Endoscopes Market Overview
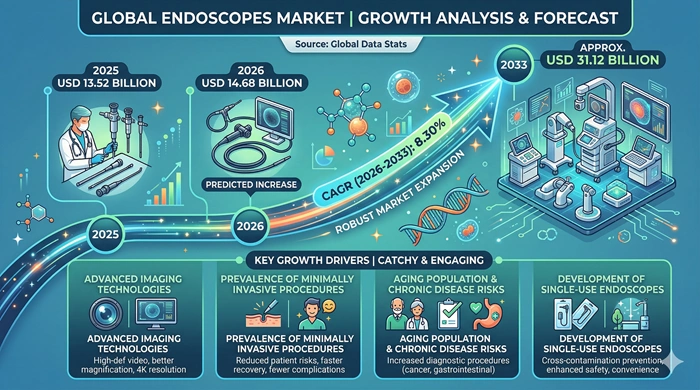
The global Endoscopes market size is valued at USD 13.52 billion in 2025 and is predicted to increase from USD 14.68 billion in 2026 to approximately USD 31.12 billion by 2033, growing at a CAGR of 8.30% from 2026 to 2033.
Endoscopes are precision-engineered minimally invasive medical instruments used by clinicians to visually examine, diagnose, and treat conditions within the body's internal structures — including the gastrointestinal tract, respiratory system, urinary tract, joints, abdominal cavity, and nasal passages — through natural body openings or small incisions, without the need for large open surgical access. Available in a broad range of configurations — including flexible gastrointestinal endoscopes, rigid laparoscopes and cystoscopes, wireless capsule endoscopes, and the rapidly emerging category of robot-assisted endoscopic systems — these instruments have become indispensable tools across virtually every clinical specialty in modern medicine. The Endoscopes market is experiencing sustained and robust growth driven by the globally rising prevalence of gastrointestinal diseases, cancers, and chronic respiratory conditions requiring endoscopic diagnosis and treatment, the accelerating adoption of minimally invasive surgical techniques across hospitals and ambulatory surgical centers, continuous technological innovation in high-definition imaging, single-use disposable endoscope platforms, and robotic endoscopy systems.
AI Impact on the Endoscopes Industry
Artificial Intelligence Is Transforming Endoscopic Lesion Detection, Real-Time Diagnostic Decision Support, Automated Quality Monitoring, and Robot-Assisted Endoscopic Procedure Navigation in Ways That Are Meaningfully Improving Diagnostic Accuracy and Procedural Safety Across Gastrointestinal, Pulmonary, and Surgical Endoscopy Applications*
Artificial intelligence is making a profound and increasingly clinically validated impact across the Endoscopes market, most powerfully through the development and commercial deployment of AI-powered real-time polyp detection and characterization systems integrated directly into colonoscopy endoscope platforms. Deep learning computer vision models — trained on millions of annotated colonoscopy image frames — are demonstrating consistent ability to detect colorectal polyps, adenomas, and early-stage malignant lesions with sensitivity levels that meet or exceed experienced endoscopist performance, and several AI-assisted colonoscopy platforms including Medtronic's GI Genius, Fujifilm's CAD EYE, and Olympus' EndoBRAIN have received regulatory clearance and are achieving growing clinical adoption in North America, Europe, and Japan. These AI polyp detection tools are generating meaningful improvements in adenoma detection rates — the most important quality metric in colonoscopy practice — with multiple randomized clinical trial publications demonstrating statistically significant adenoma detection rate improvements in AI-assisted versus standard colonoscopy, building a strong evidence base that is accelerating adoption among gastroenterology practices and hospital endoscopy units globally.
AI is also advancing rapidly in bronchoscopy and robotic endoscopy guidance applications — where computer vision algorithms and real-time anatomical mapping tools are being integrated into bronchoscope navigation platforms to improve the accuracy and success rate of transbronchial biopsy procedures targeting peripheral lung lesions that are notoriously difficult to access with conventional bronchoscopy approaches. AI-powered robotic bronchoscopy platforms including Intuitive Surgical's Ion endoluminal system and Auris Health's Monarch platform are combining flexible robotic bronchoscope hardware with AI-assisted real-time fluoroscopic and electromagnetic navigation guidance to achieve superior peripheral lung lesion biopsy accuracy compared to conventional bronchoscopy — representing a major clinical advancement that is expanding the diagnostic reach of bronchoscopic endoscopy into the critical early-stage lung cancer detection application. The progressive integration of AI diagnostic support into both diagnostic and therapeutic endoscopy platforms is creating an important technology differentiation dimension within the Endoscopes market that is driving premium product adoption and raising the competitive bar for innovation among leading endoscope manufacturers.
