Coronary Stents Market Overview
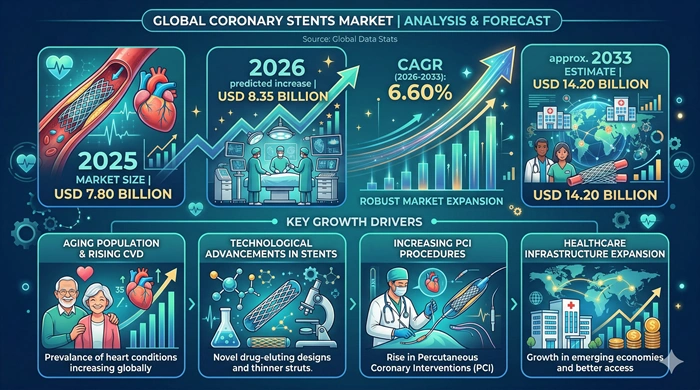
The global Coronary Stents market size is valued at USD 7.80 billion in 2025 and is predicted to increase from USD 8.35 billion in 2026 to approximately USD 14.20 billion by 2033, growing at a CAGR of 6.60% from 2026 to 2033.
Coronary stents are small, mesh-like metallic or bioresorbable tubular devices deployed within narrowed or blocked coronary arteries during percutaneous coronary intervention procedures to restore and maintain adequate blood flow to heart muscle tissue, thereby reducing the risk of myocardial infarction, relieving angina symptoms, and improving long-term cardiovascular outcomes in patients with coronary artery disease. The device category has evolved substantially over the past two decades — transitioning from early bare-metal stent designs that were prone to in-stent restenosis, through first-generation and second-generation drug-eluting stents coated with antiproliferative agents that dramatically reduced restenosis rates, to the current generation of thin-strut cobalt-chromium and platinum-chromium drug-eluting stents with biodegradable polymer coatings and increasingly refined deliverability profiles that continue to improve clinical outcomes. The Coronary Stents market is experiencing sustained commercial growth driven by the rising global burden of coronary artery disease, expanding interventional cardiology procedural volumes in emerging economies, continued clinical evidence favoring drug-eluting stent use across a broadening range of complex lesion types and patient populations, growing adoption of newer-generation thin-strut stent platforms, and the progressive development of bioresorbable scaffold technologies that may offer long-term vessel healing advantages over permanent metallic implants.
AI Impact on the Coronary Stents Industry
Artificial Intelligence Is Transforming Coronary Stent Procedural Planning, Lesion Assessment, Deployment Optimization, Patient Risk Stratification, and Post-Procedure Outcome Prediction in Ways That Are Meaningfully Improving Clinical Results and Expanding the Commercial Reach of the Coronary Stents Market*
Artificial intelligence is reshaping the Coronary Stents market most significantly through its integration into intravascular imaging analysis workflows — where AI-powered image interpretation algorithms applied to intravascular ultrasound and optical coherence tomography imaging data automatically quantify plaque burden, measure minimum lumen area, assess lesion calcification severity, identify stent underexpansion or malapposition, and recommend optimal stent sizing parameters with a speed and consistency that surpasses conventional manual image interpretation by even experienced interventional cardiologists. Companies including Philips, Boston Scientific, and Abbott are progressively embedding AI-assisted imaging analysis capabilities within their intravascular imaging systems and catheterization laboratory workflow platforms — enabling real-time procedural guidance that helps interventional cardiologists achieve more consistent stent optimization outcomes, reduces procedure time, and decreases the rate of suboptimal stent deployment results that are associated with increased rates of stent thrombosis, in-stent restenosis, and major adverse cardiac events. The progressive development of AI-powered fractional flow reserve calculation from coronary CT angiography — where deep learning algorithms generate hemodynamic lesion significance assessments from non-invasive CT imaging without the need for invasive adenosine-based FFR measurement — is additionally influencing coronary stent procedural volumes by improving the appropriateness and precision of patient selection for percutaneous coronary intervention and stent implantation.
AI is further transforming the post-procedure management dimension of the coronary stents market — where machine learning models trained on large-scale real-world clinical outcome datasets are generating patient-specific risk stratification tools that predict individual patient probabilities of stent thrombosis, in-stent restenosis, and major adverse cardiac events based on a comprehensive combination of clinical risk factors, procedural characteristics, intravascular imaging findings, and dual antiplatelet therapy adherence patterns. These AI-powered risk stratification capabilities are informing personalized dual antiplatelet therapy duration recommendations — one of the most clinically consequential management decisions in post-stent patient care — by identifying patients at highest risk of ischemic events who benefit from extended therapy versus patients at elevated bleeding risk where shorter therapy is appropriate. The integration of wearable device data streams and remote patient monitoring signals into AI-powered post-discharge surveillance platforms is additionally generating real-time patient status insights that enable earlier clinical intervention for early warning signals of stent-related complications, progressively improving long-term outcomes for the growing global population of patients living with implanted coronary stents.
