Circulating Biomarkers Market Overview
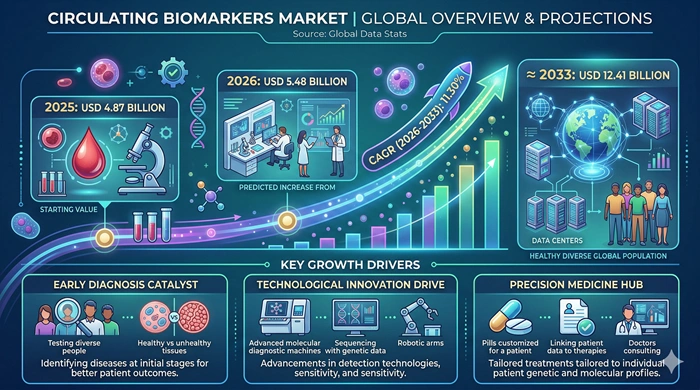
The global Circulating Biomarkers market size is valued at USD 4.87 billion in 2025 and is predicted to increase from USD 5.48 billion in 2026 to approximately USD 12.41 billion by 2033, growing at a CAGR of 11.30% from 2026 to 2033.
Circulating biomarkers are measurable biological molecules — including circulating tumor DNA, circulating tumor cells, cell-free DNA, exosomes, microRNAs, proteins, peptides, and metabolites — found in body fluids such as blood, urine, and cerebrospinal fluid, detectable through minimally invasive or non-invasive sampling procedures that provide clinically actionable information about the presence, progression, treatment response, and recurrence of diseases including cancer, cardiovascular disorders, neurological conditions, and infectious diseases. Unlike tissue biopsy approaches requiring surgical intervention, circulating biomarkers enable repeated, real-time sampling of disease biology through routine blood draws — generating dynamic longitudinal disease monitoring capability that is transforming early cancer detection, treatment selection, therapeutic monitoring, and minimal residual disease surveillance across oncology and beyond. The Circulating Biomarkers market is experiencing strong and sustained commercial growth driven by the rapid clinical adoption of liquid biopsy technologies in oncology, expanding NGS platform accessibility enabling sensitive ctDNA detection, growing regulatory approvals for blood-based biomarker companion diagnostics, escalating pharmaceutical investment in biomarker-guided clinical trial designs, and the progressive development of multi-cancer early detection testing programs that represent an enormous potential expansion of routine population screening applications.
AI Impact on the Circulating Biomarkers Industry
Artificial Intelligence Is Transforming Circulating Biomarker Signal Detection Sensitivity, Multi-Analyte Data Integration, Cancer Origin Prediction, Treatment Response Modeling, and Clinical Decision Support in Ways That Are Fundamentally Expanding the Diagnostic Utility and Commercial Scale of the Circulating Biomarkers Market*
Artificial intelligence is delivering its most clinically transformative contributions to the Circulating Biomarkers market through AI-powered multi-analyte signal integration platforms — where machine learning algorithms simultaneously analyze ctDNA methylation patterns, protein biomarker panels, cell-free RNA signals, and fragmentomic profiles from a single blood draw to generate cancer detection models with sensitivity and specificity that no individual biomarker type achieves alone. Companies including GRAIL — whose Galleri multi-cancer early detection test combines AI-powered methylation signal analysis across more than 50 cancer types — and Exact Sciences are demonstrating that AI-enabled multi-analyte liquid biopsy interpretation can identify cancer signals with clinically meaningful sensitivity at early stages where surgical cure remains possible, generating the clinical validation data that is progressively establishing multi-cancer early detection testing as a serious population screening candidate. The progressive application of deep learning algorithms to ctDNA fragmentomic analysis — where AI models detect cancer-associated DNA fragmentation pattern signatures in cell-free DNA that are invisible to conventional biomarker analysis but reproducibly detectable by trained neural networks — is generating an entirely new class of cancer detection signal that supplements traditional mutation-based ctDNA analysis and improves early-stage detection sensitivity in cancer types where somatic mutation signals are weak or absent.
AI is additionally transforming the pharmacogenomic and treatment response prediction applications of the circulating biomarkers market — where machine learning models trained on longitudinal ctDNA dynamics, protein biomarker trajectories, and clinical outcome data are generating predictive algorithms that forecast individual patient treatment response probabilities, optimal drug combination selections, and acquired resistance emergence timing with accuracy that is progressively informing real-world oncology treatment decision-making. Pharmaceutical companies including Roche, Foundation Medicine, and Guardant Health are embedding AI-powered biomarker interpretation engines within their companion diagnostic platforms — enabling oncologists to receive not just raw mutation detection results but integrated clinical interpretation recommendations based on the accumulated treatment outcome data from thousands of previously tested patients with similar biomarker profiles. This AI-powered clinical decision support capability is progressively elevating circulating biomarker testing from a diagnostic reporting service into a clinical intelligence platform — generating meaningfully higher clinical value per test, supporting premium pricing, and driving broader oncologist adoption across clinical practice settings that collectively define the Circulating Biomarkers market's commercial growth trajectory.