Growth Factors
Rising Global Prevalence of Gastrointestinal Diseases and Cancers, Accelerating Minimally Invasive Surgery Adoption, Technological Advances in Imaging and Disposable Endoscopy, and Expanding Endoscopic Screening Program Investments Are the Core Growth Drivers*
The most fundamental and broadly impactful driver of the Endoscopes market is the rapidly rising global prevalence of gastrointestinal diseases and cancers — including colorectal cancer, gastric cancer, inflammatory bowel disease, gastroesophageal reflux disease, peptic ulcers, and liver disease — that collectively create enormous and growing demand for endoscopic diagnostic and therapeutic procedures worldwide. Colorectal cancer is the second most commonly diagnosed cancer globally, and endoscopy-based colorectal cancer screening programs — most prominently colonoscopy-based screening — are progressively being implemented across more countries, age groups, and insurance coverage frameworks as the clinical evidence for screening's effectiveness in reducing colorectal cancer mortality accumulates and earns stronger public health policy endorsement. The growing global adoption of organized colorectal cancer screening programs — combined with the expanding clinical indication range for upper gastrointestinal endoscopy, capsule endoscopy, and endoscopic ultrasound — is generating a large, structurally growing, and highly predictable procedural volume base for endoscopic instruments that underpins confident long-term revenue growth projections for the endoscopy equipment market.
The global transition of surgical practice toward minimally invasive laparoscopic, arthroscopic, and endoscopic approaches — driven by the clinically demonstrated advantages of minimally invasive procedures including smaller incisions, reduced postoperative pain, shorter hospital stays, faster patient recovery, and lower complication rates compared to traditional open surgery — is simultaneously expanding the application range and procedural volume for endoscopic instruments across surgical specialties. Laparoscopic procedures have progressively become the standard of care for cholecystectomy, appendectomy, colorectal resection, bariatric surgery, and gynecological procedures — generating substantial and growing demand for high-quality rigid and flexible laparoscopes from hospital operating room and ambulatory surgical center customers. The continuing innovation in endoscope imaging technology — including the commercialization of 4K ultra-high-definition imaging systems, narrow band imaging for improved lesion characterization, fluorescence imaging for intraoperative tissue perfusion assessment, and 3D stereoscopic laparoscope visualization — is further stimulating endoscope equipment investment cycles as hospitals and surgical centers upgrade their endoscopy and laparoscopy capital equipment to access these clinically meaningful imaging performance improvements.

Market Outlook
With Single-Use Disposable Endoscope Adoption Accelerating, AI-Powered Diagnostic Assistance Achieving Clinical Mainstream Status, Robotic Endoscopy Platforms Expanding, and Emerging Market Endoscopy Infrastructure Growing Rapidly, the Endoscopes Market Outlook Is Robustly Positive Through 2033*
The long-term outlook for the Endoscopes market is firmly positive and supported by a powerful combination of clinical, technological, regulatory, and commercial growth catalysts expected to sustain strong revenue expansion throughout the 2026–2033 forecast period. The single most disruptive and commercially significant structural shift currently underway in the endoscopy product category is the rapid adoption of single-use disposable endoscopes — driven by compelling infection control advantages over reusable endoscopes that require complex, time-consuming, and imperfect high-level disinfection processes between patient uses. High-profile duodenoscope-associated infection outbreaks — where contaminated reusable duodenoscopes transmitted multidrug-resistant pathogens including carbapenem-resistant Enterobacteriaceae between patients despite adherence to standard reprocessing protocols — have created powerful infection control, patient safety, and liability risk management motivations for hospitals to adopt single-use disposable endoscope alternatives that eliminate cross-contamination risk entirely. Regulatory guidance from the FDA strongly recommending transition to disposable duodenoscope designs has reinforced this adoption trend — creating a large and durable replacement market opportunity for single-use endoscope products that is expected to progressively expand from duodenoscopes into bronchoscopes, urological endoscopes, and eventually gastrointestinal endoscope categories during the forecast period.
Robotic endoscopy represents the most transformative long-term technology frontier in the Endoscopes market — with robotic bronchoscopy systems from Intuitive Surgical and Johnson & Johnson's Auris Health, robotic colonoscopy platforms in clinical development, and flexible robotic endoscope systems for upper gastrointestinal procedures progressively validating the clinical and commercial viability of robot-assisted endoscopic approaches in high-value procedural applications. As robotic endoscopy technology matures — with improving system reliability, expanding clinical indication clearance, growing physician training programs, and progressively more compelling clinical outcome evidence — the category is expected to generate significant new capital equipment revenue streams for leading endoscopy manufacturers and their robotic surgical platform partners during the forecast period. The combination of AI-powered diagnostic intelligence, single-use infection safety advantages, and robotic procedural precision represents a compelling technology convergence that is progressively differentiating premium endoscopy platforms from commodity equipment alternatives — creating strong upgrade cycle dynamics in hospital endoscopy departments globally.
Expert Speaks
-
Gary Guthart, CEO, Intuitive Surgical — "The convergence of robotic systems, advanced imaging, and intelligent software in endoscopic and minimally invasive procedures is creating a transformative new era of surgical and diagnostic precision — enabling physicians to access and treat conditions with an accuracy and consistency that simply was not achievable with conventional manual endoscope instruments. We see the integration of AI-guided navigation and flexible robotic endoscopy as one of the most commercially important and clinically meaningful technology developments in the endoscopy market over the coming decade."
-
Michael Mahoney, CEO, Boston Scientific — "Endoscopy remains one of the most clinically important and commercially dynamic segments of Boston Scientific's medical device portfolio — with continuous innovation in therapeutic endoscopy tools, advanced imaging systems, and single-use flexible endoscope technologies creating compelling new growth opportunities in gastrointestinal, pulmonary, and urological procedure applications. The global expansion of endoscopic screening programs and the growing adoption of endoscopic interventional therapies as alternatives to open surgery are creating a sustainably growing and commercially attractive endoscopy market that we are actively and aggressively investing in."