Growth Factors
Rising Global Coronary Artery Disease Burden, Expanding Interventional Cardiology Infrastructure in Emerging Markets, Continuous Drug-Eluting Stent Platform Innovation, and Growing Adoption of Complex PCI Procedures Are the Core Growth Drivers of the Coronary Stents Market*
The most powerful and structurally sustained growth driver of the Coronary Stents market is the rising global burden of coronary artery disease — where the convergence of aging population demographics, escalating rates of obesity, type 2 diabetes, hypertension, and dyslipidemia across both developed and developing countries, sedentary lifestyle patterns, and tobacco use is generating a growing and highly prevalent patient population requiring coronary revascularization treatment at rates that are expected to continue increasing across virtually all global regions throughout the forecast period. The World Health Organization estimates that cardiovascular diseases remain the leading cause of death globally — accounting for approximately 17.9 million deaths annually — with coronary artery disease representing the single largest contributor to this mortality burden, generating large and growing clinical volumes for interventional cardiology procedures and coronary stent implantation across hospitals and cardiac catheterization laboratories worldwide. The progressive improvement in drug-eluting stent platform technology — where each successive generation of thin-strut designs, refined polymer coatings, enhanced drug release kinetics, and improved deliverability through complex anatomical configurations has expanded the range of coronary lesion types, patient populations, and clinical scenarios where percutaneous coronary intervention with stent implantation is clinically preferred over surgical bypass — has progressively broadened the total addressable patient population and procedure volume opportunity for the coronary stents market.
The enormous market development opportunity represented by expanding interventional cardiology infrastructure in Asia Pacific, Latin America, and Middle East & Africa — where rapidly growing healthcare investment, accelerating catheterization laboratory installation rates, a growing pool of trained interventional cardiologists, and progressively improving reimbursement frameworks for percutaneous coronary intervention are collectively driving rapid procedural volume growth from a relatively underpenetrated baseline compared to North American and European procedural rates — represents the most commercially significant geographic growth driver for the Coronary Stents market over the forecast period. China alone performs approximately 1.5 million percutaneous coronary intervention procedures annually — a volume that has grown dramatically over the past decade and continues to increase — representing a massive installed procedure base that drives corresponding coronary stent demand. The progressive clinical expansion of complex percutaneous coronary intervention indications — including left main coronary artery stenting, chronic total occlusion revascularization, multivessel coronary intervention, and bifurcation stenting — into patient populations previously managed surgically is additionally expanding the coronary stent addressable market by converting surgical bypass cases into percutaneous interventional cases that generate coronary stent demand.

Market Outlook
With Bioresorbable Scaffold Technology Advancing, Asia Pacific Interventional Cardiology Infrastructure Expanding Rapidly, Drug-Eluting Stent Clinical Evidence Broadening, and Minimally Invasive Coronary Intervention Adoption Growing Globally, the Coronary Stents Market Outlook Is Positive Through 2033*
The long-term outlook for the Coronary Stents market is positive and commercially well-supported — with a combination of rising coronary artery disease prevalence, geographic market penetration growth in emerging economies, continuous product platform innovation, and expanding complex PCI adoption collectively expected to sustain steady revenue growth throughout the 2026–2033 forecast period. The most commercially significant near-term development within the coronary stents ecosystem is the progressive clinical maturation and potential commercial re-emergence of bioresorbable vascular scaffold technology — where the clinical limitations of early-generation bioresorbable scaffolds including higher thrombosis rates and strut fracture issues are being systematically addressed through next-generation thin-strut bioresorbable designs by companies including Abbott, Biotronik, and several Asia Pacific manufacturers — with successful clinical validation of improved next-generation bioresorbable scaffolds potentially creating a meaningful new market segment within the broader Coronary Stents market by offering vessel restoration rather than permanent metallic implantation. The progressive adoption of physiology-guided and imaging-guided coronary intervention protocols — where routine intravascular imaging with OCT or IVUS and routine physiological assessment with FFR or iFR are used to optimize stent selection, sizing, and deployment — is simultaneously elevating procedure quality standards in ways that increase per-procedure device utilization, drive premium device adoption, and improve the clinical evidence base supporting more complex interventional strategies.
The long-term demand fundamentals for the Coronary Stents market remain robust and well-supported — with the combination of an aging global population, persistent cardiovascular risk factor prevalence, growing healthcare access in developing markets, and continuous product innovation creating a durable and growing market opportunity that is expected to sustain mid-single-digit revenue growth through 2033. The development of novel drug-eluting stent designs incorporating innovative antiproliferative agents, biocompatible polymer technologies, and advanced metallic alloy platforms continues to generate competitive differentiation among leading manufacturers — creating product upgrade cycles that drive premium stent adoption among interventional cardiologists seeking the best clinical outcomes for their patients. The integration of digital health technologies — including AI-powered catheterization laboratory guidance systems, connected stent monitoring concepts, and remote patient management platforms — into the coronary intervention ecosystem is progressively expanding the value proposition of the Coronary Stents market beyond the stent device itself to encompass a broader clinical outcome management platform that creates new revenue streams and competitive differentiation for technology-leading companies.
Expert Speaks
-
Michael Mahoney, CEO, Boston Scientific — "Coronary stent technology continues to advance meaningfully — with each new generation of drug-eluting stents delivering thinner struts, more biocompatible polymer platforms, and more precise drug delivery that collectively translate into improved clinical outcomes for patients undergoing percutaneous coronary intervention for coronary artery disease. Boston Scientific is committed to remaining at the forefront of coronary stent innovation through sustained investment in next-generation platform development, clinical evidence generation, and global market expansion that brings the benefits of best-in-class coronary intervention technology to more patients around the world."
-
Robert Ford, CEO, Abbott Laboratories — "The combination of our market-leading Xience drug-eluting stent platform, our growing IVUS and OCT intravascular imaging portfolio, and our advancing physiology assessment capabilities is creating an integrated coronary intervention ecosystem that gives interventional cardiologists the tools they need to achieve the best possible outcomes for their patients across the full spectrum of coronary artery disease complexity. Abbott is investing in next-generation bioresorbable scaffold technology development alongside our established metallic stent platforms — recognizing that the long-term future of coronary intervention may involve vessel restoration approaches that deliver optimal outcomes without a permanent metallic implant."
