Contract Sterilization Services Market Overview
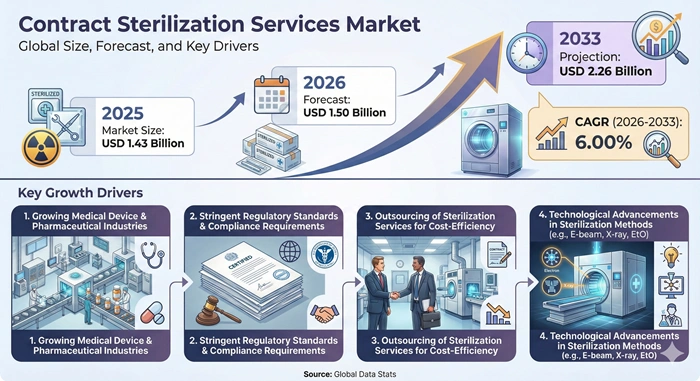
The global contract sterilization services market size is valued at USD 1.43 billion in 2025 and is predicted to increase from USD 1.50 billion in 2026 to approximately USD 2.26 billion by 2033, growing at a CAGR of 6.00% from 2026 to 2033.
Contract sterilization services represent a crucial segment within the healthcare and life sciences industry where manufacturers outsource terminal sterilization processes to specialized third-party providers. These services utilize advanced sterilization modalities including ethylene oxide, gamma irradiation, electron beam, and steam sterilization to ensure medical devices, pharmaceutical products, and surgical instruments meet stringent safety standards. The contract sterilization services market continues expanding as healthcare facilities and medical device manufacturers increasingly recognize the operational benefits of outsourcing sterilization processes rather than maintaining costly in-house infrastructure.
The rising demand stems from multiple factors including the growing volume of surgical procedures worldwide, stringent regulatory requirements for sterilization validation, and the increasing complexity of medical devices requiring specialized sterilization protocols. Healthcare-associated infections remain a persistent concern, driving hospitals and medical device companies to partner with expert sterilization service providers who maintain state-of-the-art facilities and regulatory compliance expertise.
AI Impact on the Contract Sterilization Services Industry
Artificial Intelligence and Robotics are Transforming Sterilization Efficiency, Quality Assurance, and Predictive Maintenance Across the Contract Sterilization Services Market
Artificial intelligence is revolutionizing the contract sterilization services market by introducing automation, real-time monitoring capabilities, and predictive analytics that enhance operational efficiency and patient safety outcomes. AI-driven systems enable sterilization facilities to optimize cycle parameters based on load composition, reducing processing times by up to 35 percent while maintaining consistent sterility assurance levels. Computer vision technology integrated into sterilization workflows detects instrument defects, verifies tray assembly accuracy, and ensures compliance with established protocols, significantly reducing human error rates that previously compromised sterilization quality.
Machine learning algorithms analyze historical sterilization data to predict equipment failures before they occur, enabling proactive maintenance scheduling that reduces downtime by approximately 25 percent and extends equipment lifespan. AI-powered inventory management systems track sterilization capacity in real-time, optimizing workflow distribution across multiple facilities and preventing bottlenecks during peak demand periods. These technological advancements position AI as a fundamental driver of competitive advantage within the contract sterilization services market, with early adopters reporting throughput improvements of 35 to 50 percent in partially automated processing lines. As regulatory frameworks evolve to accommodate AI-enabled medical device processing, the integration of artificial intelligence will become increasingly central to service provider differentiation and market leadership.
Growth Factors
Rising Surgical Volumes, Outsourcing Trends, and Regulatory Stringency Fuel Expansion Across the Contract Sterilization Services Market
The contract sterilization services market experiences robust growth driven by demographic shifts toward aging populations that require increased surgical interventions and chronic disease management. Global healthcare expenditure continues rising, particularly in emerging economies where expanding medical infrastructure creates substantial demand for validated sterilization services. Medical device manufacturers face mounting pressure to comply with evolving regulatory standards including the European Union's Medical Device Regulation and FDA sterilization validation requirements, prompting companies to partner with specialized contract sterilization providers who possess the technical expertise and documentation systems necessary for regulatory compliance.