-
Curt Hartman, CEO, Stryker Corporation — "The transition toward minimally invasive and endoscopic surgical approaches in orthopedics, ENT, and general surgery continues to generate strong and growing demand for high-quality arthroscope, sinuscope, and laparoscope products that enable surgeons to perform complex procedures with smaller incisions and improved patient outcomes. Stryker's endoscopy business is well-positioned to capture the growing global demand for premium visualization systems, and we are investing in next-generation 4K and 3D imaging platforms, AI-assisted surgical guidance tools, and single-use endoscope technologies that address our customers' most important clinical and operational needs."
Key Report Takeaways
-
North America dominates the Endoscopes market, holding approximately 39.20% of global market share in 2025, driven by the world's highest endoscopic procedure volumes, the most advanced colorectal cancer and other gastrointestinal screening program infrastructure, the highest per-capita healthcare spending enabling premium endoscope capital equipment investment, the leading robotic endoscopy technology ecosystem, and the most progressive regulatory environment for AI-assisted endoscopy diagnostic tool clearance — with leading companies including Olympus, Medtronic, Boston Scientific, and Stryker maintaining strong North American commercial positions.
-
Asia Pacific is the fastest-growing regional market for endoscopes, projected to expand at a CAGR of approximately 9.60% from 2026 to 2033, driven by the world's largest populations with high gastrointestinal disease prevalence in China, Japan, South Korea, and India, rapidly expanding hospital endoscopy unit infrastructure investment, growing colorectal and gastric cancer screening program adoption, rising healthcare expenditure per capita, and strong domestic endoscope manufacturer capability in Japan and China generating competitive and accessible premium endoscopy equipment for regional healthcare markets.
-
Flexible endoscopes represent the dominant product segment, accounting for approximately 56.40% of total market revenue in 2025, reflecting the enormous procedural volume of gastrointestinal endoscopy — colonoscopy, upper endoscopy, and sigmoidoscopy — that constitutes the largest single application category in the global endoscopy market and generates the highest recurring consumable and capital equipment replacement demand for flexible endoscope products from hospitals, ambulatory surgical centers, and gastroenterology practices worldwide.
-
Gastrointestinal endoscopy is the largest application segment, contributing approximately 44.80% of total market revenue in 2025, driven by the globally rising prevalence of colorectal cancer, gastric cancer, esophageal disease, inflammatory bowel disease, and other gastrointestinal conditions that require endoscopic diagnosis and therapeutic intervention — with colorectal cancer screening colonoscopy representing the single highest-volume endoscopic procedure category globally.
-
Disposable single-use endoscopes are the fastest-growing product type, projected to expand at a CAGR of approximately 16.40% through 2033, driven by compelling infection control and patient safety advantages over reusable endoscopes, FDA regulatory guidance recommending disposable duodenoscope adoption, and progressive expansion of single-use platforms into bronchoscopy, urological endoscopy, and flexible gastrointestinal endoscopy categories — representing the most structurally transformative product category shift in the endoscope industry in decades.
-
Ambulatory surgical centers are the fastest-growing end-user segment, expected to expand at a CAGR of approximately 10.20% through 2033, driven by the global trend toward outpatient endoscopic procedure delivery — including the rapid shift of colonoscopy, upper endoscopy, and arthroscopy from hospital settings to ambulatory surgical centers — enabled by improvements in endoscopy equipment portability, reprocessing efficiency, and single-use disposable endoscope adoption that make ambulatory endoscopy operationally and economically attractive for both providers and payers.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 31.12 Billion |
| Market Size by 2025 | USD 13.52 Billion |
| Market Size by 2026 | USD 14.68 Billion |
| Market Growth Rate (2026–2033) | CAGR of 8.30% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Segments Covered | Product, Type, Application, End User |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Rising GI Disease and Cancer Screening Volumes, Minimally Invasive Surgery Adoption, Single-Use Endoscope Infection Safety Demand, and AI-Powered Endoscopy Diagnostic Tool Integration Are the Four Primary Drivers of Endoscopes Market Growth*
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising global GI disease prevalence and cancer screening program expansion | ~35% | Global | Short to Long-term |
| Accelerating minimally invasive and laparoscopic surgical technique adoption | ~28% | North America, Europe, Asia Pacific | Short to Long-term |
| Single-use disposable endoscope adoption driven by infection control imperatives | ~24% | North America, Europe | Short to Medium-term |
| AI-assisted endoscopy diagnostic tool integration improving detection performance | ~13% | North America, Europe, Japan | Short to Medium-term |
The most structurally powerful and commercially durable driver of the Endoscopes market is the continuously rising global prevalence of gastrointestinal diseases and cancers — combined with the progressive expansion of organized endoscopic screening programs that systematically translate rising disease prevalence into high-volume clinical procedure demand. Colorectal cancer screening colonoscopy programs — now recommended and covered by national health systems and insurance programs across North America, most of Europe, Japan, South Korea, and Australia — collectively generate tens of millions of colonoscopy procedures annually, creating an enormous and highly predictable base of recurring endoscope equipment utilization and consumable demand that is expected to grow consistently as screening program participation rates improve and screening age thresholds are progressively lowered in line with emerging clinical evidence. The simultaneous global growth in upper gastrointestinal endoscopy for gastric cancer screening — particularly in East Asian countries with the world's highest gastric cancer incidence rates — further reinforces the procedural volume growth foundation that underpins endoscope market revenue expansion.