-
Joaquin Duato, CEO, Johnson & Johnson MedTech — "The cardiovascular device market remains one of the most strategically important and commercially attractive segments in medical technology — driven by the fundamental and growing clinical need to treat coronary artery disease more effectively and less invasively than surgical alternatives allow for the large and expanding global patient population affected by this condition. We continue to evaluate strategic portfolio opportunities in coronary intervention that would complement our existing cardiovascular technology capabilities and enable us to participate more fully in the growing coronary stents and structural heart markets."
Key Report Takeaways
-
North America dominates the Coronary Stents market, holding approximately 38.40% of global market share in 2025, driven by the highest per-capita interventional cardiology procedural rates globally, the most advanced cardiac catheterization laboratory infrastructure, the strongest commercial presence of leading device manufacturers including Abbott, Boston Scientific, and Medtronic, the most comprehensive reimbursement coverage for drug-eluting stent procedures, and the clinical culture of routine intravascular imaging and physiology-guided optimization that drives premium device utilization per procedure.
-
Asia Pacific is the fastest-growing regional market for coronary stents, projected to expand at a CAGR of approximately 8.10% from 2026 to 2033, driven by China's large and rapidly growing percutaneous coronary intervention procedure volumes, India's rapidly expanding private hospital cardiac catheterization laboratory capacity, Japan and South Korea's technologically sophisticated interventional cardiology practice culture, growing government healthcare investment across Southeast Asian markets, and the progressive development of local manufacturing capabilities in China and India that are improving device accessibility and affordability.
-
Drug-Eluting Stents are the dominant product type segment, accounting for approximately 74.60% of total market revenue in 2025, driven by their clinically demonstrated superiority over bare-metal stents in reducing in-stent restenosis rates, the progressive expansion of drug-eluting stent use across increasingly complex lesion types and high-risk patient populations, the continuous product platform improvement generating regular device upgrade cycles, and the strong clinical guideline support for drug-eluting stent use as the default coronary stent choice across virtually all interventional cardiology practice settings globally.
-
Hospitals are the dominant end-user segment, contributing approximately 68.20% of total market revenue in 2025, reflecting the concentration of high-volume, complex percutaneous coronary intervention procedures — including primary PCI for acute myocardial infarction and complex multivessel interventions requiring on-site cardiac surgery backup — within tertiary care hospital catheterization laboratory settings that combine the highest procedural volumes, the most advanced imaging and physiology guidance capabilities, and the most experienced interventional cardiology teams.
-
Acute Myocardial Infarction is the dominant application segment, accounting for approximately 43.80% of total market revenue in 2025, driven by the strong clinical guideline mandate for primary percutaneous coronary intervention with drug-eluting stent implantation as the gold-standard reperfusion treatment for ST-elevation myocardial infarction — generating large and time-sensitive coronary stent demand that is relatively insensitive to economic conditions, reimbursement constraints, and elective procedure deferral pressures that affect other PCI indications.
-
Bioresorbable Vascular Scaffolds are the fastest-growing product type segment, projected to expand at a CAGR of approximately 11.40% through 2033, driven by next-generation bioresorbable scaffold designs addressing the clinical limitations of early platforms, growing physician and patient interest in vessel restoration concepts that avoid the long-term vessel cage limitations of permanent metallic stents, and the progressive generation of clinical evidence supporting improved outcomes with newer-generation thin-strut bioresorbable scaffold technologies from companies including Abbott, Biotronik, and emerging Asian manufacturers.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 14.20 Billion |
| Market Size by 2025 | USD 7.80 Billion |
| Market Size by 2026 | USD 8.35 Billion |
| Market Growth Rate (2026–2033) | CAGR of 6.60% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Segments Covered | Product Type, Material, End User, Application |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Rising Coronary Artery Disease Prevalence, Expanding Interventional Cardiology Infrastructure in Emerging Markets, Drug-Eluting Stent Clinical Evidence Growth, and Aging Population Demographics Are the Core Pillars Driving the Coronary Stents Market*
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising global coronary artery disease prevalence driven by aging populations and lifestyle risk factors | ~35% | Global | Short to Long-term |
| Expanding cardiac catheterization laboratory infrastructure and interventional cardiology training in Asia Pacific and Latin America | ~28% | Asia Pacific, Latin America | Short to Long-term |
| Continuous drug-eluting stent platform improvement expanding complex PCI clinical indications | ~22% | North America, Europe, Asia Pacific | Short to Long-term |
| Growing adoption of physiology-guided and imaging-guided PCI protocols driving premium device utilization | ~15% | North America, Europe | Short to Medium-term |
The most powerful growth driver of the Coronary Stents market is the rising global burden of coronary artery disease — where aging population demographics, escalating metabolic risk factor prevalence including obesity and type 2 diabetes, and persistent tobacco use across developing economies are collectively expanding the patient population requiring coronary revascularization at rates that are expected to continue growing throughout the forecast period across virtually all global regions. The WHO's estimate that cardiovascular diseases cause approximately 17.9 million deaths annually — with coronary artery disease as the leading contributor — underscores the enormous and growing clinical need that drives fundamental demand for coronary stent implantation as the most common and minimally invasive coronary revascularization treatment modality. The drug-eluting stent platform improvement trajectory — driven by sustained clinical and engineering innovation by Abbott, Boston Scientific, Medtronic, and European and Asian manufacturers — has progressively expanded the range of lesion types, patient risk profiles, and clinical scenarios where percutaneous coronary intervention with stent implantation is clinically preferred over surgical coronary artery bypass grafting, growing the total addressable patient population and procedure volume for the Coronary Stents market with each successive generation of improved stent platform capabilities.