Growth Factors
Liquid Biopsy Clinical Adoption Acceleration in Oncology, NGS Platform Cost Reduction Enabling Broader Access, Companion Diagnostic Regulatory Approval Expansion, and Rising Prevalence of Chronic Diseases Are the Core Growth Drivers of the Circulating Biomarkers Market*
The most commercially powerful and structurally sustained growth driver of the Circulating Biomarkers market is the accelerating clinical adoption of liquid biopsy-based circulating tumor DNA testing across the full oncology care continuum — from treatment selection and companion diagnostic applications at diagnosis, through treatment monitoring and minimal residual disease detection during therapy, to recurrence surveillance post-treatment. The FDA's progressive expansion of approved companion diagnostic indications for blood-based biomarker tests — including Guardant Health's Guardant360 CDx, Foundation Medicine's FoundationOne Liquid CDx, and Roche's cobas EGFR Mutation Test — is generating growing oncologist adoption of liquid biopsy biomarker testing as a clinically validated and reimbursable standard of care component for biomarker-selected targeted therapy prescribing across lung, colorectal, breast, prostate, and other major cancer types. The progressive reduction in next-generation sequencing platform costs — where sequencing costs per gigabase have declined by more than a hundredfold over the past decade through platform innovation by Illumina, Oxford Nanopore, and Pacific Biosciences — is simultaneously expanding the economic accessibility of sensitive ctDNA detection and comprehensive genomic profiling from liquid biopsy samples to a broader range of clinical settings, laboratory types, and healthcare markets globally.
The enormous and rapidly developing multi-cancer early detection testing opportunity — where population-level blood-based screening programs using multi-analyte circulating biomarker panels could potentially detect multiple cancer types simultaneously from a single routine blood draw before symptomatic clinical presentation — represents the most commercially transformative potential growth driver for the Circulating Biomarkers market over the medium to long term. GRAIL's Galleri test — commercially launched in the United States and undergoing NHS evaluation in the United Kingdom — is the most advanced multi-cancer early detection product currently available, and its real-world clinical deployment is generating the clinical performance data and health economic evidence that will inform regulatory approval decisions, national screening program adoption, and insurance coverage determinations that could open a massive population screening revenue opportunity for circulating biomarker testing. The growing pharmaceutical industry investment in circulating biomarker integration within clinical trial designs — where ctDNA and other liquid biopsy biomarkers serve as enrollment selection criteria, pharmacodynamic response endpoints, and early efficacy signals in oncology drug development programs — is additionally generating strong and growing institutional demand for validated circulating biomarker testing services from pharmaceutical and biotechnology companies that represents a large and commercially attractive contract services revenue stream.

Market Outlook
With Multi-Cancer Early Detection Commercialization Advancing, Companion Diagnostic Approval Pipeline Expanding, Emerging Market Liquid Biopsy Infrastructure Developing, and Proteomics and Fragmentomics Adding New Biomarker Signal Layers, the Circulating Biomarkers Market Outlook Is Strong Through 2033*
The long-term outlook for the Circulating Biomarkers market is strongly positive and commercially well-supported — with a compelling combination of oncology clinical adoption expansion, multi-cancer early detection program development, companion diagnostic pipeline growth, pharmaceutical partnership revenue, and international market geographic expansion collectively expected to sustain strong revenue growth throughout the 2026–2033 forecast period. The most commercially significant near-term development within the circulating biomarkers ecosystem is the progressive regulatory pathway navigation and payer coverage determination process for multi-cancer early detection tests — where positive clinical utility evidence from large-scale studies including GRAIL's PATHFINDER and STRIVE trials, and NHS England's Galleri trial involving more than 140000 participants, is generating the real-world clinical performance evidence base that regulatory agencies and payers require before authorizing reimbursed population screening applications. Successful regulatory approval and major payer coverage for even a single multi-cancer early detection test would represent a step-change in the total addressable market for the Circulating Biomarkers market — transforming the potential patient population from currently diagnosed cancer patients receiving therapeutic monitoring to the entire adult screening-eligible population, a market expansion of potentially an order of magnitude in test volume.
The progressive development of next-generation circulating biomarker modalities — including exosome-based biomarker analysis, cell-free RNA profiling, circulating epigenetic markers, and AI-enabled fragmentomic signatures — is expanding the clinical information extractable from liquid biopsy samples beyond DNA mutation detection to encompass broader biological disease signals that improve both sensitivity and tissue-of-origin specificity across cancer types. The international market development opportunity for the Circulating Biomarkers market is particularly commercially significant in Asia Pacific — where China's enormous cancer incidence burden, rapidly developing genomics industry infrastructure, growing healthcare investment, and strong government emphasis on early cancer detection are generating both patient demand and institutional capability for large-scale liquid biopsy deployment. The combination of technology advancement, clinical evidence accumulation, regulatory pathway navigation, payer coverage development, and geographic market expansion will collectively sustain strong commercial growth in the circulating biomarkers market across the full forecast period through 2033.
Expert Speaks
-
Thomas Schinecker, CEO, Roche — "Circulating biomarkers and liquid biopsy represent one of the most scientifically exciting and commercially consequential areas of innovation in diagnostics and oncology — where the ability to repeatedly sample tumor biology from a simple blood draw is transforming how we detect, monitor, and guide treatment for cancer patients in ways that tissue biopsy simply cannot match for real-time therapeutic response assessment. Roche is deeply committed to advancing the clinical validation, regulatory approval, and global access infrastructure for circulating biomarker testing across the full cancer care continuum."