The global surgical transition toward minimally invasive laparoscopic and endoscopic approaches is creating equally powerful and durable demand for endoscope capital equipment and accessories in hospital operating rooms and ambulatory surgical centers — with the evidence base for minimally invasive surgery's superior patient outcome and recovery time advantages sufficiently established across virtually all major surgical specialties that the transition is now effectively irreversible in developed healthcare markets and rapidly advancing in emerging economies. The growing availability of high-quality laparoscope and arthroscope systems at competitive price points — including strong competition from domestic manufacturers in China, South Korea, and India — is simultaneously accelerating minimally invasive surgery adoption in emerging markets by making premium-quality endoscopic visualization systems accessible at cost structures compatible with developing economy healthcare budgets. This democratization of access to minimally invasive endoscopy technology is progressively expanding the Endoscopes market addressable base into healthcare systems that previously relied primarily on open surgical approaches due to endoscope cost barriers.

Restraints Impact Analysis
High Capital Equipment Costs, Complex Endoscope Reprocessing Requirements, Shortage of Trained Endoscopists, and Reimbursement Constraints in Emerging Markets Restrain Faster Global Endoscopes Market Growth*
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High capital equipment cost of premium endoscope systems limiting adoption in budget-constrained settings | ~34% | Emerging markets — Asia Pacific, Latin America, MEA | Short to Long-term |
| Complex and costly endoscope reprocessing requirements creating operational burden | ~28% | Global | Short to Long-term |
| Shortage of trained endoscopists limiting procedure capacity expansion | ~26% | Global — especially developing markets | Short to Medium-term |
| Reimbursement rate constraints limiting endoscopic procedure economic viability | ~12% | Europe, Asia Pacific | Short to Medium-term |
The most broadly experienced commercial constraint on the Endoscopes market is the high capital equipment cost of premium endoscope systems — particularly the sophisticated flexible videoendoscope platforms from leading manufacturers including Olympus, Fujifilm, and Pentax Medical that represent the global quality standard for gastrointestinal endoscopy — which creates meaningful affordability barriers for hospital endoscopy unit capital investment in cost-sensitive healthcare markets across Asia Pacific, Latin America, Africa, and the Middle East. Premium flexible endoscope systems require not only capital equipment investment in the endoscope units themselves but also substantial accompanying infrastructure investment in reprocessing equipment — including automated endoscope reprocessors, drying cabinets, and storage systems — that collectively represent large capital expenditure requirements that can delay or prevent endoscopy program establishment in resource-limited healthcare settings. The emergence of competitive lower-cost endoscope alternatives from domestic manufacturers in China and South Korea is progressively addressing this cost barrier in some markets — but achieving the imaging quality, durability, and clinical outcome equivalence of leading Japanese and U.S. manufacturer endoscope platforms at substantially lower price points remains a challenge that limits the commercial reach of lower-cost alternatives in the premium clinical endoscopy segment.
The complex, time-consuming, and operationally demanding reprocessing requirements for reusable flexible endoscopes — involving multi-step manual cleaning, leak testing, high-level disinfection in automated or manual reprocessing systems, forced-air drying, and proper storage procedures that collectively take 45 to 90 minutes per endoscope after each patient use — create significant operational burden for endoscopy unit staff and represent a persistent constraint on procedure scheduling throughput capacity. Reprocessing failures — whether from staff non-compliance with protocol steps, equipment malfunction, or inadequate drying that supports microbial growth — have been implicated in serious healthcare-associated infection outbreaks involving endoscopes, creating ongoing patient safety liability concerns that are motivating the progressive transition to single-use disposable endoscope platforms in infection-sensitive application categories. While single-use endoscopes address the reprocessing burden and safety concern simultaneously, their higher per-procedure material cost creates a different economic constraint that requires healthcare organizations to carefully evaluate total cost of care economics before committing to disposable endoscope adoption programs.
Opportunities Impact Analysis
Single-Use Disposable Endoscope Category Expansion, AI-Powered Colonoscopy CADe System Mainstream Adoption, Emerging Market Endoscopy Infrastructure Build-Out, and Robotic Flexible Endoscopy Platform Commercialization Create Major Growth Opportunities*
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Single-use disposable endoscope platform expansion beyond duodenoscopes | ~34% | North America, Europe | Short to Medium-term |
| AI-powered computer-aided detection colonoscopy system mainstream clinical adoption | ~28% | North America, Europe, Japan | Short to Medium-term |
| Emerging market endoscopy program establishment and infrastructure investment | ~26% | Asia Pacific, Latin America, MEA | Medium to Long-term |
| Robotic flexible endoscopy platform commercial expansion and clinical indication broadening | ~12% | North America, Europe | Medium to Long-term |
The rapid commercial expansion of single-use disposable endoscope platforms across an expanding range of endoscope categories represents the most immediately impactful and commercially transformative near-term opportunity in the Endoscopes market — with the compelling infection control, patient safety, operational simplification, and liability risk reduction advantages of disposable endoscopes creating strong and progressive institutional adoption momentum that is progressively expanding from duodenoscopes and bronchoscopes into urological and gastrointestinal flexible endoscope categories. Companies including Ambu, Boston Scientific, Olympus, and Karl Storz are investing substantially in single-use endoscope product portfolio development and commercialization — recognizing that the disposable endoscope transition represents one of the most significant category-defining business model transformations in the endoscopy industry's history, with recurring per-procedure disposable product revenue replacing the historically cyclical and lower-frequency capital equipment replacement revenue model of reusable endoscope business. Healthcare systems that adopt comprehensive single-use endoscope programs generate predictable and growing recurring product revenue streams for endoscope manufacturers — creating a commercially favorable business model transition that major endoscope companies are actively managing.
