eClinical Solutions Market Overview
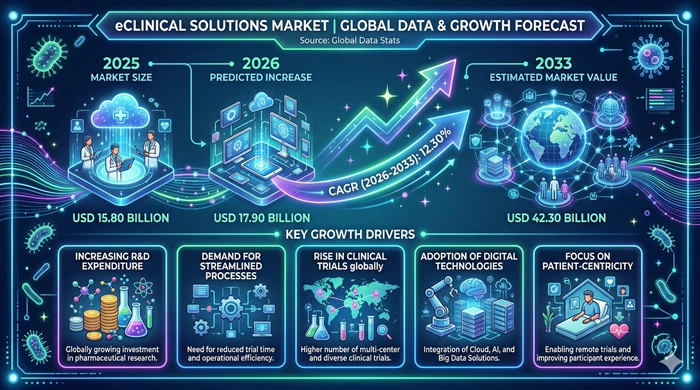
The global eClinical Solutions market size is valued at USD 15.80 billion in 2025 and is predicted to increase from USD 17.90 billion in 2026 to approximately USD 42.30 billion by 2033, growing at a CAGR of 12.30% from 2026 to 2033.
eClinical solutions encompass a broad and strategically important portfolio of technology platforms designed to digitize, automate, and streamline clinical trial operations — including electronic data capture systems, clinical trial management platforms, electronic patient-reported outcome tools, randomization and trial supply management systems, electronic trial master files, safety and pharmacovigilance platforms, and advanced clinical analytics solutions that collectively transform how pharmaceutical companies, contract research organizations, and academic institutions design, manage, and analyze clinical research programs. These interconnected digital platforms replace paper-based clinical trial workflows with validated electronic systems that improve data quality, accelerate trial timelines, reduce operational costs, and enhance regulatory compliance — addressing the pharmaceutical and life sciences industry's most urgent clinical development efficiency and productivity challenges. The eClinical Solutions market is experiencing robust and accelerating growth driven by the rising global clinical trial volume, increasing adoption of decentralized and hybrid trial models, expanding cloud-based deployment, regulatory digitization mandates, and the transformative integration of AI and machine learning into clinical data management and trial operations.
AI Impact on the eClinical Solutions Industry
Artificial Intelligence Is Transforming Clinical Data Quality Monitoring, Predictive Patient Recruitment, Automated Safety Signal Detection, Intelligent Protocol Design Optimization, and Real-Time Trial Performance Analytics in Ways That Are Fundamentally Accelerating Drug Development Timelines and Improving Clinical Research Productivity Across the Global eClinical Solutions Ecosystem*
Artificial intelligence is delivering its most commercially impactful contributions to the eClinical Solutions market through the integration of machine learning-powered risk-based monitoring algorithms into clinical data management and electronic data capture platforms — where AI systems continuously analyze incoming trial data streams to detect data quality anomalies, protocol deviations, safety signals, and site performance issues in real time with sensitivity and consistency that far exceeds traditional periodic manual data review approaches. These AI-powered risk-based monitoring platforms — integrated into EDC and CDMS solutions from leading vendors including Medidata, Oracle Health Sciences, Veeva Systems, and Ennov — are enabling clinical operations teams to focus their limited monitoring resources on the highest-risk trial sites and data domains while reducing the overall cost and time burden of clinical data oversight, directly addressing one of the pharmaceutical industry's most significant clinical development cost drivers. The FDA and EMA have both issued guidance supporting risk-based monitoring approaches that AI platforms are specifically designed to enable — creating a favorable regulatory framework that is accelerating institutional adoption of AI-powered eClinical monitoring tools as the evolving standard of clinical data quality management practice.
AI is simultaneously advancing the eClinical solutions ecosystem through intelligent patient recruitment optimization tools — where machine learning models analyze electronic health record data, demographic patterns, comorbidity profiles, and historical enrollment performance data to identify and predict the highest-probability patient recruitment sites and populations for specific clinical trial protocols — dramatically improving enrollment rate predictability and reducing the costly enrollment delays that represent one of the most significant timeline and cost risk factors in pharmaceutical clinical development programs. Natural language processing applications are also transforming clinical data management within eClinical platforms — enabling automated medical coding, narrative adverse event report processing, protocol deviation categorization, and unstructured data extraction from clinical notes and patient-reported outcome instruments — reducing manual data cleaning labor requirements and improving the speed and consistency of clinical database lock and regulatory submission readiness timelines. The combination of AI diagnostic intelligence, automated quality monitoring, and intelligent recruitment optimization across the eClinical Solutions market is creating meaningful competitive differentiation between vendors with comprehensive AI capability portfolios and those offering more traditional rule-based clinical data management functionality.