Outsourcing sterilization processes allows medical device companies and pharmaceutical manufacturers to convert fixed capital expenditures into variable operating costs, improving financial flexibility while accessing cutting-edge sterilization technologies without major infrastructure investments. Small and medium-sized enterprises particularly benefit from this model, gaining access to gamma irradiation, electron beam, and ethylene oxide sterilization capabilities that would be prohibitively expensive to develop internally. The contract sterilization services market also benefits from the proliferation of heat-sensitive medical devices including advanced polymers, biologics, and electronic surgical instruments that require specialized low-temperature sterilization methods. These convergent factors position the market for sustained expansion throughout the forecast period as healthcare quality standards intensify and medical device complexity increases.

Market Outlook
Strategic Partnerships, Capacity Expansion, and Sustainable Sterilization Methods Define the Future Trajectory of the Contract Sterilization Services Market
The contract sterilization services market outlook remains exceptionally positive as industry leaders invest in capacity expansion and next-generation sterilization technologies to meet accelerating demand. Service providers increasingly focus on establishing facilities near major medical device manufacturing hubs in Asia-Pacific and Latin America, reducing transportation costs and turnaround times for customers operating in these high-growth regions. Strategic partnerships between contract sterilization providers and medical device original equipment manufacturers are becoming more prevalent, with long-term agreements securing dedicated sterilization capacity and collaborative development of application-specific sterilization protocols.
Environmental sustainability emerges as a differentiating factor within the contract sterilization services market as regulatory agencies implement stricter emissions standards for ethylene oxide facilities. Forward-thinking providers invest in alternative sterilization modalities including vaporized hydrogen peroxide, nitrogen dioxide, and supercritical carbon dioxide systems that deliver effective sterilization with reduced environmental impact. The market will witness continued consolidation as larger service providers acquire regional facilities to expand geographic coverage and service offerings. Regulatory harmonization initiatives across international markets will simplify cross-border sterilization operations, enabling global medical device companies to standardize sterilization protocols across multiple manufacturing sites. These strategic developments position the contract sterilization services market for transformation into an increasingly integrated, technology-driven ecosystem.
Expert Speaks
-
Dan Carestio, President and CEO of STERIS plc, emphasized the company's focus on driving operational excellence and meeting customer demand across healthcare and life sciences segments, highlighting strong service revenue growth and continued investment in sterilization infrastructure to support expanding medical device manufacturing.
-
Michael Petras Jr., CEO of Sotera Health Company, outlined strategic priorities focused on winning growth markets and teaming with high-performing medtech and pharmaceutical companies, noting plans for 5-7% organic growth through investments in new facilities and cobalt-60 development for sterilization applications.
-
Industry analysts from leading market research firms indicate that contract sterilization providers leveraging automation, AI-driven monitoring, and sustainable sterilization technologies will capture disproportionate market share as customers prioritize partners demonstrating regulatory excellence and environmental responsibility.
Key Report Takeaways
-
North America leads the contract sterilization services market, commanding the largest regional market share due to the concentrated presence of medical device manufacturers, pharmaceutical companies, and stringent regulatory enforcement driving demand for validated sterilization services
-
Asia-Pacific represents the fastest-growing region with the highest CAGR during the forecast period, propelled by expanding healthcare infrastructure investments, rising medical device manufacturing activity, and increasing adoption of international quality standards across China, India, and Southeast Asian nations
-
Medical device companies constitute the largest end-user segment, utilizing contract sterilization services most extensively for surgical instruments, implantable devices, and single-use medical products requiring terminal sterilization before market distribution
-
Ethylene oxide sterilization dominates the method segment, contributing the highest market share (approximately 42-45%) and revenue due to its effectiveness for heat-sensitive materials, compatibility with complex device geometries, and long-established validation protocols
-
Off-site sterilization services maintain leadership in the delivery mode segment, offering centralized processing efficiencies and economies of scale that enable competitive pricing while maintaining quality standards for medical device manufacturers
-
The gamma irradiation segment will experience the fastest growth among sterilization methods during the forecast period, projected to expand at a CAGR exceeding 7% driven by its environmental advantages, deep penetration capabilities, and suitability for high-volume processing of packaged medical devices
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 2.26 Billion |
| Market Size by 2025 | USD 1.43 Billion |
| Market Size by 2026 | USD 1.50 Billion |
| Market Growth Rate from 2026 to 2033 | CAGR of 6.00% |
| Dominating Region | North America |
| Fastest Growing Region | Asia-Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2033 |
| Segments Covered | Service Type, Sterilization Method, Delivery Mode, End-User, Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Increasing Surgical Procedures and Healthcare Infrastructure Expansion Accelerate Contract Sterilization Services Market Growth
| Impact Factor | Details |
|---|---|
| Approximate Impact on CAGR Forecast | +2.5% to +3.0% |
| Geographic Relevance | Global, particularly strong in Asia-Pacific and Latin America |
| Impact Timeline | Immediate and sustained through 2033 |
The contract sterilization services market benefits tremendously from the escalating volume of surgical procedures performed globally as aging demographics and chronic disease prevalence drive healthcare utilization rates upward. Countries with rapidly developing healthcare systems require validated sterilization services to meet international medical device safety standards, creating substantial opportunities for contract service providers establishing regional facilities. Hospitals and ambulatory surgical centers face increasing pressure to prevent healthcare-associated infections, mandating rigorous sterilization protocols for all reusable surgical instruments and implantable devices that contact patient tissue.