Emerging market endoscopy program establishment and infrastructure build-out represents a large and commercially compelling long-term growth opportunity for the Endoscopes market — as the combination of rapidly growing middle-class populations with increasing gastrointestinal disease awareness, rising healthcare expenditure, and government investment in cancer screening and hospital quality improvement is progressively creating the conditions for meaningful endoscopic procedure volume growth in previously underserved markets across India, Southeast Asia, Africa, and Latin America. The substantial unmet clinical need for endoscopic cancer screening and gastrointestinal disease diagnosis in these large population markets — combined with the improving accessibility of cost-competitive endoscope equipment and training programs — is enabling healthcare organizations in these regions to establish or significantly expand endoscopy program capacity for the first time, creating large cohorts of first-time endoscope capital equipment purchasers entering the market during the forecast period.

Segment Analysis
By Product
Flexible Endoscopes Lead the Endoscopes Market Revenue While Single-Use Capsule and Disposable Platforms Register the Strongest Growth Momentum*
Flexible endoscopes hold the dominant product position in the Endoscopes market, accounting for approximately 56.40% of total global market revenue in 2025, reflecting the overwhelming procedural volume dominance of flexible gastrointestinal endoscopy — encompassing colonoscopy, upper endoscopy, sigmoidoscopy, endoscopic retrograde cholangiopancreatography (ERCP), and endoscopic ultrasound — that collectively represent the largest single category of endoscopic procedures performed globally and generate the highest absolute revenue contribution from both capital equipment and accessories sales. The clinical range of flexible endoscope applications has expanded continuously — with advancing therapeutic flexible endoscopy capabilities including endoscopic mucosal resection, endoscopic submucosal dissection, peroral endoscopic myotomy, and endoscopic bariatric procedures creating growing demand for premium therapeutic-grade flexible endoscope platforms with enhanced working channel capacity, improved maneuverability, and integrated advanced imaging capabilities. North America dominates flexible endoscope revenue globally — driven by the highest colonoscopy screening procedure volumes, the most reimbursement-supported upper endoscopy access, and the most commercially developed therapeutic endoscopy program infrastructure — while Asia Pacific is the fastest-growing region for flexible endoscope adoption driven by rapidly expanding gastric and colorectal cancer screening programs. Leading suppliers including Olympus Medical Systems, Fujifilm Holdings, and Hoya Corporation (Pentax Medical) maintain dominant global flexible endoscope market positions through their comprehensive flexible videoendoscope system portfolios.
Rigid endoscopes — encompassing laparoscopes, arthroscopes, cystoscopes, sinuscopes, and hysteroscopes — represent the second-largest product segment in the Endoscopes market, accounting for approximately 28.60% of total revenue in 2025, supported by the large and growing procedural volumes of laparoscopic general surgery, gynecological laparoscopy, arthroscopic orthopedic surgery, and urological cystoscopy that collectively drive substantial capital equipment and accessory demand in hospital operating rooms and ambulatory surgical centers. Rigid endoscope adoption is growing fastest in Asia Pacific — driven by the rapid expansion of minimally invasive surgical program infrastructure across Chinese and Indian hospitals, the strong domestic rigid endoscope manufacturing capability in China generating cost-competitive alternatives, and the growing surgical specialty training programs developing the endoscopist workforce needed to drive procedural volume growth. Karl Storz SE, Stryker Corporation, Smith & Nephew, and CONMED Corporation are among the leading rigid endoscope suppliers with strong global operating room market positions — competing on imaging quality, instrument ergonomics, system integration capabilities, and the breadth of complementary surgical instrument accessories available within their respective endoscopy ecosystem platforms.
By Application
Gastrointestinal Endoscopy Leads the Endoscopes Market Application Revenue While Bronchoscopy Registers the Fastest Procedure Volume and Technology Investment Growth*
Gastrointestinal endoscopy represents the dominant application segment in the Endoscopes market, contributing approximately 44.80% of total global market revenue in 2025, driven by the globally rising burden of colorectal cancer, gastric cancer, esophageal cancer, inflammatory bowel disease, and other gastrointestinal conditions that require endoscopic diagnosis, surveillance, and therapeutic intervention — creating the largest and most commercially important single endoscope procedure application category globally. The expansion of organized colorectal cancer screening colonoscopy programs across additional countries and insurance coverage frameworks — combined with the progressive lowering of recommended colonoscopy screening initiation age to 45 in the United States and equivalent policy changes in other markets — is generating structurally growing colonoscopy procedure volume growth that sustains strong and predictable gastrointestinal endoscope equipment demand. North America leads gastrointestinal endoscopy application revenue globally — driven by the world's highest colonoscopy screening rates, the most comprehensively reimbursed upper gastrointestinal endoscopy access, and the most advanced AI-assisted polyp detection technology integration in colonoscopy practice — while Asia Pacific is experiencing the fastest procedural volume growth driven by gastric cancer screening program expansion. Leading market participants serving this application include Olympus, Fujifilm, Medtronic (GI Genius AI system), and Boston Scientific — each competing across the full spectrum of diagnostic and therapeutic gastrointestinal endoscopy applications.