The transformative geographic market development opportunity in emerging economies — where rapidly expanding cardiac catheterization laboratory installation rates, growing interventional cardiology specialist workforce, and improving government reimbursement frameworks for percutaneous coronary intervention are generating rapid procedure volume growth from a relatively low baseline — represents the most commercially significant structural driver of global coronary stents market revenue growth over the forecast period. China's progression from approximately 0.4 million annual PCI procedures in 2010 to over 1.5 million procedures currently — an approximately fourfold volume increase over 15 years — illustrates the magnitude of market development potential that is being replicated at various stages across India, Southeast Asian markets, Latin America, and Middle East & Africa. The growing physician adoption of complex PCI techniques — including left main coronary artery bifurcation stenting, chronic total occlusion revascularization using drug-eluting stents, and multivessel intervention strategies guided by fractional flow reserve — is additionally expanding the per-case device utilization and revenue opportunity beyond conventional single-vessel de novo lesion stenting that historically defined the bulk of coronary stents market procedure volumes.

Restraints Impact Analysis
Drug-Eluting Stent Pricing Pressure from Local Manufacturer Competition in China, Dual Antiplatelet Therapy Compliance Challenges, Reimbursement Constraints in Cost-Sensitive Markets, and Surgical Bypass Competition for Complex Lesions Restraining Faster Coronary Stents Market Growth*
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Intense pricing pressure from local stent manufacturers in China and India eroding multinational margins | ~32% | Asia Pacific | Short to Long-term |
| Dual antiplatelet therapy compliance challenges limiting DES adoption in patients with bleeding risk or surgery needs | ~26% | Global | Short to Long-term |
| Reimbursement constraints and DRG pricing caps limiting drug-eluting stent adoption in cost-sensitive healthcare systems | ~24% | Europe, Latin America, Asia Pacific | Short to Medium-term |
| Competition from surgical coronary artery bypass grafting for complex multivessel and left main disease | ~18% | Global | Short to Long-term |
The most commercially impactful restraint on global Coronary Stents market revenue growth is the intense pricing pressure generated by domestic stent manufacturer competition in China — where the National Healthcare Security Administration's centralized volume-based procurement program has driven dramatic price reductions of 90% or more for drug-eluting stents within the Chinese public hospital market, severely compressing margins for both international manufacturers and Chinese domestic stent companies and creating a pricing benchmark that is progressively influencing reimbursement and procurement negotiations in other emerging economy markets. China's centralized volume procurement has fundamentally altered the commercial economics of the Chinese coronary stents market — transforming it from a premium device revenue opportunity into a high-volume, low-margin commodity market that effectively removes a significant portion of the world's largest procedure volume market from the premium DES revenue opportunity that international manufacturers previously enjoyed. This pricing compression dynamic is expected to persist and potentially intensify within China while also influencing procurement approaches in other major emerging economy markets including India, Brazil, and Southeast Asian countries where healthcare payers are studying the Chinese model as a potential cost control mechanism for medical device procurement.
Dual antiplatelet therapy compliance challenges — where the extended DAPT regimen required after drug-eluting stent implantation creates meaningful bleeding complications risk and surgical contraindications that limit DES use in patients requiring near-term non-cardiac surgery, those with high bleeding risk, and populations with historically poor medication adherence — continue to restrain drug-eluting stent adoption in certain patient subgroups and clinical settings. The development of newer-generation DES platforms with reduced polymer reactivity and shorter required DAPT durations has progressively mitigated this restraint — but the fundamental challenge of antiplatelet therapy management in complex, real-world patient populations with multiple comorbidities and medication polypharmacy remains a meaningful clinical constraint on the Coronary Stents market's growth rate. Competition from coronary artery bypass grafting surgery — particularly in complex multivessel coronary artery disease with diabetes, left main disease with high SYNTAX scores, and chronic total occlusions with unfavorable anatomy — continues to limit the expansion of percutaneous coronary intervention indications in patient subsets where current clinical evidence and guidelines continue to favor surgical revascularization.
Opportunities Impact Analysis
Bioresorbable Scaffold Technology Maturation, Emerging Market Infrastructure Development, Drug-Eluting Stent Indication Expansion for Complex Lesions, and Digital Health Integration Creating Significant Coronary Stents Market Opportunities*
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Next-generation bioresorbable scaffold clinical validation and commercial re-launch | ~30% | North America, Europe, Asia Pacific | Short to Long-term |
| Emerging market catheterization laboratory expansion and procedure volume growth | ~28% | Asia Pacific, Latin America, Middle East & Africa | Short to Long-term |
| Complex PCI indication expansion including CTO, left main, and multivessel stenting | ~25% | Global | Short to Long-term |
| AI-powered procedural guidance and post-stent monitoring platform development | ~17% | North America, Europe | Short to Medium-term |
The most technically significant commercial opportunity within the Coronary Stents market over the forecast period is the potential successful clinical validation and commercial re-emergence of next-generation bioresorbable vascular scaffold technology — where the well-documented clinical limitations of Abbott's first-generation Absorb bioresorbable scaffold, including elevated late scaffold thrombosis rates and technical deployment sensitivity, are being systematically addressed through next-generation thin-strut designs with improved radial strength, faster resorption kinetics, and optimized deployment protocols by Abbott, Biotronik, Elixir Medical, and several Chinese and Indian manufacturers. If next-generation bioresorbable scaffolds demonstrate non-inferiority or superiority to current-generation drug-eluting stents in adequately powered randomized clinical trials — which several are currently undergoing — their commercial adoption could create a significant and fast-growing new premium product segment within the coronary stents market that commands higher average selling prices and generates meaningful revenue growth beyond the base DES market trajectory. The vessel restoration concept — where the scaffold fully resorbs over 2–4 years leaving a healed, metal-free coronary artery that could be re-intervened upon without constraint, undergo positive vessel remodeling, and potentially restore vasomotion — addresses clinically meaningful limitations of permanent metallic stents that resonate strongly with interventional cardiologists, their patients, and healthcare payers focused on long-term outcomes.