Growth Factors
Rising Global Clinical Trial Volume, Accelerating Decentralized Trial Model Adoption, Cloud-Based eClinical Platform Expansion, Regulatory Digitization Mandates, and Growing CRO Outsourcing Demand Are the Core Growth Engines of the eClinical Solutions Market*
The most foundational and commercially durable growth driver of the eClinical Solutions market is the continuously rising global clinical trial volume — driven by the extraordinary pharmaceutical and biopharmaceutical industry pipeline across oncology, immunology, neurology, rare disease, infectious disease, and metabolic therapeutic areas that collectively generate growing demand for sophisticated eClinical technology platforms capable of managing increasingly complex, multi-site, and multinational clinical research programs. The global pharmaceutical industry is investing at record levels in clinical research and development — with global pharmaceutical R&D spending exceeding USD 250 billion annually and growing — generating expanding procurement budgets for the eClinical technology platforms that are essential infrastructure for modern clinical trial operations at every stage from study start-up through data lock and regulatory submission. Biosimilar development programs, combination product trials, adaptive trial designs, and the growing platform trial model that simultaneously evaluates multiple drug candidates within a single protocol framework are each generating specific eClinical technology requirements that are driving new platform capability investment and procurement across the clinical research technology market.
The transformative global adoption of decentralized and hybrid clinical trial models — dramatically accelerated by the COVID-19 pandemic's forced demonstration that remote trial participation, telemedicine visits, direct-to-patient drug shipment, and wearable sensor data collection can successfully replace traditional site-only trial designs — is simultaneously generating strong and growing demand for the specialized eClinical technology platforms that enable and support decentralized trial operations. Electronic patient-reported outcome platforms, remote patient monitoring data integration middleware, telemedicine visit management tools, and decentralized trial orchestration platforms are among the fastest-growing eClinical solution categories — driven by the pharmaceutical industry's growing commitment to patient-centric trial designs that reduce burden on participants, improve enrollment diversity, and accelerate recruitment by removing geographic access barriers to trial participation. The concurrent shift of eClinical platform deployment from on-premise to cloud-based software-as-a-service models — driven by the operational flexibility, scalability, implementation speed, and total cost of ownership advantages of cloud platforms — is generating strong revenue growth for cloud-native eClinical vendors while simultaneously creating challenging competitive dynamics for traditional on-premise eClinical software providers seeking to maintain customer relationships.

Market Outlook
With Decentralized Trial Technology Achieving Mainstream Adoption, AI-Powered Clinical Analytics Accelerating, Cloud eClinical Platform Migration Progressing, and Emerging Market Clinical Research Capacity Expanding Rapidly, the eClinical Solutions Market Outlook Is Exceptionally Strong Through 2033*
The long-term outlook for the eClinical Solutions market is firmly positive, supported by a compelling combination of technology innovation, regulatory evolution, pharmaceutical pipeline growth, and geographic market expansion drivers expected to sustain strong double-digit revenue growth throughout the 2026–2033 forecast period. The most commercially transformative structural shift currently reshaping the eClinical product landscape is the convergence of previously separate clinical data management, trial management, patient engagement, and clinical analytics platforms into integrated unified clinical trial technology ecosystems — where a single vendor platform manages the complete clinical data lifecycle from study design and protocol authoring through data collection, quality management, statistical analysis, and regulatory submission preparation. Leading eClinical vendors including Medidata (Dassault Systèmes), Oracle Health Sciences, Veeva Systems, and Parexel's IMPACT platform are progressively building these comprehensive clinical trial technology ecosystems — creating meaningful customer switching cost advantages and expanding the revenue addressable market per customer relationship.
The growing regulatory emphasis on real-world evidence generation, adaptive trial designs, and master protocol frameworks is simultaneously driving new eClinical technology investment requirements — as pharmaceutical companies seek eClinical platforms capable of managing the more complex data flows, adaptive randomization algorithms, and regulatory submission requirements associated with innovative trial designs. The FDA's Real-World Evidence Program, the EMA's adaptive pathways initiative, and equivalent regulatory innovation programs across major pharmaceutical regulatory jurisdictions are progressively creating regulatory pathways for innovative trial designs that require more sophisticated eClinical technology support — expanding the addressable technology investment opportunity within the eClinical Solutions market beyond standard randomized controlled trial management toward a broader spectrum of complex clinical research design support. Emerging market clinical research capacity expansion — particularly in India, China, Brazil, and Eastern Europe — is additionally creating growing new geographic revenue opportunities as clinical trial activity progressively shifts toward cost-competitive and patient-rich emerging market sites that are investing in eClinical technology adoption.
Expert Speaks
-
Chris Boerner, CEO, Bristol-Myers Squibb — "The digitization of clinical trial operations through advanced eClinical technology platforms — encompassing electronic data capture, decentralized trial tools, AI-powered monitoring, and integrated clinical analytics — is one of the most important strategic investments pharmaceutical companies can make to improve the efficiency, quality, and speed of clinical development programs that ultimately determine how quickly we can bring transformative medicines to patients. We see the continued evolution of eClinical solutions as a critical enabler of our ambition to reduce clinical development cycle times and improve the probability of technical and regulatory success across our oncology and immunology pipeline programs."