Bronchoscopy represents the fastest-growing application segment in the Endoscopes market, projected to expand at a CAGR of approximately 11.80% from 2026 to 2033, driven primarily by the extraordinary commercial momentum of robotic bronchoscopy platforms — including Intuitive Surgical's Ion and Johnson & Johnson's Monarch systems — that are enabling physicians to perform minimally invasive bronchoscopic biopsy of peripheral lung lesions that are inaccessible to conventional bronchoscopy, creating a transformative new diagnostic pathway for early-stage lung cancer detection with profound implications for lung cancer mortality outcomes. The lung cancer early detection opportunity is commercially enormous — lung cancer being the leading cause of cancer mortality globally — and the clinical validation of robotic bronchoscopy as a safe and technically feasible approach to peripheral lesion biopsy is generating rapid adoption momentum in thoracic medicine programs at leading academic medical centers and community hospitals with high-volume lung cancer care programs. North America and Europe are the primary markets for bronchoscopy application growth — driven by lung cancer screening program expansion, the concentration of thoracic medicine expertise and robotic bronchoscopy program development, and the most commercially mature market environment for robotic bronchoscopy capital equipment investment.

Regional Insights
North America
North America Leads the Global Endoscopes Market With the Highest Endoscopic Procedure Volumes, Most Advanced Cancer Screening Infrastructure, Strongest AI Endoscopy Tool Adoption, and Global Leadership in Robotic Endoscopy Technology Commercialization*
North America holds the dominant position in the global Endoscopes market, accounting for approximately 39.20% of total global market revenue in 2025, and is projected to maintain steady market leadership at a CAGR of approximately 7.80% from 2026 to 2033. The United States is overwhelmingly the primary regional revenue driver — where the combination of the world's highest colonoscopy screening procedure volumes, the most comprehensively insurance-reimbursed upper gastrointestinal endoscopy access, the leading robotic bronchoscopy and endoscopy technology ecosystem, the most progressive AI-assisted endoscopy diagnostic tool regulatory clearance environment, and the highest per-capita healthcare capital equipment investment levels collectively create the world's most commercially developed and technically advanced endoscopy market. Leading companies dominating the North American Endoscopes market include Olympus Medical Systems, Boston Scientific, Medtronic, Stryker, Karl Storz, Intuitive Surgical, and Ambu — each maintaining strong commercial positions across their respective endoscopy product categories and application segments.
Canada contributes meaningfully to North American endoscope market revenue through its well-developed colorectal cancer screening program infrastructure and consistently growing hospital endoscopy unit capital investment — with Canadian provincial health systems progressively expanding endoscopy access capacity to address growing procedural demand from aging demographics and improving colorectal cancer screening participation rates. The North American Endoscopes market outlook for 2026–2033 is strongly positive — reinforced by the structural procedural volume growth from cancer screening program expansion, the powerful single-use disposable endoscope adoption wave, the AI colonoscopy CADe system mainstream deployment, and the commercial ramp-up of robotic bronchoscopy and flexible endoscopy platforms that collectively create multiple simultaneous high-value growth vectors within the region's large and sophisticated endoscopy market.
Asia Pacific
Asia Pacific Is the Fastest-Growing Endoscopes Market, Driven by the World's Largest GI Disease Patient Populations, Rapidly Expanding Cancer Screening Programs, Growing Hospital Endoscopy Infrastructure Investment, and Strong Domestic Endoscope Manufacturing Capability in Japan, China, and South Korea*
Asia Pacific is the fastest-growing regional segment in the Endoscopes market, projected to expand at a CAGR of approximately 9.60% from 2026 to 2033 — the highest of any region globally. Japan is the most technically sophisticated and commercially mature Asia Pacific endoscope market — home to the world's leading endoscope manufacturers including Olympus Medical Systems and Fujifilm Holdings — and simultaneously the country with the world's most advanced gastric cancer screening infrastructure, highest endoscopic procedure quality standards, and strongest clinical adoption of AI-assisted endoscopy diagnostic tools including Fujifilm's CAD EYE and Olympus' EndoBRAIN systems. China and South Korea represent the most commercially dynamic high-growth endoscope markets in the region — driven by rapidly expanding colorectal and gastric cancer screening program investments, strong government healthcare infrastructure modernization funding, and growing middle-class consumer health awareness that is generating exceptional procedural volume growth across gastrointestinal and minimally invasive surgical endoscopy applications. Key companies active in the Asia Pacific Endoscopes market include Olympus, Fujifilm, Hoya (Pentax Medical), Karl Storz, and rapidly growing Chinese domestic manufacturers including Sonoscape Medical Corporation and SonoScape.