-
Shacey Petrovic, CEO, Guardant Health — "The clinical case for comprehensive liquid biopsy-based biomarker testing in oncology has never been stronger — with FDA-approved companion diagnostic indications, robust clinical evidence for treatment monitoring applications, and the first real-world multi-cancer early detection deployments collectively demonstrating that blood-based circulating biomarker testing can deliver the clinical information oncologists need to make better treatment decisions at every point in the cancer care journey. Guardant Health is executing on a strategy to establish liquid biopsy as a routine and reimbursed component of standard oncology care globally."
-
Kevin Conroy, CEO, Exact Sciences — "The convergence of sensitive multi-analyte biomarker detection, AI-powered signal interpretation, and progressively accessible next-generation sequencing is enabling a new generation of blood-based cancer tests that were simply not scientifically achievable five years ago — and the clinical evidence for their performance continues to strengthen through large-scale real-world deployment studies that are building the foundation for regulatory approval and payer coverage decisions that will define the commercial scale of this opportunity. Exact Sciences is investing aggressively in blood-based cancer detection technology development to position ourselves as a leader in the early cancer detection transformation that circulating biomarker science is enabling."
Key Report Takeaways
-
North America dominates the Circulating Biomarkers market, holding approximately 42.60% of global market share in 2025, driven by the highest concentration of FDA-approved liquid biopsy companion diagnostic tests, the most advanced oncology genomics infrastructure, the greatest density of academic cancer centers and molecular diagnostic laboratories performing circulating biomarker testing, the strongest venture capital and pharmaceutical investment in liquid biopsy company development, and the most developed reimbursement frameworks for circulating tumor DNA testing in oncology clinical practice.
-
Asia Pacific is the fastest-growing regional market for circulating biomarkers, projected to expand at a CAGR of approximately 13.40% from 2026 to 2033, driven by China's enormous cancer incidence burden representing approximately 4.5 million new cases annually, rapidly developing genomics industry infrastructure including domestic NGS platform companies such as BGI Genomics and Berry Genomics, growing government investment in precision medicine and early cancer detection programs, and the progressive development of reimbursement pathways for molecular diagnostic testing within public and private Chinese healthcare systems.
-
Oncology is the dominant application segment, accounting for approximately 67.40% of total market revenue in 2025, driven by the largest number of regulatory-approved clinical indications for circulating tumor DNA companion diagnostics, the highest clinical need for minimally invasive disease monitoring tools, the strongest pharmaceutical partnership investment in biomarker-guided oncology drug development, and the most advanced clinical evidence base for liquid biopsy applications spanning treatment selection, monitoring, minimal residual disease detection, and early detection across multiple cancer types.
-
Next-Generation Sequencing is the dominant technology segment, contributing approximately 51.80% of total market revenue in 2025, reflecting its unparalleled ability to simultaneously detect multiple mutation types across comprehensive cancer gene panels from circulating DNA at the low allele fractions characteristic of liquid biopsy samples — combined with the progressive reduction in NGS platform costs that has made comprehensive genomic profiling from plasma samples economically viable for routine clinical deployment across a growing range of healthcare settings globally.
-
Circulating Tumor DNA is the dominant biomarker type segment, accounting for approximately 45.20% of total market revenue in 2025, driven by its established clinical utility as a companion diagnostic biomarker for multiple targeted therapy prescribing decisions in lung, colorectal, breast, and prostate cancer — supported by the largest collection of regulatory approvals, clinical evidence publications, and physician familiarity among all circulating biomarker modality types currently in clinical use.
-
Exosomes and Extracellular Vesicles are the fastest-growing biomarker type segment, projected to expand at a CAGR of approximately 17.20% through 2033, driven by their cargo richness carrying protein, RNA, DNA, and lipid biomarker signals simultaneously, their biological stability in circulation, and the growing academic and commercial research investment in exosome isolation, characterization, and clinical application development that is progressively maturing exosome-based diagnostics toward commercial clinical deployment.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 12.41 Billion |
| Market Size by 2025 | USD 4.87 Billion |
| Market Size by 2026 | USD 5.48 Billion |
| Market Growth Rate (2026–2033) | CAGR of 11.30% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Segments Covered | Biomarker Type, Technology, Application, End User |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Expanding FDA-Approved Companion Diagnostic Indications, NGS Cost Reduction, Rising Cancer and Chronic Disease Prevalence, Pharmaceutical Biomarker Integration, and AI-Enhanced Detection Capabilities Are the Core Pillars Driving the Circulating Biomarkers Market*
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Expanding FDA-approved companion diagnostic indications for liquid biopsy ctDNA tests | ~33% | North America, Europe | Short to Long-term |
| Progressive NGS platform cost reduction enabling broader clinical accessibility | ~26% | Global | Short to Long-term |
| Rising cancer and chronic disease prevalence increasing demand for non-invasive diagnostics | ~23% | Global | Short to Long-term |
| Pharmaceutical industry biomarker-guided clinical trial design adoption | ~18% | North America, Europe, Asia Pacific | Short to Medium-term |
The most commercially consequential growth driver of the Circulating Biomarkers market is the expanding regulatory approval landscape for liquid biopsy-based companion diagnostics in oncology — where the FDA has progressively approved blood-based ctDNA tests as companion diagnostics for multiple targeted therapies including EGFR inhibitors in lung cancer, KRAS inhibitors in colorectal cancer, PIK3CA inhibitors in breast cancer, and AR pathway inhibitors in prostate cancer — generating clinically mandated testing indications that drive large and recurring test volumes as these therapeutic categories collectively represent millions of patient treatment decisions annually in North America and Europe alone. Each new companion diagnostic approval creates a durable, clinically required testing indication that grows total market revenue predictably as the corresponding therapeutic class expands its patient reach and as clinical guidelines progressively incorporate liquid biopsy testing as the preferred biomarker assessment approach for its speed, accessibility, and serial monitoring advantages over tissue biopsy. The additional pharmaceutical industry investment in circulating biomarker integration within oncology drug development programs — where ctDNA and protein biomarkers serve as enrollment selection, pharmacodynamic monitoring, and surrogate efficacy endpoints in clinical trials — generates large and growing contract research and reference laboratory revenue from pharmaceutical and biotechnology clients that provides commercially important revenue diversification for leading Circulating Biomarkers market participants.