The emerging and rapidly developing cardiac catheterization laboratory infrastructure across Asia Pacific, Latin America, and Middle East & Africa represents a commercially transformative opportunity for the Coronary Stents market — where the combination of large and growing coronary artery disease patient populations, rapidly increasing healthcare investment, trained interventional cardiology workforce development, and improving government reimbursement for PCI procedures is generating rapid procedural volume growth from an underpenetrated baseline. India's government initiatives to expand AIIMS and cardiac care center networks across Tier 2 and Tier 3 cities — combined with private hospital sector cardiac catheterization laboratory expansion — are generating rapidly growing PCI procedure volumes from a population base of 1.4 billion people with high and growing coronary artery disease prevalence. The integration of AI-powered procedural guidance platforms — where real-time intravascular imaging AI, FFR-CT analysis, and post-procedure outcome prediction algorithms collectively improve procedure quality, reduce complications, and enable more consistent optimal stent deployment — represents a digital health opportunity that leading device manufacturers are investing in to create integrated coronary intervention ecosystem platforms that differentiate beyond device hardware alone and generate software and platform revenue streams alongside traditional stent device revenues.

Segment Analysis
By Product Type
Drug-Eluting Stents Dominate the Coronary Stents Market While Bioresorbable Vascular Scaffolds Drive the Fastest Segment Growth Globally*
Drug-eluting stents hold the dominant revenue position in the Coronary Stents market, accounting for approximately 74.60% of total global market revenue in 2025, driven by their clinically demonstrated superiority over bare-metal stents in reducing in-stent restenosis through the controlled local release of antiproliferative agents including everolimus, zotarolimus, and sirolimus from drug-carrying polymer coatings that inhibit neointimal hyperplasia within the stented coronary artery segment. The drug-eluting stent segment benefits from continuous platform innovation — where each successive generation of thin-strut cobalt-chromium and platinum-chromium DES designs, biodegradable polymer coating chemistries, and optimized drug release kinetics delivers incremental but clinically meaningful improvements in deliverability, acute procedural success rates, and late clinical outcomes that drive regular device upgrade cycles among interventional cardiologists seeking the best stent performance for complex lesion subsets. North America generates the largest drug-eluting stent segment revenue — driven by the highest premium DES utilization rates, routine adoption of intravascular imaging-guided stent optimization, and the strongest commercial presence of Abbott's Xience, Boston Scientific's Synergy, and Medtronic's Resolute Onyx platforms — with key companies collectively generating large and growing North American drug-eluting stent revenues underpinned by strong clinical evidence bases and comprehensive commercial infrastructures. Europe is the second-largest drug-eluting stent market, driven by guideline adoption of DES for virtually all PCI indications and strong adoption of novel thin-strut ultrathin DES platforms from companies including Biotronik and Terumo across major European interventional cardiology centers.
Bioresorbable vascular scaffolds represent the fastest-growing product type within the coronary stents market — projected to expand at a CAGR of approximately 11.40% through 2033 — driven by next-generation scaffold designs that are addressing the strut thickness, thrombogenicity, and deployment sensitivity limitations of first-generation bioresorbable platforms while preserving the conceptual vessel restoration advantages of fully bioresorbable implants that offer a permanent metal-free coronary artery after complete scaffold resorption. The bioresorbable scaffold segment is experiencing renewed commercial and clinical investment — with Abbott advancing its next-generation Absorb GT1 improvements, Biotronik investigating its Magmaris magnesium-based resorbable scaffold, Elixir Medical advancing its DESolve scaffold platform, and multiple Chinese and Indian manufacturers developing bioresorbable scaffold products for their domestic markets — collectively generating a growing commercial pipeline that is expected to deliver several next-generation product launches during the forecast period. Asia Pacific is the fastest-growing region for bioresorbable scaffold research and commercial development — driven by Chinese regulatory agency interest in bioresorbable scaffold innovation, domestic manufacturer investment in proprietary bioresorbable scaffold platforms, and the strong physician interest in vessel restoration concepts among Asian interventional cardiologists managing younger patient populations where long-term vessel freedom from permanent metallic implants is particularly valued.
