Compounding Chemotherapy Market Overview
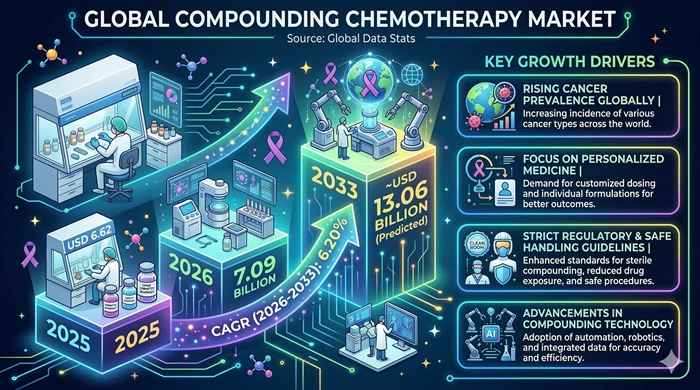
The global Compounding Chemotherapy market size is valued at USD 6.62 billion in 2025 and is predicted to increase from USD 7.09 billion in 2026 to approximately USD 13.06 billion by 2033, growing at a CAGR of 6.20% from 2026 to 2033.
Compounding chemotherapy refers to the sterile preparation of patient-specific or standardized antineoplastic drug doses in controlled pharmacy environments — tailoring formulations, concentrations, and delivery formats to meet the individual clinical needs of cancer patients that commercially manufactured oncology drugs cannot fully address. This specialized pharmaceutical service plays a critical and growing role in modern oncology care, encompassing in-house hospital workflows and outsourced sterile compounding services supported by cleanroom equipment, robotic compounding systems, gravimetric verification, isolators, and closed system transfer devices. The Compounding Chemotherapy market is expanding steadily alongside rising global cancer incidence, growing demand for personalized dosage forms, and the accelerating adoption of robotic and automated sterile compounding technologies.
AI Impact on the Compounding Chemotherapy Industry
Artificial Intelligence Is Transforming Compounding Chemotherapy Dose Optimization, Stability Prediction, Quality Assurance Automation, and Inventory Management in Ways That Are Meaningfully Improving Patient Safety and Pharmacy Operational Efficiency
Artificial intelligence is making a significant and growing impact across the Compounding Chemotherapy market, most notably through AI-powered analytics platforms being integrated into sterile compounding pharmacy workflows for dose calculation optimization, stability prediction, and real-time quality verification. AI and advanced analytics tools are now being applied to predict the physicochemical stability of compounded chemotherapy formulations under different storage conditions — enabling pharmacies to establish more accurate beyond-use dating and reduce product waste from premature expiry. The June 2025 pilot announcement by St Vincent's Hospital Melbourne — which adopted an AI-driven, cloud-based laser spectrometry solution from New Zealand-based company Veriphi to verify intravenous drug identity and concentration at the point of compounding — exemplifies the growing real-world deployment of AI-powered verification in oncology compounding environments, adding a critical layer of accuracy and safety assurance to the chemotherapy preparation process.
AI-driven tools are also being applied to optimize inventory management in high-volume oncology compounding operations — analyzing prescription demand patterns, drug usage trends, and supply chain signals to maintain optimal stock levels of hazardous drug active pharmaceutical ingredients and compounding consumables. This inventory intelligence reduces the risk of critical drug shortages affecting compounded chemotherapy preparation timelines, improving service reliability for hospital and oncology center clients. AI integration into robotic compounding systems is further elevating precision, reducing contamination risk, and enabling the compounding pharmacy sector to manage the growing complexity of individualized cancer treatment protocols with greater accuracy and efficiency. Collectively, these AI applications are accelerating the quality, safety, and scalability of the compounding chemotherapy market.