-
Albert Bourla, CEO, Pfizer Inc. — "The COVID-19 vaccine development experience demonstrated conclusively that digital clinical trial technology — including decentralized trial platforms, real-time data monitoring, and AI-powered trial operations tools — can dramatically compress development timelines without compromising data quality or regulatory compliance when implemented with rigor and intention. Pfizer is committed to sustained investment in eClinical platform capabilities that enable faster, more efficient, and more patient-friendly clinical trial designs — recognizing that technology-enabled clinical development acceleration translates directly into faster patient access to transformative medicines and stronger commercial performance."
-
Pascal Soriot, CEO, AstraZeneca — "Digital transformation of clinical research operations — built on advanced eClinical data management, AI-assisted monitoring, decentralized patient engagement, and integrated regulatory technology platforms — represents one of the most commercially significant and strategically important operational efficiency investments in pharmaceutical development today. AstraZeneca is actively advancing its digital clinical development capabilities across our global trial portfolio — leveraging the most advanced eClinical solutions available to improve enrollment efficiency, data quality, and regulatory submission readiness across our oncology, cardiovascular, and respiratory pipeline programs."
Key Report Takeaways
-
North America dominates the eClinical Solutions market, holding approximately 43.20% of global market share in 2025, driven by the world's highest clinical trial activity concentration, the most extensive pharmaceutical and biopharmaceutical company R&D investment base, the most progressive regulatory digitization environment under FDA oversight, the highest adoption rates of cloud-based eClinical platform deployment, and the concentration of leading global eClinical technology vendors including Medidata, Oracle Health Sciences, Veeva Systems, and Parexel with dominant North American commercial customer bases.
-
Asia Pacific is the fastest-growing regional market for eClinical solutions, projected to expand at a CAGR of approximately 14.60% from 2026 to 2033, driven by the rapidly growing clinical trial activity in China, India, South Korea, and Japan — combined with pharmaceutical company efforts to enroll large and genetically diverse patient populations in Asian markets — and progressive government regulatory digitization programs across Asian pharmaceutical authorities that are establishing eClinical data submission requirements equivalent to FDA and EMA standards.
-
Electronic Data Capture is the dominant product segment, accounting for approximately 32.40% of total market revenue in 2025, reflecting EDC's status as the foundational eClinical technology investment across virtually every clinical trial regardless of therapeutic area, trial phase, or organizational type — generating the largest single category of eClinical technology procurement from pharmaceutical companies, CROs, and academic research institutions that collectively conduct thousands of clinical trials annually requiring validated electronic data collection infrastructure.
-
Cloud-based deployment is the dominant and fastest-growing delivery mode, contributing approximately 61.30% of total market revenue in 2025 and growing at a CAGR of approximately 14.20% through 2033, driven by the superior implementation speed, operational flexibility, automatic update management, and total cost of ownership advantages of cloud-based eClinical platforms that are progressively compelling pharmaceutical companies and CROs to migrate from on-premise legacy systems toward cloud-native software-as-a-service eClinical technology deployments.
-
Pharmaceutical and biopharmaceutical companies represent the largest end-user segment, contributing approximately 48.60% of total market revenue in 2025, driven by the enormous and continuously growing R&D investment these organizations make in clinical development programs across all therapeutic areas — with large pharmaceutical companies operating portfolios of dozens to hundreds of simultaneous clinical trials that generate comprehensive eClinical technology procurement requirements spanning EDC, CTMS, CDMS, safety systems, eTMF, and clinical analytics platforms.
-
Contract Research Organizations are the fastest-growing end-user segment, expected to expand at a CAGR of approximately 15.40% through 2033, driven by the pharmaceutical industry's progressive outsourcing of clinical trial operations to CROs that manage increasingly large and complex trial portfolios requiring enterprise-grade eClinical technology infrastructure — with leading CROs including IQVIA, Covance, PPD, and Syneos Health collectively managing thousands of active clinical trials generating massive and growing eClinical technology procurement demand.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 42.30 Billion |
| Market Size by 2025 | USD 15.80 Billion |
| Market Size by 2026 | USD 17.90 Billion |
| Market Growth Rate (2026–2033) | CAGR of 12.30% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Segments Covered | Product, Delivery Mode, End User |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Rising Clinical Trial Volume, Decentralized Trial Adoption, Cloud eClinical Platform Migration, Regulatory Digitization Mandates, and CRO Outsourcing Growth Are the Five Pillars Powering eClinical Solutions Market Revenue Expansion*
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising global clinical trial volume and pharmaceutical R&D investment growth | ~32% | Global | Short to Long-term |
| Accelerating decentralized and hybrid clinical trial model adoption | ~27% | North America, Europe | Short to Long-term |
| Cloud-based eClinical platform deployment migration from on-premise legacy systems | ~25% | Global | Short to Medium-term |
| Regulatory digitization mandates and electronic submission requirement expansion | ~16% | North America, Europe, Asia Pacific | Short to Medium-term |
The most enduring and commercially significant driver of the eClinical Solutions market is the continuously rising global clinical trial volume — generated by the pharmaceutical and biopharmaceutical industry's record R&D pipeline investment across oncology, immunology, rare disease, and neurology that collectively produce growing numbers of clinical development programs requiring comprehensive eClinical technology support at every stage of the clinical research lifecycle. The complexity of modern clinical trial designs — incorporating adaptive randomization, Bayesian statistical approaches, platform trial architectures, combination therapy evaluations, and biomarker-stratified patient selection — is simultaneously increasing the eClinical technology sophistication requirements per trial beyond what previous-generation electronic data capture and trial management systems were designed to support. This combination of growing trial volume and rising per-trial technology complexity is generating a compounding eClinical technology investment growth dynamic that sustains the market's strong double-digit CAGR throughout the forecast period.