By Application
Acute Myocardial Infarction Leads the Coronary Stents Market While Chronic Total Occlusion Applications Register the Fastest Growth Driven by Expanding Complex PCI Adoption*
Acute myocardial infarction holds the dominant application position in the Coronary Stents market, accounting for approximately 43.80% of total market revenue in 2025, driven by the strong and guideline-mandated clinical indication for primary percutaneous coronary intervention with drug-eluting stent implantation as the gold-standard reperfusion treatment for ST-elevation myocardial infarction — generating large and time-critical coronary stent demand that is relatively insensitive to economic conditions and elective procedure deferral pressures that affect other PCI application segments. The AMI application segment benefits from the non-discretionary nature of emergency cardiac intervention demand — where the clinical urgency of restoring coronary blood flow in STEMI patients creates mandatory stent utilization that generates stable and growing revenue from the large and persistent global burden of acute coronary syndromes. North America and Europe generate the largest AMI application segment revenues — driven by mature primary PCI networks with rapid door-to-balloon time capabilities, comprehensive reimbursement for emergency PCI procedures, and well-established STEMI intervention protocols — with Abbott, Boston Scientific, and Medtronic collectively supplying the majority of drug-eluting stents used in STEMI primary PCI procedures across these regions. The AMI application segment within the coronary stents market is expected to sustain steady growth throughout the forecast period driven by both the growing underlying prevalence of coronary artery disease generating higher STEMI event rates and the geographic expansion of primary PCI networks in emerging economies that are progressively achieving the catheterization laboratory density and response time capabilities required for systematic primary PCI delivery.
Chronic total occlusion and complex lesion applications represent the fastest-growing application segment within the coronary stents market — driven by the progressive adoption of advanced CTO percutaneous coronary intervention techniques by a growing community of dedicated CTO operators who have developed specialized skills in retrograde and antegrade wire escalation approaches, dissection-reentry techniques, and complex wire manipulation strategies that enable successful CTO revascularization with drug-eluting stent implantation in a growing proportion of previously unrevascularized occluded coronary segments. The CTO PCI segment is experiencing rapid procedural volume growth in North America, Europe, and Asia Pacific — driven by growing evidence from registries and randomized trials supporting improved patient symptoms, functional capacity, and potentially survival with successful CTO revascularization, combined with the progressive development of dedicated CTO PCI training programs, specialized CTO operator networks, and advanced guidewire and microcatheter technologies that are improving CTO procedural success rates and safety profiles. Japan has historically led the world in CTO PCI technique development and adoption — with Japanese interventional cardiologists developing many of the key techniques and device technologies that have enabled the global expansion of complex CTO revascularization — and continues to generate premium-tier coronary stent utilization from high-volume CTO and complex lesion PCI practices that drive above-average per-case device revenues and represent a commercially attractive application segment for leading coronary stent manufacturers.

Regional Insights
North America
North America Leads the Global Coronary Stents Market at 38.40% Revenue Share, Driven by the Highest Interventional Cardiology Procedural Rates, Premium Drug-Eluting Stent Adoption, and Strongest Commercial Infrastructure*
North America holds the dominant position in the global Coronary Stents market, accounting for approximately 38.40% of total global market revenue in 2025, and is projected to sustain market leadership at a CAGR of approximately 6.10% from 2026 to 2033. The United States generates the overwhelming majority of North American coronary stents revenue — driven by the highest per-capita percutaneous coronary intervention procedural rates globally, the most comprehensive commercial insurance and Medicare reimbursement coverage for drug-eluting stent procedures, the routine clinical adoption of intravascular imaging-guided and physiology-guided stent optimization protocols that drive premium device utilization per case, and the dominant commercial presence of Abbott Vascular, Boston Scientific, and Medtronic — whose combined drug-eluting stent portfolios including Xience, Synergy, and Resolute Onyx respectively command the majority of US coronary stent implantation volume. North America additionally benefits from the strongest clinical research investment in coronary intervention technology — where NIH-funded and industry-sponsored cardiovascular clinical trials regularly generate new evidence supporting broader and more complex drug-eluting stent applications that progressively expand the US coronary stent market revenue opportunity.
Canada contributes a meaningful share of North American coronary stents revenue — through its well-developed public hospital cardiac catheterization laboratory network, growing adoption of complex PCI techniques including CTO revascularization, and progressive provincial health insurance coverage expansion for newer-generation drug-eluting stent platforms. The United States market is additionally experiencing meaningful revenue growth from the expanding adoption of AI-powered intravascular imaging and physiology guidance systems — including Philips' IntelliSpace platform, Boston Scientific's ACIST and Avvigo intravascular imaging capabilities, and Abbott's emerging digital catheterization laboratory workflow tools — that are increasing per-procedure device utilization and premium stent adoption rates among the growing community of imaging-guided PCI practitioners across American interventional cardiology centers.
Asia Pacific
Asia Pacific Is the Fastest-Growing Coronary Stents Market at 8.10% CAGR, Driven by China's Large PCI Procedure Volumes, India's Rapidly Expanding Cardiac Infrastructure, and Government Healthcare Investment*
Asia Pacific is the fastest-growing regional segment in the Coronary Stents market, projected to expand at a CAGR of approximately 8.10% from 2026 to 2033 — the highest regional growth rate globally. China represents the most commercially significant and volume-generating Asia Pacific coronary stents market — performing over 1.5 million PCI procedures annually across a large and rapidly growing catheterization laboratory network — although the National Healthcare Security Administration's centralized volume-based procurement program has dramatically compressed coronary stent selling prices within the Chinese public hospital market, fundamentally reshaping the commercial economics for both international device manufacturers and domestic Chinese stent producers including MicroPort Medical, Lepu Medical, and JW Medical Systems that collectively dominate Chinese domestic market stent volume following the procurement price reductions. Key international manufacturers including Abbott, Boston Scientific, and Medtronic continue to maintain commercial presences in China through local partnerships and manufacturing investments — focusing increasingly on premium, complex PCI, and private hospital channels where pricing dynamics are more favorable than in the public procurement system.
