. Beyond infection safety, single-use devices eliminate reprocessing labor, equipment, and quality assurance costs — creating an improving total cost of care economic case that healthcare organizations are progressively recognizing and incorporating into their endoscopy program product decisions.
Question 5: How is artificial intelligence changing diagnostics in the Endoscopy Devices market?
Answer: AI-powered computer-aided detection systems integrated into colonoscope and gastrointestinal endoscopy platforms — including commercially cleared products from Medtronic, Fujifilm, and Olympus — are demonstrating significant improvements in adenoma detection rates supported by multiple randomized clinical trial publications, creating a growing clinical quality imperative for gastroenterology programs to adopt AI-assisted colonoscopy as standard of care. The AI application range in the Endoscopy Devices market is expanding rapidly beyond colonoscopy polyp detection into upper GI lesion characterization, capsule endoscopy automated reading, and bronchoscopy navigation guidance — with AI integration expected to become a standard performance expectation across premium endoscopy platforms throughout the 2026–2033 forecast period.