The decentralized clinical trial revolution is simultaneously generating entirely new eClinical technology category creation — with electronic patient-reported outcome platforms, remote patient monitoring data integration middleware, direct-to-patient medication management tools, and virtual site visit management systems emerging as fast-growing eClinical solution categories that generate incremental revenue streams alongside traditional EDC and CTMS platforms. Regulatory agencies including the FDA, EMA, and PMDA have all published guidance supporting decentralized trial designs and the eClinical technologies that enable them — creating a favorable regulatory framework that accelerates pharmaceutical company adoption of decentralized trial approaches and the associated eClinical technology investments. The pharmaceutical industry's growing recognition that decentralized trial designs improve patient enrollment diversity, reduce dropout rates, and accelerate recruitment timelines — all commercially valuable outcomes — is generating strong internal organizational motivation for the eClinical technology investments required to operationalize decentralized trial capabilities.

Restraints Impact Analysis
High Implementation Costs, System Integration Complexity, Data Privacy Regulation Compliance Burden, and Workforce Digital Skill Gaps Restrain Faster eClinical Solutions Market Penetration*
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High eClinical platform implementation and validation costs for smaller organizations | ~32% | Asia Pacific, Latin America, smaller sponsors | Short to Long-term |
| Complex system integration requirements across multi-vendor eClinical technology ecosystems | ~28% | Global | Short to Long-term |
| Growing data privacy regulation compliance burden across multiple jurisdictions | ~26% | Europe, North America, Asia Pacific | Short to Medium-term |
| Workforce digital skill gaps limiting eClinical platform utilization effectiveness | ~14% | Global — especially emerging markets | Short to Medium-term |
The most commonly experienced commercial barrier constraining broader and faster adoption of advanced eClinical solutions is the significant implementation cost, system validation investment, and organizational change management effort required to deploy and operationalize enterprise eClinical platforms — particularly for smaller pharmaceutical companies, academic research institutions, and emerging market CROs that lack the dedicated clinical technology infrastructure teams and implementation budget that large pharmaceutical companies routinely commit to eClinical deployment programs. Computer system validation requirements under FDA 21 CFR Part 11 and EU Annex 11 — which mandate comprehensive documented validation of every eClinical system used in regulated clinical research — create substantial upfront time and resource investment requirements that extend eClinical implementation timelines and increase total cost of adoption beyond the software licensing costs that procurement decision-makers typically evaluate initially. The proliferation of multiple eClinical technology platforms within pharmaceutical company clinical operations — where EDC, CTMS, safety, eTMF, RTSM, and clinical analytics systems from different vendors must exchange data reliably and in compliance with CDISC data standards — creates persistent and costly system integration complexity that consumes significant clinical technology team resources throughout the operational lifetime of each eClinical platform deployment.
The growing complexity of data privacy regulatory compliance across multiple jurisdictions — including GDPR in Europe, HIPAA in the United States, PDPA across Asian markets, and a proliferating landscape of national data localization and patient data protection regulations — is creating substantial compliance burden for pharmaceutical companies and CROs conducting multinational clinical trials that collect patient data across dozens of countries simultaneously. eClinical platform vendors must continuously invest in privacy engineering, data residency capabilities, consent management tools, and regulatory compliance documentation to maintain compliance with evolving data privacy requirements — generating ongoing development costs that reduce the R&D investment available for platform capability innovation. Pharmaceutical companies conducting global trials must simultaneously manage eClinical platform configurations, data transfer agreements, and patient consent frameworks that satisfy the data privacy regulatory requirements of every country where trial sites operate — creating operational complexity that disproportionately burdens smaller organizations with limited regulatory affairs resources.