Medical device manufacturers launching innovative products face compressed development timelines and increasingly complex regulatory pathways requiring extensive sterilization validation documentation. Partnering with established contract sterilization providers allows these companies to accelerate market entry while leveraging specialized expertise in sterilization process development and regulatory submissions. The contract sterilization services market also gains momentum from pharmaceutical companies expanding biologic drug production, which necessitates sterile manufacturing environments and validated sterilization of production equipment. These combined factors create powerful tailwinds driving sustained market expansion throughout the forecast period.

Restraints Impact Analysis
Environmental Regulations and High Capital Requirements Present Challenges for Contract Sterilization Services Market Growth
| Impact Factor | Details |
|---|---|
| Approximate Impact on CAGR Forecast | -1.0% to -1.5% |
| Geographic Relevance | North America and Europe primarily |
| Impact Timeline | Ongoing through 2030, potential moderation afterward |
The contract sterilization services market faces significant headwinds from increasingly stringent environmental regulations targeting ethylene oxide emissions, which remain a primary sterilization method for heat-sensitive medical devices. Regulatory agencies in the United States and European Union have implemented stricter emissions limits and community notification requirements that increase compliance costs and extend permitting timelines for new facility development. Several contract sterilization facilities have faced temporary closures or operational restrictions due to environmental concerns, creating capacity constraints that temporarily disrupted medical device supply chains.
Establishing contract sterilization facilities requires substantial capital investment ranging from tens to hundreds of millions of dollars depending on sterilization modalities offered, facility size, and regulatory compliance infrastructure. These high entry barriers limit market access for new competitors and constrain capacity expansion among existing providers facing capital allocation priorities. The contract sterilization services market also encounters challenges from supply chain disruptions affecting cobalt-60 availability for gamma irradiation and raw material costs for facility construction. Despite these restraints, the fundamental demand drivers for sterilization services remain robust, ensuring continued market growth albeit at moderated rates reflecting these operational and regulatory challenges.
Opportunities Impact Analysis
Emerging Markets and Alternative Sterilization Technologies Create Significant Growth Opportunities in the Contract Sterilization Services Market
| Impact Factor | Details |
|---|---|
| Approximate Impact on CAGR Forecast | +1.5% to +2.0% |
| Geographic Relevance | Asia-Pacific, Latin America, Middle East |
| Impact Timeline | Accelerating from 2027 through 2033 |
The contract sterilization services market presents substantial untapped opportunities in emerging economies where medical device manufacturing activity is rapidly expanding but local sterilization infrastructure remains underdeveloped. Countries including India, China, Vietnam, and Mexico are establishing themselves as global medical device production hubs, creating immediate demand for validated contract sterilization services that meet international regulatory standards. Service providers establishing first-mover advantages in these high-growth markets can secure long-term partnerships with medical device manufacturers seeking reliable regional sterilization capacity.
Development of environmentally sustainable sterilization technologies represents another significant opportunity as medical device companies seek alternatives to traditional ethylene oxide sterilization facing regulatory scrutiny. Innovative modalities including vaporized hydrogen peroxide, supercritical carbon dioxide, and nitrogen dioxide sterilization offer effective microbial inactivation with reduced environmental impact, positioning early adopters as preferred partners for environmentally conscious customers. The contract sterilization services market can also expand service offerings beyond basic sterilization to include validation consulting, regulatory submission support, and comprehensive sterility assurance programs that create higher value relationships and improved customer retention. These strategic opportunities enable forward-thinking service providers to differentiate their offerings and capture disproportionate market share during the forecast period.