Growth Factors
Rising Global Cancer Incidence, Growing Demand for Personalized Chemotherapy Dosage Forms, Robotic and Automated Compounding Technology Adoption, and Recurring Commercial Drug Shortage Management Needs Are the Core Drivers
The most fundamental and enduring driver of the Compounding Chemotherapy market is the continuously rising global cancer burden, which is expanding the chemotherapy patient population year after year and growing the clinical demand for individualized compounded drug preparations. The WHO reports approximately 20 million new cancer cases diagnosed globally each year — a figure projected to exceed 29 million annually by 2040 as populations age and lifestyle risk factors compound. A critical and growing proportion of these patients require individualized compounded formulations — including pediatric cancer patients needing weight-based dosing unavailable in commercial formats, patients with renal or hepatic impairment requiring dose adjustments, individuals with drug allergies requiring alternative excipients, and patients on complex multi-drug regimens not commercially available in combination. These patient-specific needs make compounding pharmacy services an essential and irreplaceable component of comprehensive oncology care delivery globally.
The growing adoption of robotic and automated sterile compounding technologies is simultaneously transforming the operational capabilities and commercial appeal of compounding chemotherapy services. Robotic compounding systems reduce human error, minimize pharmacist and technician exposure to hazardous chemotherapy agents, improve dose preparation accuracy, and significantly increase throughput capacity — enabling compounding pharmacies to serve growing oncology patient volumes with greater safety and consistency. Advanced gravimetric verification systems, compounding aseptic isolators, and closed system transfer devices are further elevating sterility assurance and worker safety standards in compounding chemotherapy preparation environments. The accelerating adoption of these advanced technologies across hospital pharmacy departments and outsourced sterile compounding facilities is strengthening the clinical credibility and institutional adoption of compounding chemotherapy services throughout the forecast period.

Market Outlook
With Global Cancer Cases Rising, Personalized Medicine Expanding, Home Infusion Chemotherapy Growing, and Robotic Compounding Technology Scaling, the Compounding Chemotherapy Market Is Well-Positioned for Sustained Growth Through 2033
The long-term outlook for the Compounding Chemotherapy market is firmly positive and structurally well-supported. The forecast period from 2026 to 2033 will be characterized by continued growth in global cancer patient volumes, the progressive expansion of precision oncology treatment protocols requiring individualized drug formulations, and the steady development of oncology care infrastructure — including hospitals, ambulatory infusion centers, and home infusion pharmacy networks — across emerging markets in Asia Pacific, Latin America, and the Middle East. As per-capita healthcare expenditure rises and cancer treatment access expands across these high-population regions, demand for compounding chemotherapy services will grow meaningfully beyond the current North American and European market concentration, creating important new revenue opportunities for established compounding pharmacy operators with international capabilities.
The rapid growth of home infusion chemotherapy programs represents a particularly important demand driver for the Compounding Chemotherapy market during the forecast period. As healthcare systems globally shift cancer treatment from inpatient hospital settings toward cost-effective ambulatory and home environments — driven by patient preference, payer pressure to reduce hospitalization costs, and strong clinical evidence supporting safe outpatient chemotherapy delivery — the demand for pre-prepared, patient-specific, sterile compounded chemotherapy doses suitable for home administration is growing substantially. This shift is creating new high-volume opportunities for quality-certified compounding pharmacy providers capable of preparing, validating, and reliably supplying home infusion-ready chemotherapy formulations at scale — making the home infusion pharmacies segment one of the most commercially dynamic growth channels in the compounding chemotherapy landscape through 2033.
Expert Speaks
-
Albert Bourla, CEO, Pfizer — "Personalized cancer treatment is the future of oncology, and the ability to prepare individualized chemotherapy formulations through expert sterile compounding is an essential component of precision cancer care delivery. Ensuring that every patient receives exactly the right dose in the right formulation — including through high-quality compounded chemotherapy products — is fundamental to achieving the best possible treatment outcomes."
-
Joaquin Duato, CEO, Johnson & Johnson — "The pharmaceutical industry and the compounding pharmacy sector must work together to ensure that every cancer patient has access to the precise formulation they need — whether that means a commercially manufactured drug or a specially compounded preparation tailored to their individual clinical profile. Compounding chemotherapy plays a vital and growing role in delivering truly personalized oncology care globally."
-
Christopher Boerner, CEO, Bristol-Myers Squibb — "As oncology treatment protocols become increasingly complex and individualized, the role of specialized compounding pharmacy services in cancer care is becoming more — not less — important. The compounding chemotherapy sector's commitment to safety, sterility, and precision is a critical enabler of the personalized medicine transformation that is reshaping cancer treatment."