The structural cost reduction trajectory of next-generation sequencing platforms — enabled by continuous innovation in sequencing chemistry, instrument throughput, and bioinformatics processing efficiency by Illumina, MGI Tech, Oxford Nanopore, and competing platform developers — is progressively lowering the per-test cost of comprehensive ctDNA genomic profiling to levels that make routine clinical deployment economically viable across an expanding range of hospital laboratory, independent diagnostic laboratory, and international market settings. This cost reduction is simultaneously improving the economic case for payer coverage of liquid biopsy testing — where declining per-test costs improve the health economic cost-effectiveness ratios that inform payer coverage determination and reimbursement rate setting decisions — progressively converting the reimbursement access constraint from a market growth barrier into an enabling factor as costs reach levels where clinical value per dollar clearly justifies coverage. Combined with the strong and growing evidence base for clinical utility of ctDNA testing across oncology indications, the cost reduction dynamic is generating a powerful and mutually reinforcing driver of Circulating Biomarkers market expansion through both volume growth and market access broadening simultaneously.

Restraints Impact Analysis
Analytical Sensitivity Limitations in Early-Stage Detection, High Test Complexity and Cost, Reimbursement Coverage Gaps, Pre-Analytical Variability, and Clinical Interpretation Standardization Challenges Restraining Faster Circulating Biomarkers Market Growth*
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Limited detection sensitivity for early-stage cancers with low ctDNA shedding | ~34% | Global | Short to Long-term |
| Incomplete insurance and payer reimbursement coverage beyond approved companion diagnostics | ~27% | North America, Europe | Short to Long-term |
| Pre-analytical sample handling variability affecting result reproducibility | ~24% | Global | Short to Medium-term |
| Lack of standardized clinical interpretation frameworks across different platforms | ~15% | Global | Short to Long-term |
The most fundamentally limiting technical constraint on the Circulating Biomarkers market's expansion into early cancer detection applications is the challenge of detecting cancer-associated circulating biomarker signals — particularly ctDNA — at the extremely low concentrations and allele fractions characteristic of early-stage and localized cancers, where total tumor burden is small, ctDNA shedding rates are low relative to background cell-free DNA, and signal-to-noise discrimination requires analytical sensitivity levels at the boundary of current sequencing technology capabilities. Early-stage solid tumor ctDNA detection sensitivity limitations — where Stage I solid tumors may have ctDNA allele fractions below 0.1% that current standard NGS approaches cannot reliably detect — create a clinical gap between the early detection application that would generate the greatest healthcare value and the analytical performance currently achievable. This sensitivity limitation is the primary technical barrier that multi-analyte AI-powered approaches — combining ctDNA with methylation, protein, and fragmentomic signals — are specifically designed to overcome, and the clinical evidence generated by GRAIL's Galleri and competing multi-analyte platforms will progressively determine how comprehensively this gap can be closed within the forecast period.
Incomplete and inconsistent payer reimbursement coverage for liquid biopsy circulating biomarker testing — beyond the established companion diagnostic indications with formal FDA approval — represents a significant commercial growth constraint for the broader Circulating Biomarkers market across monitoring, surveillance, early detection, and emerging clinical use cases. In the United States, Medicare coverage for comprehensive genomic profiling liquid biopsy tests under MolDX coverage policies has been progressing but remains incomplete for many clinical indications — creating reimbursement uncertainty for both laboratories and physicians that limits test ordering confidence and patient accessibility for indications without firm coverage determinations. Pre-analytical variability — where differences in blood collection tube type, processing time, centrifugation protocol, and plasma storage conditions can meaningfully affect ctDNA recovery efficiency — creates result reproducibility challenges across different clinical collection sites and laboratory workflows that complicate multi-site clinical trial biomarker analysis and real-world clinical interpretation.
Opportunities Impact Analysis
Multi-Cancer Early Detection Population Screening Programs, Minimal Residual Disease Monitoring Clinical Adoption, Emerging Market Liquid Biopsy Infrastructure Development, and Pharmaceutical Companion Diagnostic Partnership Expansion Creating Significant Circulating Biomarkers Market Opportunities*
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Multi-cancer early detection population screening regulatory approval and payer coverage | ~35% | North America, Europe | Short to Long-term |
| Minimal residual disease ctDNA monitoring clinical guideline integration and reimbursement | ~27% | North America, Europe, Asia Pacific | Short to Medium-term |
| Emerging market liquid biopsy infrastructure development in China, India, and Southeast Asia | ~23% | Asia Pacific | Medium to Long-term |
| Proteomics and epigenomics multi-analyte signal integration expanding clinical utility | ~15% | North America, Europe | Short to Long-term |
The most commercially transformative opportunity within the Circulating Biomarkers market is the potential regulatory approval and payer coverage of multi-cancer early detection blood tests for population screening — where the successful navigation of FDA approval pathways and subsequent Medicare and commercial insurance coverage determinations for GRAIL's Galleri or competing multi-cancer early detection platforms would unlock an addressable market of tens of millions of annual screening tests among adults aged 50 and above in the United States alone. The large-scale NHS Galleri trial — enrolling more than 140000 participants in the United Kingdom and generating prospective real-world clinical performance data expected to inform both NHS national screening program adoption decisions and international regulatory reference datasets — represents the most significant ongoing clinical validation program for multi-cancer early detection. The progressive development of minimal residual disease ctDNA monitoring as a clinically adopted and reimbursed post-treatment surveillance tool — where serial ctDNA testing after curative-intent cancer surgery or chemotherapy detects molecular recurrence months to years before imaging-detectable disease progression — is expanding the circulating biomarker testing market into a large and recurring post-treatment surveillance application that generates long-term, repeat-testing revenue from the growing global population of cancer survivors.