India represents one of the most commercially important emerging growth opportunities in the Asia Pacific endoscopes market — where the combination of a 1.4 billion population with high gastrointestinal disease burden, rapidly expanding private hospital network endoscopy infrastructure investment, and growing healthcare expenditure are creating exceptional first-time endoscope capital equipment adoption volume. Southeast Asian markets — including Thailand, Malaysia, Indonesia, and Vietnam — are experiencing rapid endoscopy program establishment and expansion driven by government hospital quality improvement programs, medical tourism development initiatives, and the growing availability of cost-competitive endoscope systems from Japanese and Chinese manufacturers that are making premium endoscopy equipment accessible within regional healthcare budgets. The breadth and demographic scale of Asia Pacific's endoscopy market development opportunity — spanning Japan's technical leadership through China's scale through India and Southeast Asia's infrastructure build-out — positions the region as the most commercially exciting and fastest-expanding frontier in the global Endoscopes market through 2033.
Top Key Players
-
Olympus Corporation — Olympus Medical Systems (Japan)
-
Fujifilm Holdings Corporation (Japan)
-
Hoya Corporation — Pentax Medical (Japan)
-
Boston Scientific Corporation (United States)
-
Medtronic plc (Ireland / United States)
-
Karl Storz SE & Co. KG (Germany)
-
Stryker Corporation (United States)
-
Ambu A/S (Denmark)
-
Intuitive Surgical Inc. (United States)
-
Smith & Nephew plc (United Kingdom)
-
CONMED Corporation (United States)
-
Sonoscape Medical Corporation (China)
Recent Developments
-
Olympus Corporation (2025) — Launched its next-generation EVIS X1 endoscopy platform featuring a new RealLumi imaging technology that enables simultaneous white light, narrow band imaging, and texture and color enhancement imaging display modes — combined with an AI-powered Endo-Therapy Plus intelligent endoscope control system that automatically adjusts imaging parameters in real time to optimize visualization for each procedure phase — representing Olympus's most significant flagship endoscope platform advancement in a decade and reinforcing its global market leadership in premium flexible videoendoscope systems for gastrointestinal, pulmonary, and urological applications.
-
Ambu A/S (2024) — Reported strong commercial revenue growth for its single-use bronchoscope portfolio — including the aScope 5 Broncho range — driven by accelerating hospital adoption of disposable bronchoscopy as the standard of care approach for ICU and bronchoscopy suite applications, with Ambu simultaneously announcing the expansion of its single-use endoscope product pipeline into new urological and gastrointestinal flexible endoscope categories — reinforcing its position as the global leader in the strategically critical single-use endoscope category and demonstrating its first-mover commercial advantage as hospital infection control programs progressively mandate disposable endoscope adoption.
-
Intuitive Surgical (2025) — Reported continued strong commercial expansion of its Ion endoluminal bronchoscopy system — with growing robotic bronchoscopy program establishment across community and academic hospital thoracic medicine programs in the United States and initial international market launches in Europe and Japan — achieving a growing installed base of Ion systems and generating expanding procedure volume growth as thoracic medicine specialists gain experience with robot-assisted peripheral lung lesion biopsy and integrate Ion into their standard diagnostic pathway for screen-detected lung nodule evaluation.
-
Boston Scientific (2025) — Launched the Exalt Model D single-use duodenoscope as the centerpiece of its disposable endoscopy strategy — achieving strong commercial uptake among U.S. hospital endoscopy programs responding to FDA guidance recommending transition away from reusable duodenoscopes — while simultaneously advancing its AI-assisted endoscopy capability through the integration of its LumenVision imaging enhancement platform across its therapeutic gastrointestinal endoscope product line, creating a differentiated premium endoscopy offering that combines infection safety, advanced imaging, and AI-assisted lesion detection in a comprehensive therapeutic endoscopy platform.
-
Karl Storz (2024) — Introduced its 4K IMAGE1 S Rubina fluorescence imaging system for laparoscopic and robotic surgery — enabling surgeons to perform real-time near-infrared fluorescence imaging during minimally invasive procedures for intraoperative tissue perfusion assessment, sentinel lymph node mapping, and bile duct visualization — representing a significant technical advancement in rigid endoscope imaging capability that expands the clinical utility of laparoscopic visualization systems beyond standard white light imaging and addresses growing surgical quality and safety requirements in oncological and reconstructive laparoscopic surgery.
Market Trends
The Transformative Shift Toward Single-Use Disposable Endoscope Platforms and the Mainstream Clinical Integration of AI-Powered Computer-Aided Detection Systems Are the Two Most Defining Trends Reshaping the Endoscopes Market*
The most commercially transformative structural trend reshaping the Endoscopes market is the accelerating institutional adoption of single-use disposable endoscopes — driven by compelling infection control, patient safety, and operational simplification advantages that are progressively making disposable alternatives the standard of care choice across an expanding range of endoscope categories. The high-profile regulatory and patient safety pressure on reusable duodenoscopes — combined with the growing clinical availability of performance-comparable single-use alternatives from Ambu, Boston Scientific, and Olympus — has established a clear and broadly recognized infection safety case for single-use endoscope adoption that hospital infection control committees, risk management teams, and endoscopy medical directors are progressively translating into institutional product conversion decisions. As single-use endoscope manufacturing scale increases, per-unit product costs decline, and the total cost of care economics of disposable versus reusable approaches become more favorable — particularly when the full operational costs of reprocessing labor, equipment, and quality assurance are accurately accounted for — the commercial adoption trajectory of single-use endoscopes is expected to accelerate substantially across bronchoscopy, urological endoscopy, and flexible gastrointestinal applications during the forecast period.