Key Report Takeaways
-
North America dominates the Compounding Chemotherapy market, holding approximately 38.40% of global market share in 2025, driven by the world's highest oncology drug expenditure, a mature and highly regulated compounding pharmacy infrastructure including a large network of FDA-registered 503B outsourcing facilities, strong institutional demand from major hospital oncology centers, and the presence of leading compounding pharmacy organizations including Baxter International, Fresenius Kabi, Fagron, and PharMEDium concentrated in the United States.
-
Asia Pacific is the fastest-growing regional market for compounding chemotherapy, driven by rapidly rising cancer incidence across China, India, Japan, and South Korea, significant investment in hospital pharmacy automation and robotic compounding infrastructure, growing adoption of personalized and dose-specific cancer treatment approaches, and increasing demand for cost-effective oncology drug compounding solutions across the region's rapidly expanding healthcare systems.
-
Hospital pharmacies are the dominant care setting segment, holding approximately 49.60% of total market share in 2025, driven by their availability of trained personnel, expertise in managing drug shortages, advanced sterile compounding capabilities, and the growing adoption of robotic and automated compounding technologies that enable hospital pharmacies to prepare high volumes of individualized chemotherapy doses with maximum precision and patient safety.
-
Outsourced sterile compounding services lead the offering segment, accounting for approximately 34.60% of market share in 2025, reflecting the strong and growing preference of hospital oncology departments to engage specialized 503B-registered outsourcing facilities for their compounded chemotherapy supply — driven by quality assurance requirements, operational cost management, and the high complexity of maintaining in-house sterile hazardous drug compounding infrastructure.
-
Patient-specific doses are the dominant preparation format segment, contributing approximately 41.50% of total revenue share in 2025, reflecting the fundamental clinical purpose of compounding chemotherapy — preparing individualized anticancer drug formulations precisely tailored to each patient's weight, renal function, tumor type, and treatment protocol rather than applying a standardized one-size-fits-all dosing approach.
-
Robotic compounding systems are the fastest-growing compounding technology segment, expected to expand at a CAGR of approximately 10.80% through 2033, driven by growing demand for automated precision dosing, reduced contamination risk, improved worker safety from hazardous drug exposure, enhanced throughput capacity in high-volume oncology compounding environments, and the long-term operational and economic benefits of robotic compounding relative to manual preparation methods.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 13.06 Billion |
| Market Size by 2025 | USD 6.62 Billion |
| Market Size by 2026 | USD 7.09 Billion |
| Market Growth Rate (2026–2033) | CAGR of 6.20% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Segments Covered | Offering, Preparation Format, Care Setting, Compounding Technology |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Rising Cancer Prevalence, Personalized Dosage Form Demand, Robotic Sterile Compounding Adoption, and Commercial Drug Shortage Management Needs Are the Four Pillars Driving Compounding Chemotherapy Market Growth
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising global cancer incidence expanding chemotherapy patient volumes | ~34% | Global — especially North America, Asia Pacific, Europe | Short to Long-term |
| Growing demand for personalized dosage forms tailored to patient medical history | ~28% | North America, Europe | Short to Medium-term |
| Adoption of robotic and automated sterile compounding technologies | ~24% | North America, Asia Pacific | Short to Long-term |
| Recurring commercial chemotherapy drug shortages driving compounding demand | ~14% | North America, Europe | Short to Medium-term |
The foundational and most structurally embedded demand driver of the Compounding Chemotherapy market is the relentlessly rising global cancer incidence, which continuously expands the chemotherapy patient population and proportionally grows the clinical volume of individualized compounded drug preparations required. An estimated 20 million new cancer cases are diagnosed globally each year — a figure projected to grow significantly through 2040 driven by population aging, lifestyle risk factors, and improving cancer detection rates in previously underdiagnosed regions. A critical and growing proportion of these patients require compounded formulations — including pediatric patients, those with organ function impairments, individuals with drug intolerances, and patients on complex multi-agent protocols — ensuring a robust and structurally growing demand foundation for compounding pharmacy services globally.