The enormous liquid biopsy market development opportunity in Asia Pacific — particularly in China, where domestic genomics companies including Berry Genomics, Burning Rock Biotech, and Genetron Health are building commercially competitive ctDNA testing platforms and clinical laboratory networks capable of serving China's enormous cancer patient population at locally competitive pricing — represents a significant geographic market expansion of the global Circulating Biomarkers market addressable revenue that will become progressively more commercially important throughout the forecast period. China's National Healthcare Security Administration is progressively expanding reimbursement coverage for molecular diagnostic testing within Chinese public health insurance — a development that could dramatically accelerate liquid biopsy adoption across the massive Chinese cancer patient population currently accessing testing primarily through self-pay channels. The growing integration of proteomics-based circulating biomarker panels — where sensitive proximity extension assay technology from Olink Proteomics and SomaScan technology from SomaLogic enable simultaneous measurement of hundreds to thousands of plasma proteins from a single blood sample — with genomic liquid biopsy approaches is generating multi-analyte biomarker discovery and clinical application opportunities that are progressively expanding the scientific and commercial frontier of the Circulating Biomarkers market into new disease indications and precision medicine use cases beyond oncology.

Segment Analysis
By Biomarker Type
Circulating Tumor DNA Leads the Market in Revenue While Exosomes and Extracellular Vesicles Drive the Fastest Segment Growth in the Circulating Biomarkers Market*
Circulating tumor DNA holds the dominant revenue position in the Circulating Biomarkers market, accounting for approximately 45.20% of total global market revenue in 2025, driven by its established and growing collection of FDA-approved companion diagnostic indications across multiple oncology therapeutic classes, its strong clinical evidence base supporting utility across treatment selection, monitoring, and surveillance applications, and the maturity of commercially deployed ctDNA testing platforms from Guardant Health, Foundation Medicine, and Roche that have achieved broad clinical adoption and commercial reimbursement across North American and European oncology practice settings. The ctDNA segment benefits from a particularly strong commercial ecosystem — where pharmaceutical company partnerships for companion diagnostic co-development, academic medical center research collaborations, and clinical laboratory service contracts collectively generate diversified and recurring revenue streams that drive consistent segment growth independent of any single clinical application. North America leads ctDNA segment revenue — driven by the largest FDA-approved companion diagnostic portfolio, the strongest insurance reimbursement coverage for approved ctDNA indications, and the highest oncology genomic testing utilization rates globally — with key companies including Guardant Health and Foundation Medicine dominating the commercial ctDNA testing market. Europe is the second-largest ctDNA market, driven by CE-IVD approved ctDNA platforms and growing European Society for Medical Oncology guideline adoption of liquid biopsy recommendations across major European cancer research centers.
Exosomes and extracellular vesicles represent the fastest-growing biomarker type segment in the circulating biomarkers market — projected to expand at a CAGR of approximately 17.20% through 2033 — driven by their unique biological properties as naturally occurring nano-sized vesicles that encapsulate and protect a diverse cargo of protein, RNA, DNA, and lipid biomarkers reflecting the physiological or pathological state of their cells of origin, providing a potentially richer and more disease-informative signal than cell-free nucleic acid analysis alone. The exosome segment is experiencing rapid growth in both academic discovery research and commercial clinical application development — with companies including Exosome Diagnostics (Bio-Techne), Exosome Sciences, and a growing pipeline of venture-backed exosome diagnostic startups advancing exosome-based biomarker tests toward clinical validation and commercial launch across oncology, neurology, and cardiovascular disease applications. Asia Pacific is the fastest-growing region for exosome research and commercial development — driven by strong government research investment in exosome biology at Chinese, Japanese, and South Korean academic institutions, growing biotechnology company formation in the exosome diagnostic space, and the region's broad interest in next-generation liquid biopsy technologies that go beyond first-generation ctDNA testing. The clinical translation of exosome-based biomarkers from discovery research into validated clinical diagnostic products is expected to progressively accelerate through the forecast period as platform standardization, clinical validation evidence, and regulatory pathway clarity develop in parallel with growing industry investment in commercial exosome diagnostic product development.