Opportunities Impact Analysis
Unified Clinical Trial Technology Platform Convergence, AI Clinical Analytics Mainstream Adoption, Emerging Market Clinical Research Expansion, and Real-World Evidence Platform Integration Create Major eClinical Solutions Market Opportunities*
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Unified integrated clinical trial technology ecosystem platform development | ~33% | North America, Europe | Short to Medium-term |
| AI-powered clinical analytics and risk-based monitoring mainstream adoption | ~28% | North America, Europe, Japan | Short to Medium-term |
| Emerging market clinical research capacity expansion and eClinical adoption | ~27% | Asia Pacific, Latin America, Eastern Europe | Medium to Long-term |
| Real-world evidence platform integration with clinical trial eClinical systems | ~12% | North America, Europe | Medium to Long-term |
The most commercially transformative strategic opportunity reshaping the eClinical Solutions market is the platform convergence trend — where the pharmaceutical industry's longstanding frustration with managing fragmented multi-vendor eClinical technology ecosystems is creating strong demand for unified integrated clinical trial technology platforms that manage the complete clinical data lifecycle within a single connected system architecture. Vendors that successfully build comprehensive eClinical ecosystems — spanning EDC, CTMS, CDMS, eTMF, safety, RTSM, and clinical analytics within a unified data model — are capturing significant competitive advantages including faster system implementation, lower total integration cost, improved data quality from eliminating cross-system data transfers, and superior regulatory submission readiness that are compelling pharmaceutical companies to consolidate eClinical vendor relationships around platform ecosystem leaders. Medidata's Rave platform, Oracle's Clinical One, and Veeva's Vault Clinical suite represent the most advanced current expressions of the integrated eClinical ecosystem strategy — each progressively expanding platform scope to capture a larger share of the total eClinical technology spending per customer.
The mainstream adoption of real-world evidence generation programs — where pharmaceutical companies supplement randomized controlled trial data with evidence from electronic health records, claims databases, patient registries, and wearable sensor networks to support regulatory submissions, label expansions, and post-market surveillance requirements — is creating a compelling new adjacent opportunity within the eClinical solutions ecosystem. eClinical vendors that develop integrated real-world data ingestion, harmonization, and analysis capabilities alongside traditional clinical trial data management functionality are positioned to serve the pharmaceutical industry's growing real-world evidence program requirements — expanding their addressable revenue opportunity significantly beyond the clinical trial operations market into the broader real-world evidence generation and analytics market that is developing rapidly under supportive FDA and EMA regulatory frameworks.

Segment Analysis
By Product
Electronic Data Capture Leads the eClinical Solutions Market Revenue While Clinical Analytics and Decentralized Trial Platforms Register the Fastest Investment Growth*
Electronic Data Capture holds the dominant position in the eClinical Solutions market, accounting for approximately 32.40% of total global market revenue in 2025, reflecting EDC's foundational role as the most universally deployed eClinical technology — present in virtually every regulated clinical trial regardless of therapeutic area, phase, or organizational sponsor type — that captures, validates, and manages the primary clinical trial data that forms the evidentiary basis for regulatory submissions and scientific publications. EDC platforms from leading vendors including Medidata Rave, Oracle Clinical One, OpenClinica, Veeva Vault EDC, and Ennov EDC have progressively evolved from basic electronic data entry tools into sophisticated clinical data management ecosystems with integrated risk-based monitoring, real-time data quality alerting, and direct CDISC CDASH data standards compliance that dramatically streamline the path from data collection through database lock and regulatory submission. North America leads EDC product segment revenue globally — driven by the highest clinical trial activity concentration and the most mature eClinical technology adoption infrastructure — while Europe is the second-largest market. The EDC product segment is growing fastest in Asia Pacific — where the expanding clinical trial activity in China, India, and South Korea is driving first-time EDC adoption at pharmaceutical companies and CROs establishing eClinical technology infrastructure for Asia Pacific trial operations.
Clinical Trial Management Systems represent the second-largest and fastest-growing traditional product segment in the eClinical Solutions market, accounting for approximately 18.60% of total market revenue in 2025 and growing at a CAGR of approximately 13.80% through 2033 as pharmaceutical companies and CROs invest in more sophisticated trial operations management platforms capable of supporting the growing complexity of multinational, multi-site, and decentralized clinical trial portfolios. CTMS platforms manage the operational logistics of clinical trial execution — including site identification and selection, investigator and site management, regulatory document tracking, patient enrollment monitoring, protocol deviation management, and operational performance analytics — with enterprise CTMS solutions from Medidata, Veeva Vault CTMS, Parexel CTMS, and Forte Research Systems serving the needs of large and mid-size pharmaceutical companies and CROs managing complex multi-trial portfolios. Europe is the fastest-growing CTMS market — driven by the expanding clinical trial activity of European pharmaceutical companies, growing regulatory requirements for real-time trial oversight documentation, and the strong adoption of integrated clinical trial technology ecosystems among European pharmaceutical companies that are progressively standardizing CTMS platforms across their trial operations functions.