The accelerating adoption of robotic compounding systems and gravimetric verification technology is simultaneously strengthening the clinical safety credentials and operational scalability of compounding chemotherapy services in ways that are driving broader institutional adoption. Robotic compounding systems reduce human error, minimize pharmacist and technician exposure to hazardous chemotherapy agents, and significantly increase preparation throughput — enabling compounding pharmacies to serve growing oncology patient volumes with greater safety consistency and regulatory compliance assurance. As more hospital pharmacy departments and outsourced compounding facilities deploy robotic and automated compounding infrastructure, the quality and reliability of compounded chemotherapy products improves materially — driving greater institutional confidence in and procurement of these services across the healthcare system.

Restraints Impact Analysis
Stringent USP and FDA Regulatory Compliance, High Operational Cost of Hazardous Drug Compounding, Drug Stability Limitations, and Skilled Workforce Shortages Restrain Faster Compounding Chemotherapy Market Growth
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Stringent USP 797/800 and FDA regulatory compliance requirements | ~34% | North America, Europe | Short to Long-term |
| High capital and operational cost of sterile hazardous drug compounding facilities | ~28% | Global | Short to Medium-term |
| Drug stability limitations constraining beyond-use dating of compounded preparations | ~24% | Global | Short to Medium-term |
| Skilled pharmacy technician shortages in specialized compounding environments | ~14% | North America, Europe | Medium-term |
The increasingly rigorous regulatory compliance framework governing sterile and hazardous drug compounding — particularly in the United States, where USP Chapter 797 sterile compounding standards and USP Chapter 800 hazardous drug handling requirements impose demanding infrastructure, process, and documentation obligations on all compounding chemotherapy providers — creates substantial operational complexity and cost burdens for the sector. Compliance requires significant investment in cleanroom facilities, beyond-use dating validation programs, environmental monitoring systems, staff training programs, and documentation infrastructure — creating meaningful barriers to entry for smaller operations and ongoing compliance cost pressures for established providers. FDA inspection activity and state board of pharmacy oversight add further operational risk for compounding pharmacies that may face facility shutdowns or operational consent agreements if compliance gaps are identified during regulatory reviews.
The shortage of skilled pharmacy technicians specialized in operating, calibrating, and troubleshooting advanced robotic compounding equipment represents a growing operational constraint on the Compounding Chemotherapy market's ability to scale. As compounding pharmacies invest in increasingly sophisticated automated and robotic technologies, the demand for technicians with specialized training in robotic compounding system management, gravimetric verification, and sterile hazardous drug preparation protocols is growing faster than the available trained workforce — creating staffing bottlenecks that limit throughput expansion and impose higher labor costs on compounding pharmacy operators. This workforce challenge is most acute in North America and Europe, where the combination of growing oncology compounding volumes and advanced technology adoption is outpacing the training pipeline for qualified compounding pharmacy staff.
Opportunities Impact Analysis
Home Infusion Pharmacy Expansion, Emerging Market Oncology Infrastructure Growth, Robotic Compounding Scalability, and AI-Powered Quality Verification Deployment Create the Most Significant Untapped Market Opportunities
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rapid growth of home infusion pharmacy chemotherapy delivery programs | ~34% | North America, Europe | Short to Medium-term |
| Emerging market oncology infrastructure expansion driving new demand | ~28% | Asia Pacific, Latin America, MEA | Medium to Long-term |
| Robotic compounding technology enabling scalable high-volume outsourcing | ~26% | North America, Asia Pacific | Short to Medium-term |
| AI-powered point-of-care drug verification improving safety and adoption | ~12% | North America, Europe, Asia Pacific | Medium-term |
The rapid expansion of home infusion chemotherapy delivery programs represents the most immediately significant commercial growth opportunity in the Compounding Chemotherapy market. As healthcare systems globally shift cancer treatment from inpatient hospital settings to cost-effective home and ambulatory environments, the demand for pre-prepared, patient-specific, sterile compounded chemotherapy doses safe for home administration is growing substantially. Home infusion pharmacies that can prepare, validate, and reliably supply high-quality compounded chemotherapy admixtures for home administration are positioned to capture this fast-growing new demand channel — one that offers attractive revenue per patient relative to traditional hospital supply agreements and that is being actively supported by payer policy in North America and Europe seeking to reduce expensive inpatient oncology care costs.