By Application
Oncology Dominates the Circulating Biomarkers Market While Neurological Disorder Applications Register the Fastest Growth Driven by Blood-Based Alzheimer's Biomarker Adoption*
Oncology holds the overwhelmingly dominant application position in the Circulating Biomarkers market, accounting for approximately 67.40% of total global market revenue in 2025, driven by the largest collection of regulatory-approved clinical indications, the most advanced clinical evidence base, the strongest pharmaceutical partnership investment, and the highest oncologist familiarity with circulating tumor DNA and protein biomarker applications across cancer diagnosis, treatment selection, monitoring, and surveillance use cases. The oncology segment encompasses a comprehensive and growing range of circulating biomarker clinical applications — from EGFR and KRAS mutation testing for targeted therapy selection in lung and colorectal cancer, to HER2 amplification and PIK3CA mutation testing for breast cancer treatment decisions, to AR-V7 splice variant detection for prostate cancer therapeutic sequencing — collectively generating large and expanding test volumes across multiple major cancer types. North America generates the largest oncology application segment revenue — driven by the most advanced liquid biopsy clinical adoption culture, the highest targeted therapy prescribing rates, and the most developed reimbursement infrastructure for ctDNA companion diagnostics — with Guardant Health, Foundation Medicine, and Roche collectively dominating clinical ctDNA testing volumes across the US oncology market. The oncology application segment within the circulating biomarkers market is expected to sustain strong revenue growth throughout the forecast period as companion diagnostic indications expand, monitoring applications gain reimbursement, and multi-cancer early detection commercialization potentially creates an enormous new oncology screening revenue category.
Neurological disorder applications represent the fastest-growing application segment in the circulating biomarkers market — projected to expand at a CAGR of approximately 20.40% through 2033 — driven primarily by the landmark clinical validation and commercial adoption of blood-based Alzheimer's disease biomarkers, including plasma phosphorylated tau 217 (p-tau217) and amyloid ratio tests from companies including Roche, Fujirebio, ALZpath, and Lilly's Precivity AD2 test, that are progressively replacing expensive and invasive PET imaging and cerebrospinal fluid testing as the preferred Alzheimer's diagnostic confirmation approach for patients presenting with cognitive impairment symptoms. The regulatory approval of anti-amyloid Alzheimer's therapies including lecanemab (Leqembi) and donanemab — which require amyloid confirmation before prescribing — is generating large and growing clinical demand for accessible blood-based amyloid biomarker testing as a gating diagnostic for therapy eligibility, creating a rapidly expanding companion diagnostic market within the neurology application segment. Europe and Asia Pacific are particularly fast-growing neurological biomarker regions — driven by strong academic neuroscience research ecosystems and the progressive development of memory clinic infrastructure creating institutional capacity for blood-based Alzheimer's biomarker adoption. Key companies including Roche Diagnostics, Fujirebio, and ALZpath are leading neurological circulating biomarker commercial development globally.

Regional Insights
North America
North America Leads the Global Circulating Biomarkers Market at 42.60% Revenue Share, Driven by the Most Advanced Liquid Biopsy Clinical Adoption, FDA Companion Diagnostic Approvals, and Strongest Research Investment*
North America holds the dominant position in the global Circulating Biomarkers market, accounting for approximately 42.60% of total global market revenue in 2025, and is projected to sustain market leadership at a CAGR of approximately 11.10% from 2026 to 2033. The United States drives the overwhelming majority of North American revenue — generating the world's most commercially advanced liquid biopsy and circulating biomarker testing market through the combination of the largest portfolio of FDA-approved companion diagnostic ctDNA tests, the most developed commercial insurance reimbursement coverage for approved liquid biopsy indications, the highest oncology genomic testing utilization rates globally, and the greatest concentration of pioneering liquid biopsy companies including Guardant Health, Foundation Medicine, Exact Sciences, GRAIL, and Tempus AI that collectively define the global competitive frontier of circulating biomarker commercial innovation. North America benefits uniquely from the FDA's regulatory framework that has established clear approval pathways for liquid biopsy companion diagnostics — progressively authorizing blood-based ctDNA tests as primary companion diagnostics for multiple targeted oncology therapies and generating the reimbursement credibility that drives clinical adoption at scale across the US oncology practice ecosystem.
Canada contributes meaningfully to North American circulating biomarker revenue — through a growing adoption of ctDNA companion diagnostic testing in Canadian academic cancer center oncology practices and progressive provincial health insurance coverage expansion for molecular diagnostic testing. The United States National Cancer Institute and NIH continue to fund large-scale liquid biopsy research programs — including Cancer Moonshot initiative components focused on early cancer detection — that generate both academic evidence supporting clinical translation and trained research talent fueling commercial company innovation pipelines. North America's combination of regulatory leadership, commercial infrastructure maturity, research investment, and clinical adoption culture positions it to sustain clear Circulating Biomarkers market leadership through 2033.
Asia Pacific
Asia Pacific Is the Fastest-Growing Circulating Biomarkers Market at 13.40% CAGR, Driven by China's Cancer Burden, Domestic Genomics Industry Strength, and Government Precision Medicine Investment*
Asia Pacific is the fastest-growing regional segment in the Circulating Biomarkers market, projected to expand at a CAGR of approximately 13.40% from 2026 to 2033 — the highest regional growth rate globally. China represents the most commercially significant and fastest-growing Asia Pacific circulating biomarker market — driven by the world's largest absolute cancer incidence burden of approximately 4.5 million new cases annually, a rapidly maturing domestic genomics and liquid biopsy industry anchored by companies including Burning Rock Biotech, Berry Genomics, Genetron Health, and Novogene, strong government investment in precision medicine infrastructure and early cancer detection programs, and progressive National Healthcare Security Administration reimbursement expansion for molecular diagnostic testing within public health insurance. Key international companies including Roche, Illumina, and Guardant Health are simultaneously establishing growing commercial presences in China's liquid biopsy market through local partnerships and regulatory submissions, creating a competitive but commercially expanding market environment that collectively drives strong revenue growth.