India represents the most commercially exciting emerging opportunity within the Asia Pacific Coronary Stents market — where rapidly expanding private hospital cardiac catheterization laboratory networks, a growing and highly trained interventional cardiology specialist community, government-sponsored cardiac care programs expanding PCI access to previously underserved populations, and a large and growing coronary artery disease patient population are collectively generating rapid procedure volume growth that is expected to make India one of the world's largest coronary stent procedural volume markets during the forecast period. Key companies including Abbott, Boston Scientific, Medtronic, and local Indian manufacturers including Sahajanand Medical Technologies and Translumina Therapeutics are competing actively for Indian market share — with local manufacturers deploying significant cost and accessibility advantages in price-sensitive public and semi-urban hospital settings while international manufacturers target premium urban private hospital and complex PCI procedure segments. Japan and South Korea contribute premium-tier Asia Pacific coronary stents revenue — driven by sophisticated interventional cardiology practices that routinely employ intravascular imaging guidance, complex CTO PCI, and multivessel intervention strategies that generate above-average per-case device utilization and premium stent adoption.
Top Key Players
-
Abbott Laboratories (United States)
-
Boston Scientific Corporation (United States)
-
Medtronic plc (Ireland)
-
Biotronik SE & Co. KG (Germany)
-
Terumo Corporation (Japan)
-
MicroPort Scientific Corporation (China)
-
Lepu Medical Technology Co. Ltd. (China)
-
Sahajanand Medical Technologies Pvt. Ltd. (India)
-
JW Medical Systems Ltd. (China)
-
B. Braun Melsungen AG (Germany)
-
Translumina Therapeutics LLP (India)
-
Elixir Medical Corporation (United States)
Recent Developments
-
Abbott Laboratories (2025) — Advanced the clinical development of its next-generation bioresorbable vascular scaffold program while reporting continued strong commercial performance for its Xience drug-eluting stent platform globally — with Xience maintaining its market-leading position in the United States, Europe, and multiple Asia Pacific markets — and expanding its intravascular imaging portfolio through the commercial launch of enhanced ILUMIEN INTELLIMAP OPTIS OCT-guided PCI system capabilities that integrate AI-powered automated stent optimization recommendations with real-time procedural guidance for interventional cardiologists.
-
Boston Scientific Corporation (2025) — Reported strong global commercial growth for its Synergy biodegradable polymer everolimus-eluting stent platform and expanded its coronary intervention portfolio through strategic investments in intravascular imaging and physiology-guided PCI workflow capabilities — while advancing the clinical development of its next-generation coronary stent and bioresorbable scaffold pipeline programs and reporting growing complex PCI procedure volume adoption of its ACIST intravascular imaging systems across major US and European cardiac catheterization laboratory customers.
-
Medtronic plc (2025) — Continued to advance the global commercial rollout of its Resolute Onyx drug-eluting stent platform featuring a proprietary BioLinx polymer coating designed for extended elution and excellent biocompatibility — while reporting growing adoption of its Resolute Onyx DES for complex lesion subsets including small vessels and bifurcations — and pursuing strategic portfolio expansion opportunities in coronary intervention digital workflow and AI-powered guidance platforms that would complement its established stent device business with higher-growth digital health capabilities.
-
MicroPort Medical (2024) — Successfully maintained its strong competitive position within the Chinese domestic coronary stents market following the implementation of centralized volume-based procurement — adapting its commercial strategy to optimize volume capture within the procurement program while simultaneously developing next-generation premium DES and bioresorbable scaffold product candidates targeting the Chinese private hospital and international export markets — and reporting growing international market revenues from its coronary stent portfolio in emerging economy markets across Asia Pacific, Latin America, and Middle East & Africa where its competitive pricing advantages drive adoption.
-
Biotronik (2025) — Advanced the clinical development of its Magmaris magnesium-based bioresorbable scaffold program — reporting continued long-term clinical follow-up data demonstrating favorable safety and efficacy outcomes consistent with permanent metallic DES benchmarks — while simultaneously reporting strong commercial performance for its Orsiro thin-strut sirolimus-eluting drug-eluting stent platform in European and international markets where its ultra-thin strut design and biodegradable polymer coating have established a premium clinical reputation among interventional cardiologists specializing in complex and small-vessel coronary intervention.
Market Trends
Ultrathin Strut Drug-Eluting Stent Platform Adoption Accelerating, Physiology-Guided and Imaging-Guided PCI Protocols Becoming Standard of Care, and Bioresorbable Scaffold Technology Experiencing a Second Clinical Generation Renaissance Are the Most Commercially Defining Trends Reshaping the Coronary Stents Market Through 2033*
The most commercially impactful product trend reshaping the Coronary Stents market is the progressive shift toward ultrathin strut drug-eluting stent designs — where advances in cobalt-chromium and platinum-chromium alloy metallurgy and manufacturing precision have enabled strut thickness reductions to 60–80 microns in leading current-generation DES platforms, compared to 140+ micron strut thicknesses in first-generation DES designs — with clinical trial evidence from BIOSCIENCE, BIOFLOW, and TALENT studies demonstrating that ultrathin strut platforms deliver superior target lesion failure outcomes compared to thicker-strut DES comparators across both simple and complex coronary lesion subsets. This ultrathin strut design trend is simultaneously improving deliverability through complex tortuous and calcified coronary anatomies, reducing the mechanical footprint within the coronary artery lumen, and enabling more complete endothelialization that contributes to the improved clinical outcomes and reduced DAPT duration requirements associated with the best current-generation DES platforms. The routine clinical adoption of physiology-guided PCI using FFR and iFR — combined with increasingly widespread use of OCT and IVUS intravascular imaging for stent sizing, deployment optimization, and post-dilation guidance — is progressively elevating the standard of interventional cardiology practice in a way that benefits premium stent device manufacturers whose products are most compatible with image-optimized deployment protocols and whose commercial strategies include integrated catheterization laboratory workflow platforms.