The growing investment in oncology care infrastructure across Asia Pacific — particularly China's major government investment in hospital pharmacy automation and robotic compounding capabilities — is creating a large and strategically important emerging market growth opportunity for compounding chemotherapy providers with regional capabilities. China's focus on improved regulatory standards for sterile compounding, combined with the country's growing demand for targeted therapies and personalized cancer treatment, is progressively building the institutional framework required for large-scale compounding chemotherapy service adoption. India, South Korea, and Southeast Asian markets are similarly investing in hospital pharmacy modernization, creating new first-time adoption opportunities for both compounding technology providers and outsourced sterile compounding service organizations during the 2026–2033 forecast period.

Segment Analysis
By Offering
Outsourced Sterile Compounding Services Lead the Compounding Chemotherapy Market as the Dominant Institutional Supply Channel, While Closed System Transfer Devices Emerge as the Fastest-Growing Offering Segment
Outsourced sterile compounding services hold the dominant position within the offering segment of the Compounding Chemotherapy market, accounting for approximately 34.60% of total global market revenue in 2025. This segment's leadership reflects the strong and growing preference of hospital oncology pharmacy departments to engage specialized external compounding facilities — particularly FDA-registered 503B outsourcing facilities in the United States — for their compounded chemotherapy supply, driven by the high capital and compliance costs of maintaining in-house sterile hazardous drug compounding infrastructure, the need for USP-compliant patient-specific sterile formulations, and the operational benefit of eliminating contamination risk and managing skilled personnel shortages through contracted outsourcing arrangements. North America accounts for the largest outsourced sterile compounding service revenue share, where the established 503B outsourcing facility sector — represented by major companies including Baxter International, Fresenius Kabi, Fagron Sterile Services, PharMEDium, QuVa Pharma, and CAPS — generates significant contracted supply revenues from hospital oncology center procurement agreements. The segment benefits from growing institutional recognition that outsourced 503B-quality compounding provides a more reliable, quality-assured, and cost-effective alternative to in-house preparation for many standardized and semi-standardized oncology admixture needs.
Closed system transfer devices are the fastest-growing segment within the offering category of the Compounding Chemotherapy market, projected to expand at a CAGR of approximately 8.20% from 2026 to 2033, driven by heightened worker and patient safety requirements, rising oncology case volumes, increasingly stringent regulatory guidelines for hazardous drug handling, and the need to prevent chemotherapy agent exposure during drug formulation and administration. These devices — including syringe safety devices, bag access devices, vial access devices, and Luer-lock systems — are essential safety tools that maintain the integrity of compounded chemotherapy preparations while protecting pharmacists, nurses, and patients from hazardous drug exposure. North America leads closed system transfer device adoption within the compounding chemotherapy setting, where OSHA, NIOSH, and USP Chapter 800 guidelines actively encourage or in some contexts mandate their use in hazardous drug compounding and administration workflows. Key companies in this segment include ICU Medical, B. Braun Melsungen, and BD (Becton Dickinson).
By Care Setting
Hospital Pharmacies Anchor the Compounding Chemotherapy Market With the Highest Preparation Volume, While Home Infusion Pharmacies Rapidly Emerge as the Fastest-Growing Care Setting Segment
Hospital pharmacies represent the dominant care setting segment in the Compounding Chemotherapy market, accounting for approximately 49.60% of total global market revenue in 2025. Their dominance reflects the central role of hospital pharmacy departments as the primary compounding and dispensing point for intravenous chemotherapy across the world's largest and most active oncology treatment centers. Hospital pharmacies benefit from direct integration with clinical oncology teams, immediate patient-specific prescription access, advanced sterile compounding infrastructure, and the clinical flexibility to rapidly prepare and adjust individualized chemotherapy formulations in response to changing patient condition. The growing adoption of robotic compounding systems and gravimetric verification technology within hospital pharmacy departments is further strengthening their capacity to serve expanding oncology patient volumes with maximum precision and safety. North America is the largest regional contributor to hospital pharmacy compounding chemotherapy revenue, where major academic cancer center hospital systems — including MD Anderson, Memorial Sloan Kettering, and large GPO-affiliated hospital networks — generate the highest institutional compounding preparation volumes. Leading technology and service providers serving this segment include Baxter International, Fresenius Kabi, Grifols, and ICU Medical.