Japan and South Korea represent the most technically advanced and commercially sophisticated Asia Pacific circulating biomarker markets outside China — with Japan's well-developed clinical laboratory infrastructure, strong academic liquid biopsy research ecosystem, and progressive national health insurance coverage for approved molecular diagnostic indications generating a mature domestic market for ctDNA and protein biomarker testing in oncology clinical practice. India represents an emerging but rapidly developing Asia Pacific circulating biomarker market — where a combination of growing cancer incidence, rapid expansion of private hospital molecular diagnostic capabilities, and strong domestic bioinformatics talent is creating the foundation for significant market development over the forecast period. Asia Pacific's combination of scale, government support, domestic industry strength, and rapidly developing clinical infrastructure positions it as the most commercially dynamic Circulating Biomarkers market geography globally through 2033.
Top Key Players
-
Guardant Health Inc. (United States)
-
Foundation Medicine Inc. — Roche Subsidiary (United States)
-
Roche Diagnostics (Switzerland)
-
Exact Sciences Corporation (United States)
-
GRAIL Inc. — Illumina Subsidiary (United States)
-
Illumina Inc. (United States)
-
Bio-Techne Corporation — Exosome Diagnostics (United States)
-
Burning Rock Biotech Limited (China)
-
Berry Genomics Co. Ltd. (China)
-
Fujirebio Inc. (Japan)
-
Tempus AI Inc. (United States)
-
Veracyte Inc. (United States)
Recent Developments
-
Guardant Health (2025) — Received FDA approval for its Shield blood test for colorectal cancer screening — marking a landmark regulatory milestone as the first FDA-approved blood-based colorectal cancer screening test for average-risk adults — while simultaneously reporting strong commercial launch momentum with growing physician ordering adoption, advancing CMS Medicare coverage determinations for the Shield test, and continuing to expand its comprehensive ctDNA companion diagnostic portfolio across additional oncology therapeutic indications through pharmaceutical company co-development partnerships.
-
GRAIL — Illumina (2025) — Continued commercial expansion of its Galleri multi-cancer early detection test in the United States while reporting ongoing enrollment progress in the large-scale NHS Galleri trial in the United Kingdom — with updated real-world clinical performance data continuing to demonstrate meaningful cancer signal detection across more than 50 cancer types, while simultaneously engaging with FDA on regulatory pathway discussions and working with commercial insurance carriers on coverage policy development for multi-cancer early detection screening applications.
-
Foundation Medicine — Roche (2025) — Launched expanded FoundationOne Liquid CDx panel updates incorporating additional biomarker indications aligned with new FDA companion diagnostic co-approvals — while reporting strong growth in pharmaceutical company clinical trial biomarker testing partnerships generating increasing contract research revenue alongside growing clinical diagnostic volumes — and advancing integration of Foundation Medicine's liquid biopsy capabilities within Roche's broader companion diagnostics strategy across its oncology drug development portfolio.
-
Burning Rock Biotech (2024) — Expanded its circulating tumor DNA testing service portfolio with new clinical applications addressing esophageal, gastric, and hepatocellular carcinoma — cancer types with particularly high incidence in Chinese and broader Asian patient populations — while reporting growing pharmaceutical company partnership revenue from ctDNA biomarker services in clinical trials and advancing discussions with China's National Medical Products Administration for regulatory approval of additional ctDNA companion diagnostic indications aligned with targeted oncology therapies available in the Chinese market.
-
Exact Sciences (2025) — Advanced development of its next-generation blood-based multi-cancer detection program — reporting positive interim data from its BLUE-C clinical study evaluating a blood-based colorectal cancer detection test — while simultaneously expanding its established Cologuard colorectal cancer stool DNA screening test commercial infrastructure and investing in AI-powered multi-analyte biomarker signal integration capabilities that will underpin its future multi-cancer blood test product development strategy.
Market Trends
Multi-Analyte AI-Powered Liquid Biopsy Panels Replacing Single-Analyte Tests, Blood-Based Neurological Biomarker Adoption Expanding, and Minimal Residual Disease Monitoring Becoming a Standard Post-Treatment Tool Are the Most Commercially Defining Trends Reshaping the Circulating Biomarkers Market Through 2033*
The most commercially transformative trend reshaping the Circulating Biomarkers market is the progressive transition from single-analyte circulating biomarker tests toward comprehensive multi-analyte AI-powered liquid biopsy panels that simultaneously measure ctDNA mutations, methylation patterns, fragmentomics signatures, protein biomarkers, and cell-free RNA signals from a single blood draw and integrate these diverse biological signals through machine learning algorithms to generate more sensitive and specific diagnostic and monitoring outputs than any single biomarker can provide. This multi-analyte integration trend is most commercially advanced in the multi-cancer early detection application — where GRAIL's Galleri and competing platforms are demonstrating that AI-powered methylation signal analysis across multiple biomarker types can detect cancer signals across dozens of cancer types with clinically meaningful sensitivity — but is progressively expanding into oncology monitoring, recurrence surveillance, and companion diagnostic applications as the clinical performance advantages of multi-analyte approaches accumulate in published research. Companies that invest in building comprehensive multi-analyte biomarker measurement capabilities combined with sophisticated AI interpretation platforms are progressively differentiating commercially from single-analyte competitors — generating stronger clinical evidence, broader approved indication portfolios, and higher clinical value per test that support premium pricing and stronger payer coverage positioning.