By Delivery Mode
Cloud-Based Deployment Dominates the eClinical Solutions Market and Drives the Fastest Adoption Across All End-User Segments and Geographies*
Cloud-based delivery mode holds the dominant position in the eClinical Solutions market, accounting for approximately 61.30% of total global market revenue in 2025 and projected to grow at a CAGR of approximately 14.20% through 2033 — driven by the compelling operational, financial, and strategic advantages of cloud-based eClinical platform deployment that are progressively compelling pharmaceutical companies, CROs, and academic research institutions to migrate from on-premise legacy eClinical systems toward cloud-native software-as-a-service platforms. The advantages of cloud eClinical deployment — including elimination of on-premise infrastructure maintenance costs, automatic platform updates that always deliver current regulatory compliance features, elastic scalability that accommodates trial portfolio fluctuations without fixed infrastructure investment, and the ability to support decentralized trial patient and site access from any global location — are compelling and well-understood by clinical technology procurement decision-makers who are progressively mandating cloud-first eClinical platform strategies across their organizations. North America leads cloud-based eClinical deployment revenue globally — driven by the highest cloud technology adoption maturity and the strongest organizational commitment to SaaS platform strategies among North American pharmaceutical companies and CROs — while Asia Pacific is the fastest-growing cloud eClinical deployment region at approximately 15.20% CAGR through 2033 as newly establishing clinical research organizations default to cloud-native eClinical platforms rather than legacy on-premise systems.
On-premise delivery mode accounts for approximately 24.40% of the eClinical Solutions market revenue in 2025 — representing legacy pharmaceutical company and CRO eClinical infrastructure investments that persist in organizations with existing long-term contracts, highly customized system configurations, specific data localization requirements, or institutional technology governance frameworks that complicate or delay cloud migration. The on-premise segment is declining as a share of total eClinical revenue — progressively displaced by cloud and hybrid alternatives — but remains commercially relevant in the near term for large pharmaceutical company enterprise eClinical deployments where the cost and operational complexity of migrating validated on-premise systems to cloud platforms remains a significant short-term barrier. European pharmaceutical companies represent a disproportionate share of remaining on-premise eClinical deployments — driven by both the historically strong on-premise enterprise technology preferences of European pharmaceutical companies and the data sovereignty considerations under GDPR that motivated earlier caution about cloud-based clinical data management that is progressively being addressed by cloud eClinical vendors through EU-region data hosting and contractual data processing safeguards.

Regional Insights
North America
North America Commands the Global eClinical Solutions Market With the World's Highest Clinical Trial Activity Concentration, Most Progressive Regulatory Digitization Environment, Leading AI-Powered eClinical Innovation Ecosystem, and Dominant Cloud-Based Platform Adoption*
North America holds the dominant position in the global eClinical Solutions market, accounting for approximately 43.20% of total global market revenue in 2025, and is projected to maintain steady leadership at a CAGR of approximately 11.80% from 2026 to 2033. The United States drives the overwhelming majority of North American revenue — where the combination of the world's largest pharmaceutical and biopharmaceutical R&D investment base, the highest clinical trial activity concentration, the most progressive FDA regulatory digitization and electronic submission framework, and the highest cloud-based eClinical platform adoption rates collectively create the world's most commercially developed and technology-advanced eClinical solutions market. Leading companies commanding the North American eClinical Solutions market include Medidata Solutions (Dassault Systèmes), Oracle Health Sciences, Veeva Systems, IQVIA Technologies, ERT (now Nuvation Health), and Parexel — each maintaining dominant positions through comprehensive platform ecosystems, deep pharmaceutical customer relationships, and continuous AI capability investment that strengthens their competitive positioning.
Canada contributes meaningfully to North American market revenue through its growing clinical research sector, increasing pharmaceutical company R&D investment, and the progressive alignment of Health Canada's eClinical data submission requirements with FDA standards that drives comparable eClinical technology adoption among Canadian trial sponsors and CROs. The North American eClinical solutions market is at the forefront of AI integration, decentralized trial technology commercialization, and unified platform ecosystem development — serving as the primary commercial incubation market for eClinical innovation that subsequently diffuses to European, Asian, and emerging market clinical research organizations. North America's combination of pharmaceutical industry scale, regulatory sophistication, and eClinical technology vendor concentration positions it to maintain clear market leadership throughout the 2026–2033 forecast period.
Asia Pacific
Asia Pacific Is the Fastest-Growing eClinical Solutions Market, Driven by Rapidly Expanding Clinical Trial Activity in China and India, Growing Pharmaceutical Company R&D Investment, and Progressive Regulatory Digitization Across Asian Pharmaceutical Authorities*
Asia Pacific is the fastest-growing regional segment in the eClinical Solutions market, projected to expand at a CAGR of approximately 14.60% from 2026 to 2033 — the highest regional growth rate globally. China is the most commercially dynamic and rapidly growing Asia Pacific eClinical market — driven by the National Medical Products Administration's progressive implementation of eClinical data submission standards aligned with ICH guidelines, the extraordinary growth of Chinese pharmaceutical and biotech company R&D investment generating growing domestic clinical trial activity, and the increasing participation of multinational pharmaceutical company global clinical trials at Chinese research sites that require eClinical platform adoption compliant with international data standards. India represents the second-largest and equally dynamic Asia Pacific eClinical Solutions market — driven by the country's highly cost-competitive clinical research environment, large and diverse patient populations attractive for global pharmaceutical company trial enrollment, and the growing eClinical technology maturity of India's leading CRO sector. Key companies serving Asia Pacific eClinical solutions market growth include Medidata, Oracle Health Sciences, Veeva Systems, and IQVIA Technologies alongside regional Asian clinical technology providers including Cato Research and EBM Technologies serving domestic pharmaceutical customers.