Home infusion pharmacies represent the fastest-growing care setting segment in the Compounding Chemotherapy market, projected to expand at a CAGR of approximately 8.10% from 2026 to 2033, driven by the accelerating shift of chemotherapy delivery from inpatient hospital settings to home-based environments — a trend that received major momentum during the COVID-19 pandemic and has continued to strengthen as both patients and payers recognize the clinical safety and significant cost advantages of home chemotherapy administration. Home infusion pharmacies prepare customized, sterile, and accurately dosed chemotherapy medications for direct patient home administration, ensuring that elderly patients and those who cannot regularly visit hospital infusion suites receive safe and effective oncology treatment in the comfort of their own homes. North America and Europe are the primary growth regions for home infusion compounding chemotherapy, where established home infusion pharmacy networks and supportive payer reimbursement frameworks for home-based oncology care are enabling this rapidly growing care delivery model. Companies including Coram CVS Specialty Infusion Services, BioMatrix Specialty Pharmacy, and Option Care Health serve as leading home infusion providers in the North American market.

Regional Insights
North America
North America Leads the Global Compounding Chemotherapy Market With the Highest Cancer Treatment Volumes, Most Advanced 503B Outsourcing Infrastructure, and Greatest Robotic Compounding Technology Adoption
North America holds the dominant position in the global Compounding Chemotherapy market, accounting for approximately 38.40% of total global market revenue in 2025, and is projected to maintain its regional leadership at a CAGR of approximately 5.90% from 2026 to 2033. The United States is the primary revenue driver, where the highest global oncology drug expenditure, the largest and most mature network of FDA-registered 503B outsourcing facilities, strong institutional demand from academic cancer centers and regional oncology programs, recurring commercial chemotherapy drug shortage management needs, and the growing adoption of robotic compounding systems and closed system transfer devices collectively create the world's most commercially developed Compounding Chemotherapy market. The U.S. market alone was valued at approximately USD 1.99 billion in 2025, with major compounding pharmacy and outsourcing facility operators — including Baxter International, Fresenius Kabi, Fagron, PharMEDium (Cencora), QuVa Pharma, and CAPS — serving the institutional demand of large hospital oncology networks through multi-year supply contracts.
Canada contributes to regional market revenue through its hospital pharmacy compounding programs and a growing outsourced sterile compounding sector serving oncology centers in major urban healthcare hubs. The progressive regulatory tightening around sterile and hazardous drug compounding standards — driven by continuing FDA and state board of pharmacy oversight of the 503B sector — is expected to drive market consolidation toward larger, quality-compliant outsourcing facility operators throughout the forecast period, concentrating compounding chemotherapy supply volumes with well-capitalized market leaders and creating both competitive pressure and acquisition opportunity for mid-tier participants in the North American compounding pharmacy landscape.
Asia Pacific
Asia Pacific Is the Fastest-Growing Compounding Chemotherapy Market, Driven by Surging Cancer Incidence, Rapid Hospital Pharmacy Automation Investment, and Growing Demand for Personalized Oncology Dosage Forms
Asia Pacific is the fastest-growing regional segment in the Compounding Chemotherapy market, driven by surging cancer incidence — China alone accounts for approximately 23.8% of global new cancer cases annually — combined with significant government and private sector investment in hospital pharmacy automation, robotic compounding infrastructure, and sterile compounding quality upgrades across the region's major healthcare markets. China's major investment in hospital pharmacy automation and robotic compounding systems, its growing demand for targeted therapies and personalized oncology treatment, and improving regulatory standards for sterile compounding are collectively building the institutional framework for large-scale compounding chemotherapy service adoption. Japan and South Korea represent the most mature compounding chemotherapy markets within the region, characterized by advanced hospital pharmacy infrastructure, sophisticated oncology programs, and growing precision medicine adoption that generates strong demand for individualized compounded formulations. Key companies with Asia Pacific compounding chemotherapy presence include B. Braun Melsungen, Fresenius Kabi, Grifols, and a growing number of regional specialty compounding pharmacy operators expanding across China, India, and Southeast Asia.