The second-generation renaissance of bioresorbable scaffold technology — where next-generation devices are specifically designed to address the strut thickness, resorption kinetics, and deployment protocol limitations that contributed to the adverse clinical outcomes observed in the first-generation Absorb GT1 clinical trials — represents the most commercially transformative medium-term trend for the Coronary Stents market if clinical validation studies currently underway demonstrate non-inferiority to permanent DES benchmarks. Biotronik's Magmaris magnesium resorbable scaffold — which uses a magnesium alloy rather than the PLLA polymer of first-generation bioresorbable scaffolds, enabling faster resorption over 12 months rather than the 2–3 year resorption timeline of polymer scaffolds — has generated encouraging early and medium-term clinical data that is building physician confidence in the second-generation bioresorbable scaffold concept. The strong and growing clinician and patient preference for vessel restoration implant concepts — particularly in younger patients with de novo coronary lesions where long-term vessel freedom from permanent metallic implants after complete scaffold resorption is valued for future intervention flexibility, vessel remodeling potential, and quality of life considerations — positions bioresorbable scaffolds as a commercially important growth driver for the coronary stents market across the full forecast period through 2033.
Segments Covered in the Report
By Product Type:
-
Drug-Eluting Stents
-
Cobalt-Chromium Drug-Eluting Stents
-
Platinum-Chromium Drug-Eluting Stents
-
Stainless Steel Drug-Eluting Stents
-
Others
-
-
Bare-Metal Stents
-
Cobalt-Chromium Bare-Metal Stents
-
Stainless Steel Bare-Metal Stents
-
-
Bioresorbable Vascular Scaffolds
-
Others
By Material:
-
Cobalt-Chromium
-
Stainless Steel
-
Platinum-Chromium
-
Nitinol
-
Others
By End User:
-
Hospitals
-
Ambulatory Surgical Centers
-
Cardiac Catheterization Laboratories
-
Others
By Application:
-
Acute Myocardial Infarction
-
Stable Angina
-
Unstable Angina
-
Chronic Total Occlusion
-
Others
By Region:
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the Coronary Stents market and what is its projected value by 2033?
Answer: The Coronary Stents market was valued at USD 7.80 billion in 2025 and is projected to reach USD 14.20 billion by 2033, growing at a CAGR of 6.60% from 2026 to 2033. This steady growth is underpinned by rising global coronary artery disease prevalence, expanding interventional cardiology infrastructure in Asia Pacific and Latin America, continuous drug-eluting stent platform innovation, and growing adoption of complex PCI procedures across a progressively broader patient population.
Question 2: What are the different types of coronary stents and which is most commonly used?
Answer: The main types of coronary stents include drug-eluting stents, bare-metal stents, and bioresorbable vascular scaffolds — with drug-eluting stents representing the dominant product type accounting for approximately 74.60% of total Coronary Stents market revenue in 2025 due to their clinically proven superiority in reducing in-stent restenosis. Drug-eluting stents release antiproliferative drugs such as everolimus or sirolimus from polymer coatings on the stent surface to inhibit the smooth muscle cell proliferation that caused restenosis in earlier bare-metal stent designs.
Question 3: Which region dominates the Coronary Stents market and which is growing the fastest?
Answer: North America leads the Coronary Stents market with approximately 38.40% of global revenue in 2025 — driven by the highest per-capita PCI procedural rates globally, comprehensive reimbursement coverage, and the dominant commercial presence of Abbott, Boston Scientific, and Medtronic. Asia Pacific is the fastest-growing region at approximately 8.10% CAGR through 2033 — driven by China's large procedure volumes, India's rapidly expanding cardiac care infrastructure, and growing government healthcare investment across Southeast Asian markets.
Question 4: What is a drug-eluting stent and how does it differ from a bare-metal stent?
Answer: A drug-eluting stent is a metallic coronary stent coated with a polymer that elutes an antiproliferative drug — such as everolimus, zotarolimus, or sirolimus — locally into the arterial wall after implantation to inhibit the neointimal tissue growth that previously caused bare-metal stent restenosis requiring repeat revascularization. In the Coronary Stents market, drug-eluting stents have largely replaced bare-metal stents as the default stent choice because multiple large randomized clinical trials have demonstrated their superiority in reducing clinically driven target lesion revascularization rates across virtually all coronary lesion types and patient populations.
Question 5: What future technologies are expected to drive Coronary Stents market growth through 2033?
Answer: The most commercially significant future technology driver for the Coronary Stents market is next-generation bioresorbable vascular scaffold technology — where fully bioresorbable stent platforms that completely dissolve within 2–4 years of implantation could offer vessel restoration advantages including metal-free coronary arteries, restored vasomotion, and re-intervention flexibility that permanent metallic DES cannot provide, potentially creating a large new premium product segment if clinical trials validate their safety and efficacy. AI-powered intravascular imaging analysis platforms, physiology-guided PCI workflow systems, and digital catheterization laboratory integration technologies represent additional important innovation drivers that will expand the per-procedure device utilization and clinical value of the coronary stents market ecosystem beyond stent hardware revenue alone.