Segment Analysis
By Sterilization Method
Ethylene Oxide Sterilization Maintains Market Leadership Through Versatility and Established Validation for Heat-Sensitive Medical Devices
Ethylene oxide sterilization dominates the contract sterilization services market, commanding approximately 42-45% market share in 2025 and projected to maintain leadership throughout the forecast period with steady CAGR growth of 5.2-5.5%. This sterilization method remains indispensable for processing heat-sensitive medical devices including catheters, surgical kits, implantable devices, and complex instruments containing electronic components or polymeric materials that cannot withstand high-temperature steam sterilization. North America represents the largest regional market for ethylene oxide sterilization services, driven by the concentrated presence of major medical device manufacturers and well-established regulatory frameworks governing sterilization validation. Leading contract sterilization providers including STERIS, Sterigenics (Sotera Health), and Getinge operate extensive ethylene oxide sterilization networks across the United States with facilities strategically located near medical device manufacturing clusters.
The segment faces regulatory challenges from environmental agencies implementing stricter emissions controls, prompting service providers to invest in advanced abatement systems and process modifications that reduce ethylene oxide releases while maintaining sterilization efficacy. Despite these headwinds, the contract sterilization services market continues relying heavily on ethylene oxide due to its deep material penetration, compatibility with diverse packaging configurations, and decades of validated performance data supporting regulatory submissions. European markets show particular strength in ethylene oxide demand as medical device companies navigate Medical Device Regulation requirements necessitating robust sterilization validation documentation. Asia-Pacific emerges as a high-growth region for ethylene oxide sterilization as contract service providers establish facilities supporting the region's expanding medical device manufacturing sector. The segment benefits from continuous process improvements including cycle optimization algorithms and enhanced monitoring technologies that improve throughput while reducing environmental impact.
Gamma Irradiation Sterilization Experiences Fastest Growth Driven by Environmental Benefits and High-Volume Processing Capabilities
Gamma irradiation represents the fastest-growing segment within the contract sterilization services market, projected to expand at a robust CAGR exceeding 7% during 2026-2033 as medical device manufacturers increasingly adopt this environmentally friendly sterilization modality. Gamma sterilization utilizes cobalt-60 isotopes to deliver penetrating ionizing radiation that effectively inactivates microorganisms throughout packaged medical devices without chemical residues or temperature elevation. The method particularly suits high-volume disposable medical device production including syringes, wound care products, surgical gowns, and single-use diagnostic kits that constitute rapidly growing market segments. Major contract sterilization providers operate dedicated gamma irradiation facilities across North America and Europe, with Sotera Health (through its Sterigenics brand) maintaining the largest global gamma sterilization network with strategic facilities positioned near major medical device manufacturing regions.
Asia-Pacific demonstrates exceptional growth potential for gamma irradiation services as contract providers invest in new facilities supporting the region's emerging role as a global medical device production center, particularly in China, India, and Southeast Asian nations. The contract sterilization services market benefits from gamma sterilization's consistent dose delivery, predictable validation parameters, and continuous processing capabilities that enable rapid turnaround times critical for just-in-time medical device manufacturing operations. Leading companies in this segment include BGS Beta-Gamma-Service operating extensive European capacity, STERIS with integrated gamma facilities, and regional providers expanding capacity in Latin America and Asia-Pacific. Regulatory acceptance of gamma sterilization across all major markets, combined with its environmental advantages over chemical methods, positions this segment for sustained above-market growth throughout the forecast period as sustainability considerations increasingly influence medical device manufacturer sterilization method selection.

Regional Insights
North America
Established Medical Device Industry and Regulatory Rigor Position North America as the Dominant Contract Sterilization Services Market
North America maintains commanding leadership in the contract sterilization services market, accounting for approximately 42-45% of global market share in 2025 and projected to sustain dominance throughout the forecast period with steady CAGR growth of 5.5-6.0%. The region's market strength derives from the concentrated presence of global medical device manufacturers including Medtronic, Johnson & Johnson, Boston Scientific, and Abbott Laboratories that outsource substantial sterilization volumes to specialized contract providers. The United States hosts the world's most extensive contract sterilization infrastructure with major facilities operated by STERIS, Sterigenics (Sotera Health), Getinge, and E-BEAM Services strategically positioned near medical device manufacturing clusters in the Midwest, Southeast, and California regions.