India and Southeast Asian markets — including Singapore, Thailand, and Malaysia — are increasingly investing in hospital pharmacy modernization and sterile compounding capability development as their healthcare systems scale to meet rising oncology care demands. India's rapidly expanding private hospital oncology center network and its growing middle-class population seeking advanced cancer treatment are creating important new first-time adoption opportunities for both compounding technology equipment providers and outsourced sterile compounding service organizations. As regulatory frameworks for pharmaceutical compounding mature across Asian markets and as awareness of the clinical value of personalized compounded chemotherapy formulations grows among regional oncologists, Asia Pacific is expected to remain the most commercially dynamic and fastest-growing regional market in global Compounding Chemotherapy market throughout the 2026–2033 forecast period.
Top Key Players
-
Baxter International Inc. (United States)
-
B. Braun Melsungen AG (Germany)
-
Fresenius Kabi AG (Germany)
-
Grifols S.A. (Spain)
-
ICU Medical, Inc. (United States)
-
PharMEDium — Cencora, formerly AmerisourceBergen (United States)
-
Nephron Pharmaceuticals Corporation (United States)
-
QuVa Pharma, Inc. (United States)
-
Wells Pharma of Houston, LLC (United States)
-
Fagron NV (Belgium)
-
Wedgewood Pharmacy (United States)
-
McKesson Corporation — Oncology & Specialty (United States)
-
CAPS — Central Admixture Pharmacy Services (United States)
-
Cantrell Drug Company (United States)
-
Empower Pharmacy (United States)
Recent Developments
-
St Vincent's Hospital Melbourne (June 2025) — Announced the pilot deployment of an AI-driven, cloud-based drug verification solution provided by New Zealand-based company Veriphi within its compounding pharmacy department. The technology uses laser spectrometry to verify intravenous drug identity and concentration at the precise point of compounding — adding a critical and automated layer of accuracy verification to sterile chemotherapy preparation and representing one of the most clinically significant AI safety innovations deployed in oncology compounding globally.
-
Georgetown Public Hospital Corporation (June 2025) — Commissioned a new non-hazardous compounding hood at its Maternity Pharmacy, officially launching GPHC's Compounding Sterile Preparation IV Program in Guyana — a landmark development that marks the establishment of formalized sterile compounding infrastructure in a country with previously limited pharmaceutical compounding capabilities, reflecting the growing global expansion of compounding pharmacy services into emerging healthcare markets.
-
Fagron NV (2024) — Expanded its sterile compounding services network across North America and Europe by investing in additional 503B-compliant outsourcing facility capacity and advanced robotic compounding infrastructure, reinforcing its position as one of the world's leading providers of outsourced sterile compounding services for oncology and hospital pharmacy clients and strengthening its ability to meet growing institutional demand for quality-assured compounded chemotherapy products.
-
Baxter International (2025) — Advanced its pharmaceutical compounding and admixture portfolio by expanding its INTRAVIA bag-based ready-to-administer chemotherapy product range, targeting the growing institutional demand for pre-prepared, sterile, and convenience-optimized oncology drug admixtures from hospital oncology centers and ambulatory infusion facilities seeking to reduce in-house preparation burden and improve workflow efficiency.
-
ICU Medical, Inc. (2024) — Strengthened its closed system transfer device portfolio for oncology and hazardous drug compounding applications through product line extensions targeting compatibility with a broader range of chemotherapy drug vials and syringe configurations, responding to growing regulatory and institutional demand for comprehensive hazardous drug containment solutions across hospital pharmacy and ambulatory infusion center settings.
Market Trends
Robotic Compounding Automation Scaling Across Hospital and Outsourcing Facility Settings and the Accelerating Shift Toward Home-Based Chemotherapy Delivery Are the Two Dominant Trends Reshaping the Compounding Chemotherapy Market
The most transformative operational trend reshaping the Compounding Chemotherapy market is the rapid scaling of robotic compounding systems and automated gravimetric verification technology across both hospital pharmacy departments and 503B outsourcing facilities. Robotic compounding arms — capable of preparing hazardous chemotherapy doses with sub-milligram precision, in fully contained sterile environments, and at throughput rates far exceeding manual preparation capacity — are transitioning from early-adopter novelty to mainstream institutional standard across high-volume oncology compounding settings. These systems address multiple simultaneous challenges including pharmacist and technician hazardous drug exposure risk, preparation accuracy and dose consistency requirements, workforce shortage pressures, and the growing regulatory scrutiny of sterile compounding quality standards — making robotic compounding technology adoption both a quality imperative and a commercial differentiator for leading compounding pharmacy operators.