The emergence of blood-based neurological biomarker testing as a rapidly growing and commercially significant new application category — anchored by the clinical validation of plasma p-tau217 and amyloid ratio tests as accessible Alzheimer's diagnostic tools — is expanding the Circulating Biomarkers market beyond its traditional oncology focus into neurology in a way that is creating substantial new revenue streams and attracting diagnostic company investment in neurological biomarker platform development. The progressive adoption of minimal residual disease ctDNA monitoring as a standard post-treatment surveillance component in adjuvant therapy decision-making for colorectal, breast, and lung cancer — where guidelines from ESMO and ASCO are progressively incorporating ctDNA MRD monitoring recommendations — is simultaneously converting post-treatment surveillance from an occasional imaging-based recurrence check into a regular, molecularly-informed clinical management practice generating recurring liquid biopsy test volumes from the large and growing global cancer survivor population. These converging trends collectively reinforce the strong and broad-based commercial growth trajectory of the Circulating Biomarkers market through 2033 and beyond.
Segments Covered in the Report
By Biomarker Type:
-
Circulating Tumor DNA (ctDNA)
-
Circulating Tumor Cells (CTCs)
-
Exosomes and Extracellular Vesicles
-
Cell-Free DNA (cfDNA)
-
MicroRNA (miRNA)
-
Proteins and Peptides
-
Metabolites
-
Others
By Technology:
-
Next-Generation Sequencing (NGS)
-
Polymerase Chain Reaction (PCR)
-
Immunoassay
-
Microarray
-
Mass Spectrometry
-
Others
By Application:
-
Oncology
-
Cardiovascular Disease
-
Neurological Disorders
-
Infectious Diseases
-
Autoimmune Diseases
-
Others
By End User:
-
Hospitals and Diagnostic Laboratories
-
Academic and Research Institutes
-
Pharmaceutical and Biotechnology Companies
-
Contract Research Organizations
By Region:
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the Circulating Biomarkers market and what is its projected value by 2033?
Answer: The Circulating Biomarkers market was valued at USD 4.87 billion in 2025 and is projected to reach USD 12.41 billion by 2033, growing at a CAGR of 11.30% from 2026 to 2033. This strong growth is driven by expanding FDA-approved liquid biopsy companion diagnostic indications in oncology, progressive NGS platform cost reduction, growing pharmaceutical biomarker integration investment, and the advancing development of multi-cancer early detection programs that could dramatically expand the total market addressable population.
Question 2: What are circulating biomarkers and how are they used clinically?
Answer: Circulating biomarkers are measurable biological molecules — including ctDNA, circulating tumor cells, exosomes, cell-free DNA, proteins, and microRNAs — detectable in body fluids such as blood through minimally invasive sampling that provide clinically actionable information about disease presence, progression, and treatment response without requiring surgical tissue biopsy. In the Circulating Biomarkers market, the most established clinical applications are companion diagnostic biomarker testing for targeted cancer therapy prescribing, treatment response monitoring, minimal residual disease detection after cancer surgery, and cancer recurrence surveillance — with multi-cancer early detection representing the most commercially significant emerging application.
Question 3: Which region dominates the Circulating Biomarkers market and which is growing fastest?
Answer: North America leads the Circulating Biomarkers market with approximately 42.60% of global revenue in 2025 — driven by the largest portfolio of FDA-approved liquid biopsy companion diagnostics, the most mature clinical adoption culture, and the greatest concentration of leading commercial companies including Guardant Health, Foundation Medicine, GRAIL, and Exact Sciences. Asia Pacific is the fastest-growing region at approximately 13.40% CAGR through 2033 — driven primarily by China's enormous cancer incidence burden, rapidly developing domestic genomics industry infrastructure, and growing government investment in precision medicine and early cancer detection programs.
Question 4: What is the difference between liquid biopsy and circulating biomarkers?
Answer: Liquid biopsy is the clinical procedure — the minimally invasive blood draw or other biofluid sampling — while circulating biomarkers are the specific biological molecules detected and measured within those biofluid samples to generate clinically useful diagnostic or monitoring information. The Circulating Biomarkers market encompasses the full ecosystem of biomarker types, detection technologies, clinical applications, and commercial products that collectively enable liquid biopsy-based clinical testing — with ctDNA currently the most commercially prominent circulating biomarker type, followed by circulating tumor cells, exosomes, and protein biomarkers across a growing range of clinical indications.
Question 5: How is AI transforming the Circulating Biomarkers market?
Answer: Artificial intelligence is transforming the Circulating Biomarkers market most significantly through multi-analyte signal integration — where machine learning algorithms combine ctDNA mutation, methylation, fragmentomic, and protein biomarker signals from a single blood draw to generate cancer detection and tissue-of-origin prediction models with sensitivity and specificity that no individual biomarker achieves alone, most visibly in multi-cancer early detection platforms like GRAIL's Galleri. AI is additionally advancing clinical decision support through treatment response prediction models trained on longitudinal ctDNA dynamics, improving the clinical value of biomarker reporting from raw mutation detection results into integrated therapeutic interpretation recommendations that progressively elevate liquid biopsy testing from a diagnostic service into a clinical intelligence platform.