The integration of AI-powered computer-aided detection and characterization systems into clinical endoscope practice — most advanced in colonoscopy where multiple commercially cleared AI polyp detection platforms including Medtronic's GI Genius and Fujifilm's CAD EYE are achieving growing adoption — represents the second major transformative trend progressively reshaping quality standards and technology differentiation dynamics in the Endoscopes market. The clinical evidence demonstrating that AI-assisted colonoscopy significantly improves adenoma detection rates compared to standard colonoscopy is now supported by multiple randomized controlled trial publications — creating a growing clinical quality imperative for gastroenterology practices and hospital endoscopy programs to adopt AI-assisted colonoscopy as standard of care to meet evolving quality benchmark requirements. As AI endoscopy applications expand beyond colonoscopy adenoma detection into upper gastrointestinal lesion characterization, Barrett's esophagus surveillance, bronchoscopy navigation guidance, and capsule endoscopy automated reading — the technology is expected to become a standard performance expectation integrated into premium endoscope platforms across multiple clinical specialties throughout the 2026–2033 forecast period.
Segments Covered in the Report
By Product:
-
Flexible Endoscopes
-
Gastrointestinal Endoscopes
-
Bronchoscopes
-
Urologic Endoscopes
-
Arthroscopes
-
Rigid Endoscopes
-
Laparoscopes
-
Cystoscopes
-
Sinuscopes
-
Capsule Endoscopes
-
Robot-Assisted Endoscopes
By Type:
-
Disposable Endoscopes
-
Reusable Endoscopes
By Application:
-
Gastrointestinal Endoscopy
-
Laparoscopy
-
Arthroscopy
-
Bronchoscopy
-
Urology
-
Otolaryngology
-
Gynecology
By End User:
-
Hospitals
-
Ambulatory Surgical Centers
-
Specialty Clinics
-
Diagnostic Imaging Centers
By Region:
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the Endoscopes market and what is its projected value by 2033?
Answer: The Endoscopes market was valued at USD 13.52 billion in 2025 and is projected to reach USD 31.12 billion by 2033. It is expected to grow at a CAGR of 8.30% from 2026 to 2033, driven by rising global gastrointestinal disease prevalence, expanding cancer screening programs, accelerating minimally invasive surgery adoption, single-use disposable endoscope platform growth, and AI-assisted endoscopy diagnostic tool integration.
Question 2: What are the key types of endoscopes available in the Endoscopes market?
Answer: The Endoscopes market offers a comprehensive range of instrument types — from flexible videoendoscopes used for gastrointestinal, bronchoscopic, and urological examination through rigid laparoscopes and arthroscopes for minimally invasive surgery, wireless capsule endoscopes for small intestine imaging, to the rapidly emerging category of robot-assisted flexible endoscopes for peripheral lung lesion biopsy and complex intraluminal procedures. Single-use disposable endoscope versions are available across bronchoscope, duodenoscope, and urological endoscope categories and represent the fastest-growing product type in the market — driven by their compelling infection control and patient safety advantages over reusable alternatives.
Question 3: Which region leads the Endoscopes market and which is the fastest growing?
Answer: North America dominates the Endoscopes market with approximately 39.20% of global revenue in 2025, driven by the world's highest endoscopic procedure volumes, most advanced cancer screening infrastructure, strongest AI endoscopy tool adoption, and global leadership in robotic endoscopy technology commercialization. Asia Pacific is the fastest-growing region at approximately 9.60% CAGR through 2033, driven by the world's largest gastrointestinal disease patient populations, rapidly expanding cancer screening programs, growing hospital endoscopy infrastructure investment, and strong domestic endoscope manufacturing capability in Japan, China, and South Korea.
Question 4: Why are single-use disposable endoscopes growing so rapidly in the Endoscopes market?
Answer: Single-use disposable endoscopes are experiencing exceptional growth in the Endoscopes market — projected at approximately 16.40% CAGR through 2033 — primarily because they completely eliminate the cross-contamination infection risk associated with reusable endoscopes that require complex high-level disinfection reprocessing between patients, following high-profile duodenoscope-associated infection outbreaks involving multidrug-resistant pathogens that generated strong FDA regulatory guidance recommending disposable alternatives. Beyond infection safety, disposable endoscopes also eliminate reprocessing labor and equipment costs, simplify operational workflows, and remove the quality assurance burden of reprocessing validation — creating a compelling total operational value proposition that healthcare organizations are progressively recognizing and acting upon.
Question 5: How is artificial intelligence transforming diagnostic performance in the Endoscopes market?
Answer: AI-powered computer-aided detection systems integrated into colonoscope platforms — including commercially cleared products from Medtronic, Fujifilm, and Olympus — are demonstrating statistically significant improvements in adenoma detection rates compared to standard colonoscopy in randomized clinical trial data, establishing a compelling clinical quality case for AI-assisted colonoscopy adoption that is progressively translating into mainstream gastroenterology practice adoption across North America, Europe, and Japan. Beyond colonoscopy polyp detection, AI applications in the Endoscopes market are expanding into upper gastrointestinal lesion characterization, capsule endoscopy automated image reading, and robotic bronchoscopy navigation guidance — with the breadth and clinical validation of AI endoscopy applications expected to grow substantially throughout the 2026–2033 forecast period, making AI integration a standard performance expectation for premium endoscope platforms.