The accelerating shift of chemotherapy delivery from hospital inpatient settings toward home and ambulatory environments is simultaneously creating a major structural transformation in how compounded chemotherapy products are prepared, packaged, and distributed. Ready-to-administer IV bags and pre-filled syringes — which allow chemotherapy to be safely and conveniently administered by home infusion nurses or patient caregivers outside hospital settings — are among the fastest-growing preparation format segments in the market, reflecting this care setting transition. As home infusion pharmacy networks scale, as payer reimbursement for home chemotherapy delivery expands, and as clinical evidence supporting the safety of outpatient chemotherapy administration strengthens, the demand for sterile, shelf-stable, patient-specific compounded chemotherapy products suitable for home use will continue to grow significantly — making home-oriented compounding service capabilities a critical strategic investment for leading compounding chemotherapy market participants through 2033.
Segments Covered in the Report
By Offering:
-
Outsourced Sterile Compounding Services
-
In-House Compounding Systems
-
Closed System Transfer Devices
-
Sterile Compounding Consumables
-
Validation and Compliance Services
By Preparation Format:
-
Patient-Specific Doses
-
Batch-Compounded Doses
-
Ready-to-Administer Syringes
-
Ready-to-Administer IV Bags
-
Clinical Trial Preparations
By Care Setting:
-
Hospital Pharmacies
-
Oncology Clinics
-
Ambulatory Infusion Centers
-
Home Infusion Pharmacies
-
Academic Medical Centers
By Compounding Technology:
-
Biological Safety Cabinets
-
Compounding Aseptic Isolators
-
Robotic Compounding Systems
-
Gravimetric Verification Systems
-
Environmental Monitoring Systems
By Region:
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the Compounding Chemotherapy market and what is its projected value by 2033?
Answer: The Compounding Chemotherapy market was valued at USD 6.62 billion in 2025 and is projected to reach USD 13.06 billion by 2033. It is expected to grow at a CAGR of 6.20% from 2026 to 2033, driven by rising global cancer incidence, growing demand for personalized dosage forms, and rapid adoption of robotic and automated sterile compounding technologies.
Question 2: What are the primary growth drivers of the Compounding Chemotherapy market?
Answer: Rising global cancer prevalence generating growing demand for individualized chemotherapy formulations and the accelerating adoption of robotic and automated sterile compounding technologies are the two most powerful drivers of the Compounding Chemotherapy market. Growing demand for personalized dosage forms tailored to patient medical history and recurring commercial chemotherapy drug shortages are also meaningfully expanding market demand.
Question 3: Which region leads the Compounding Chemotherapy market and which is the fastest growing?
Answer: North America dominates the Compounding Chemotherapy market with approximately 38.40% of global revenue in 2025, driven by high oncology treatment volumes, mature 503B outsourcing infrastructure, and advanced robotic compounding adoption. Asia Pacific is the fastest-growing region, driven by surging cancer incidence, rapid hospital pharmacy automation investment, and growing demand for personalized oncology compounding across China, India, and Southeast Asia.
Question 4: Which offering segment and care setting lead the Compounding Chemotherapy market?
Answer: Outsourced sterile compounding services lead the offering segment with approximately 34.60% of total Compounding Chemotherapy market revenue in 2025, while closed system transfer devices are the fastest-growing offering. Hospital pharmacies dominate the care setting segment with approximately 49.60% market share, while home infusion pharmacies are growing fastest at approximately 8.10% CAGR through 2033.
Question 5: How is robotic technology shaping the future of the Compounding Chemotherapy market?
Answer: Robotic compounding systems are transforming the Compounding Chemotherapy market by improving dose preparation precision, reducing hazardous drug exposure for healthcare workers, managing workforce shortages, and enabling significantly higher throughput capacity in high-volume oncology compounding environments. This segment is expected to grow at the highest CAGR of approximately 10.80% through 2033, making robotic compounding technology adoption a critical strategic differentiator for leading market participants.