Japan and South Korea represent the most technically sophisticated and commercially mature Asia Pacific eClinical markets — with well-established pharmaceutical industry eClinical technology adoption, strong regulatory electronic submission requirements, and high cloud-based eClinical platform deployment rates comparable to North American standards. Southeast Asian clinical research market development — particularly in Singapore, Thailand, Australia, and Taiwan — is generating progressive first-time enterprise eClinical technology adoption as pharmaceutical company regional trial activity expands and CRO sector development matures. The combination of China and India's clinical trial volume growth, Japan and Korea's technology adoption maturity, and Southeast Asia's emerging market development collectively positions Asia Pacific as the most commercially exciting and fastest-growing regional eClinical solutions market through 2033.
Top Key Players
-
Medidata Solutions Inc. — Dassault Systèmes (United States / France)
-
Oracle Health Sciences (United States)
-
Veeva Systems Inc. (United States)
-
IQVIA Technologies Inc. (United States)
-
Parexel International Corporation (United States)
-
ERT — Nuvation Health (United States)
-
BioClinica Inc. (United States)
-
Ennov Group (France)
-
OpenClinica LLC (United States)
-
Forte Research Systems Inc. (United States)
-
Merge Healthcare — IBM Watson Health (United States)
-
Castor EDC (Netherlands)
Recent Developments
-
Medidata Solutions — Dassault Systèmes (2025) — Announced the expansion of its Medidata AI clinical intelligence platform with new generative AI-powered protocol design optimization capabilities — enabling pharmaceutical company sponsors to leverage large language model analysis of historical trial protocol performance data to identify enrollment-risk design elements and optimize inclusion/exclusion criteria before study start-up — while simultaneously reporting strong commercial momentum for its Rave EDC and Medidata Rave Decentralized platform combination among large pharmaceutical company customers transitioning to hybrid and fully decentralized trial designs.
-
Veeva Systems (2025) — Reported strong commercial revenue growth for its Vault Clinical suite — encompassing Vault EDC, Vault CTMS, Vault eTMF, and Vault Safety platforms — driven by accelerating pharmaceutical company adoption of Veeva's integrated clinical development cloud strategy that consolidates multiple previously separate eClinical platforms into a unified Veeva Vault architecture, reducing integration complexity and improving regulatory submission readiness for pharmaceutical company customers managing large global clinical trial portfolios.
-
Oracle Health Sciences (2024) — Launched Oracle Clinical One 4.0 — a major platform update introducing enhanced AI-powered risk-based monitoring capabilities, improved decentralized trial workflow support, and next-generation CDISC data standards compliance tools — while announcing expanded partnerships with major global CROs including Covance and PRA Health Sciences to deploy Oracle Clinical One as the preferred EDC and study management platform for their pharmaceutical company sponsor customer portfolios across North American and European markets.
-
IQVIA Technologies (2025) — Announced the commercial launch of its IQVIA Decentralized Trials Platform — a comprehensive integrated eClinical technology suite combining eDiary patient engagement tools, remote patient monitoring data integration, telemedicine visit management, and direct-to-patient medication logistics management within a unified platform — targeting pharmaceutical company sponsors seeking to operationalize fully decentralized or hybrid trial designs for therapeutic areas where patient travel burden to traditional clinical sites creates enrollment and retention challenges.
-
Parexel International (2024) — Completed a significant expansion of its PAREXEL IMPACT eClinical technology platform through the acquisition of ConvergeHEALTH — a specialized clinical data integration and analytics company — adding advanced real-world data harmonization, clinical trial and real-world evidence integration, and AI-powered clinical analytics capabilities to the IMPACT platform ecosystem, positioning Parexel to serve pharmaceutical company demand for integrated clinical trial and real-world evidence data management within a single connected eClinical technology infrastructure.
Market Trends
The Convergence of Unified Integrated eClinical Platform Ecosystems and the Mainstream Adoption of AI-Powered Clinical Analytics and Risk-Based Monitoring Are the Two Most Commercially Defining Trends Reshaping the eClinical Solutions Market Through 2033*
The most commercially transformative structural trend in the eClinical Solutions market is the pharmaceutical industry's accelerating consolidation of fragmented multi-vendor eClinical technology stacks toward unified integrated platform ecosystems — where a single vendor provides a comprehensive connected clinical trial technology suite spanning EDC, CTMS, eTMF, safety, RTSM, and clinical analytics within a unified data architecture. Pharmaceutical companies managing large and complex clinical trial portfolios are recognizing that the integration maintenance burden, data quality risks, and regulatory compliance complexity of operating six to twelve separate eClinical point solutions from different vendors represents a strategic operational liability — motivating increasingly bold vendor consolidation decisions that favor the few eClinical vendors capable of delivering comprehensive platform ecosystems over best-of-breed point solution providers. This platform consolidation trend is simultaneously raising the competitive entry barriers within the eClinical solutions industry — as smaller specialized eClinical technology vendors face growing displacement by ecosystem platform providers — while creating significant expansion revenue opportunities for the platform leaders that successfully convert multi-point-solution customers into consolidated platform ecosystem relationships.