Stringent regulatory oversight by the FDA drives medical device companies toward partnership with established contract sterilization providers possessing comprehensive validation expertise and documented quality systems meeting 21 CFR Part 820 requirements. The contract sterilization services market in North America benefits from high surgical procedure volumes, advanced healthcare infrastructure, and strong infection prevention protocols that mandate rigorous sterilization for all medical devices contacting sterile body tissues. Leading regional players invest continuously in capacity expansion and technology upgrades including automated handling systems, real-time monitoring platforms, and alternative sterilization modalities addressing environmental regulations. Canada represents a significant secondary market with contract sterilization facilities supporting the country's medical device manufacturing sector and serving as strategic hubs for cross-border sterilization services. The region faces capacity constraints in certain markets due to environmental regulatory pressures on ethylene oxide facilities, creating opportunities for gamma irradiation and electron beam sterilization expansion. Despite mature market characteristics, North America sustains solid growth driven by medical device innovation, regulatory compliance requirements, and ongoing outsourcing adoption among small and mid-sized medical device manufacturers.
Asia-Pacific
Rapid Healthcare Infrastructure Expansion and Medical Device Manufacturing Growth Drive Asia-Pacific as the Fastest-Growing Contract Sterilization Services Market
Asia-Pacific represents the fastest-growing regional segment in the contract sterilization services market, projected to expand at an exceptional CAGR of 8.5-9.0% during 2026-2033 as the region transforms into a global medical device manufacturing powerhouse. Countries including China, India, Japan, South Korea, Singapore, and Vietnam attract substantial medical device production investment driven by favorable labor costs, improving technical capabilities, and supportive government policies promoting healthcare sector development. The contract sterilization services market in Asia-Pacific benefits from multinational medical device companies establishing regional manufacturing operations requiring validated sterilization services meeting international quality standards for products destined for global markets.
International contract sterilization providers including STERIS, Sterigenics, and European-based companies actively invest in Asia-Pacific facility development, establishing strategic partnerships with regional medical device manufacturers and local sterilization service providers. China emerges as the largest national market within Asia-Pacific, supported by government healthcare reform initiatives expanding medical insurance coverage and hospital infrastructure that drives medical device consumption and corresponding sterilization demand. India demonstrates particularly strong growth potential with contract sterilization facilities supporting the country's expanding pharmaceutical and medical device manufacturing sectors serving domestic and export markets. Leading regional players include established providers like Steri-Tech and emerging companies investing in modern facilities equipped with multiple sterilization modalities. The contract sterilization services market in Southeast Asian nations grows rapidly as medical device manufacturers relocate production from higher-cost markets, creating immediate demand for validated sterilization infrastructure. Regulatory convergence toward international standards including ISO 11135 and ISO 11137 facilitates market entry for global contract sterilization providers while enabling regional manufacturers to access developed market customers. Investment in workforce training and technology transfer from established markets accelerates the development of local technical expertise supporting sustainable market growth throughout the forecast period.
Top Key Players
-
STERIS plc (Ireland)
-
Sotera Health Company (United States)
-
Getinge AB (Sweden)
-
Stryker Corporation (United States)
-
E-BEAM Services Inc. (United States)
-
BGS Beta-Gamma-Service GmbH & Co. KG (Germany)
-
Medistri SA (Switzerland)
-
Noxilizer Inc. (United States)
-
Cantel Medical Corporation (United States)
-
Cosmed Group Inc. (Canada)
-
Life Science Outsourcing Inc. (United States)
-
Synergy Health plc (United Kingdom)
-
Steri-Tech Inc. (South Africa)
-
Midwest Sterilization Corporation (United States)
Recent Developments
-
STERIS plc (2025) – Reported strong fiscal performance with 10% revenue growth in Applied Sterilization Technologies segment, driven by expanded service capacity and successful contract renewals with major medical device manufacturers across North America and Europe
-
Sotera Health Company (2024) – Announced strategic growth plan targeting 5-7% organic revenue growth through 2027, supported by investments in new gamma irradiation facilities and cobalt-60 supply development to strengthen sterilization capacity in high-growth markets
-
Getinge AB (2024) – Completed acquisition of regional sterilization service provider in Asia-Pacific, expanding the company's geographic footprint and adding specialized electron beam sterilization capabilities for pharmaceutical and medical device customers
-
E-BEAM Services Inc. (2025) – Commissioned new electron beam sterilization facility in the Southeastern United States with advanced automation and real-time monitoring systems, increasing regional processing capacity by 40% to meet growing customer demand
-
Sotera Health Company (2024) – Successfully resolved regulatory challenges related to ethylene oxide emissions through implementation of advanced abatement technologies and community engagement programs, enabling continued operations at key sterilization facilities
Market Trends
Sustainability Focus, Automation Integration, and Regional Capacity Expansion Shape the Evolution of the Contract Sterilization Services Market
The contract sterilization services market experiences transformative shifts as industry participants respond to environmental pressures, technological opportunities, and changing customer requirements. Service providers increasingly prioritize sustainable sterilization alternatives including vaporized hydrogen peroxide, nitrogen dioxide, and supercritical carbon dioxide technologies that deliver effective microbial inactivation without ethylene oxide emissions or radioactive material handling. These environmentally friendly modalities appeal to medical device manufacturers facing stakeholder pressure for reduced environmental footprints and regulatory agencies implementing stricter emissions standards. Investment in alternative sterilization technologies requires substantial capital but positions early adopters as preferred partners for sustainability-conscious customers.
Automation and artificial intelligence integration represent another dominant trend transforming operational efficiency within the contract sterilization services market. Leading providers implement robotic handling systems, computer vision inspection platforms, and machine learning algorithms that optimize sterilization cycle parameters, reduce manual labor requirements, and enhance quality consistency. Real-time monitoring systems provide customers with immediate access to sterilization status, validation data, and batch release documentation through cloud-based platforms that improve supply chain visibility and accelerate product distribution. Geographic capacity expansion continues as contract sterilization providers establish regional facilities near emerging medical device manufacturing hubs in Asia-Pacific, Latin America, and Eastern Europe. These strategic investments reduce transportation costs, shorten turnaround times, and strengthen customer relationships through localized technical support and regulatory expertise tailored to regional requirements.
Segments Covered in the Report
By Service Type
-
Contract Sterilization Services
-
Validation Services
-
Consulting Services
-
Testing Services
By Sterilization Method
-
Ethylene Oxide Sterilization
-
Gamma Irradiation
-
Electron Beam Sterilization
-
Steam Sterilization
-
Vaporized Hydrogen Peroxide
-
Others (X-Ray, Nitrogen Dioxide)
By Delivery Mode
-
Off-Site Sterilization
-
On-Site Sterilization
By End-User
-
Medical Device Companies
-
Hospitals and Healthcare Facilities
-
Pharmaceutical and Biotechnology Companies
-
Contract Manufacturing Organizations
-
Research Laboratories
-
Others
By Region
-
North America (United States, Canada, Mexico)
-
Europe (Germany, United Kingdom, France, Italy, Spain, Rest of Europe)
-
Asia-Pacific (China, Japan, India, South Korea, Australia, Southeast Asia, Rest of Asia-Pacific)
-
Latin America (Brazil, Argentina, Rest of Latin America)
-
Middle East & Africa (GCC Countries, South Africa, Rest of Middle East & Africa)
Frequently Asked Questions
Question 1: What is the projected size of the contract sterilization services market by 2033?
Answer: The contract sterilization services market is projected to reach approximately USD 2.26 billion by 2033, growing from USD 1.50 billion in 2026. This expansion reflects increasing demand from medical device manufacturers and pharmaceutical companies for validated sterilization solutions.
Question 2: Which region dominates the contract sterilization services market currently?
Answer: North America dominates the contract sterilization services market with the largest market share, driven by concentrated medical device manufacturing activity and stringent regulatory requirements. The region benefits from established sterilization infrastructure and high healthcare quality standards.
Question 3: What factors drive growth in the contract sterilization services market?
Answer: The contract sterilization services market grows due to increasing surgical procedure volumes, stringent regulatory compliance requirements, and medical device manufacturer outsourcing trends. Rising healthcare-associated infection awareness and expanding medical device complexity also contribute significantly to market expansion.
Question 4: Which sterilization method holds the largest share in the contract sterilization services market?
Answer: Ethylene oxide sterilization maintains the largest share in the contract sterilization services market, accounting for approximately 42-45% of market revenue. This method remains preferred for heat-sensitive medical devices requiring terminal sterilization with deep material penetration.
Question 5: How does the contract sterilization services market benefit from technological advancements?
Answer: The contract sterilization services market benefits from AI integration, automation, and alternative sterilization technologies that improve processing efficiency and environmental sustainability. Real-time monitoring systems and predictive maintenance reduce operational costs while enhancing quality consistency for customers.