The mainstream integration of AI-powered clinical analytics, risk-based monitoring, and operational intelligence into eClinical platforms is simultaneously becoming a baseline technology expectation rather than a premium differentiator — as pharmaceutical companies and CROs accumulate experience with AI monitoring tools and recognize their tangible clinical data quality and monitoring cost efficiency benefits. As AI eClinical capabilities mature and clinical evidence accumulates demonstrating their impact on trial timeline acceleration and regulatory submission quality, the competitive pressure for all major eClinical vendors to offer comprehensive and clinically validated AI clinical intelligence capabilities is intensifying — progressively elevating the AI technology development investment requirement for maintaining competitive positioning in the eClinical solutions market and accelerating the pace of AI capability expansion across the entire eClinical platform landscape.
Segments Covered in the Report
By Product:
-
Electronic Data Capture (EDC)
-
Clinical Trial Management Systems (CTMS)
-
Clinical Data Management Systems (CDMS)
-
Electronic Patient-Reported Outcomes (ePRO)
-
Electronic Trial Master File (eTMF)
-
Randomization and Trial Supply Management (RTSM)
-
Safety and Pharmacovigilance Systems
-
Clinical Analytics Platforms
By Delivery Mode:
-
On-Premise
-
Cloud-Based
-
Hybrid
By End User:
-
Pharmaceutical and Biopharmaceutical Companies
-
Contract Research Organizations (CROs)
-
Medical Device Companies
-
Academic and Research Institutes
-
Hospitals and Healthcare Providers
By Region:
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the eClinical Solutions market and what is its projected value by 2033?
Answer: The eClinical Solutions market was valued at USD 15.80 billion in 2025 and is projected to reach USD 42.30 billion by 2033, growing at a CAGR of 12.30% from 2026 to 2033. This growth is driven by rising global clinical trial volume, accelerating decentralized trial adoption, cloud-based platform migration, and the mainstream integration of AI-powered clinical analytics across eClinical technology platforms.
Question 2: What are the main types of eClinical solutions used in clinical trials?
Answer: The primary eClinical solutions used in clinical trials include Electronic Data Capture (EDC), Clinical Trial Management Systems (CTMS), Clinical Data Management Systems (CDMS), electronic patient-reported outcomes (ePRO), electronic Trial Master File (eTMF), and Randomization and Trial Supply Management (RTSM) platforms. The eClinical Solutions market is progressively evolving toward unified integrated platform ecosystems that combine multiple previously separate eClinical technology categories within a single connected data architecture.
Question 3: Which region leads the eClinical Solutions market and which is growing the fastest?
Answer: North America leads the eClinical Solutions market with approximately 43.20% of global revenue in 2025 — driven by the world's highest clinical trial activity concentration, most progressive FDA regulatory digitization environment, and dominant cloud-based eClinical platform adoption rates. Asia Pacific is the fastest-growing region at approximately 14.60% CAGR through 2033 — driven by rapidly expanding clinical trial activity in China and India, growing pharmaceutical R&D investment, and progressive regulatory digitization across Asian pharmaceutical authorities.
Question 4: How is the shift to decentralized clinical trials driving growth in the eClinical Solutions market?
Answer: The global adoption of decentralized and hybrid clinical trial models is generating entirely new eClinical technology category creation — including electronic patient-reported outcome platforms, remote patient monitoring data integration tools, and virtual site visit management systems — that generate incremental technology investment alongside traditional EDC and CTMS platforms in the eClinical Solutions market. Regulatory guidance from the FDA and EMA supporting decentralized trial designs is accelerating pharmaceutical company adoption of decentralized trial approaches and the associated eClinical technology investments required to operationalize these patient-centric trial models effectively.
Question 5: Why is cloud-based deployment growing so rapidly in the eClinical Solutions market?
Answer: Cloud-based deployment is growing rapidly in the eClinical Solutions market because it eliminates on-premise infrastructure maintenance costs, delivers automatic regulatory compliance updates, provides elastic scalability for trial portfolio fluctuations, and enables decentralized patient and site access from any global location — advantages that are compelling pharmaceutical companies and CROs to migrate progressively from on-premise legacy systems toward cloud-native SaaS platforms. Cloud-based eClinical platforms accounted for approximately 61.30% of total market revenue in 2025 and are projected to grow at approximately 14.20% CAGR through 2033 as the remaining on-premise installed base progressively migrates toward cloud deployment models.
