Bone Densitometer Devices Market Overview
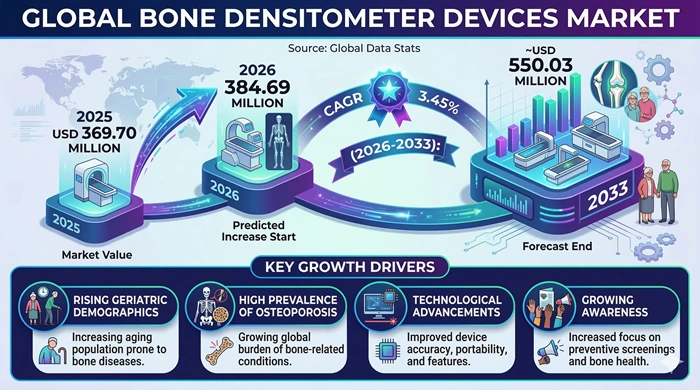
The global Bone Densitometer Devices market size is valued at USD 369.70 million in 2025 and is predicted to increase from USD 384.69 million in 2026 to approximately USD 550.03 million by 2033, growing at a CAGR of 3.45% from 2026 to 2033.
Bone densitometer devices are specialized medical imaging systems used to measure bone mineral density (BMD) and assess fracture risk — with their most critical clinical application being the early diagnosis and ongoing monitoring of osteoporosis and related skeletal conditions. The rising global burden of osteoporosis — driven by rapidly aging populations, increasingly sedentary lifestyles, nutritional deficiencies, and growing postmenopausal populations — is creating steady and expanding clinical demand for bone density testing equipment across all major healthcare markets. As healthcare systems increasingly emphasize preventive screening and early intervention for bone health, the Bone Densitometer Devices market continues on a measured but well-supported growth path through 2033.
AI Impact on the Bone Densitometer Devices Industry
Artificial Intelligence Is Reshaping Bone Mineral Density Analysis, Fracture Risk Prediction, and Clinical Workflow Efficiency in Ways That Are Meaningfully Expanding the Diagnostic Value and Operational Performance of Modern Bone Densitometry Systems
Artificial intelligence is becoming an increasingly integrated part of the Bone Densitometer Devices market, most significantly through AI-powered image analysis algorithms embedded within DXA and QCT systems. AI-assisted tools have demonstrated strong diagnostic performance — comparable to clinical expert assessment — and are now being actively developed for bone health screening applications in both hospital and community settings. Researchers in Japan, for instance, have developed AI-assisted diagnostic systems capable of estimating lumbar spine and femoral bone mineral density using standard anteroposterior X-ray images, a development that could dramatically expand opportunistic osteoporosis screening beyond dedicated densitometry appointments. Recent systematic reviews and meta-analyses confirm that AI algorithms can achieve diagnostic performance on par with clinical specialists, and that combined AI-and-specialist approaches deliver even better outcomes — a finding that is driving healthcare institutions to actively seek AI-enabled bone densitometry platforms.
Beyond image interpretation, AI is supporting the integration of machine learning and deep learning tools alongside digital detector technologies to advance the accuracy and speed of bone health diagnostics comprehensively. AI-powered algorithms automatically analyze imaging data, identify fracture risks in real time, and assist clinicians in making more precise and consistent diagnostic and treatment decisions — reducing the inter-operator variability that has historically been a notable limitation of conventional bone densitometry interpretation. These advances are also enabling opportunistic osteoporosis diagnosis from routine imaging performed for other indications — greatly expanding the population that can be screened for low bone mineral density without the need for dedicated densitometer appointments. As AI capabilities mature and gain regulatory validation, they are expected to become a standard feature of next-generation bone densitometer platforms, strengthening the clinical and commercial value proposition of premium device offerings.
Growth Factors
Rising Osteoporosis Prevalence Among Aging Populations, Expanding Clinical Screening Guidelines, Growing Preventive Healthcare Focus, and Advanced AI-Integrated Imaging Technologies Are the Core Forces Driving Bone Densitometer Devices Market Growth
The most fundamental and enduring driver of the Bone Densitometer Devices market is the rapidly escalating global burden of osteoporosis and bone-related disorders, driven primarily by the unprecedented pace of global population aging. Osteoporosis disproportionately affects postmenopausal women and elderly individuals — populations growing in absolute size across virtually every major healthcare market — and remains widely underdiagnosed even following traumatic fracture events, underscoring the critical importance of proactive bone density screening. The International Osteoporosis Foundation estimates that more than 200 million women worldwide are affected by osteoporosis, with millions of fragility fractures occurring annually — a burden that carries enormous healthcare system costs in terms of hospitalization, surgical intervention, rehabilitation, and long-term disability care. This growing epidemiological pressure is directly translating into expanding clinical demand for bone densitometer devices across hospitals, diagnostic centers, and orthopedic clinics globally.
Growing awareness of bone health as a preventive healthcare priority — among both patients and healthcare providers — is simultaneously expanding the active screening population for bone densitometer devices. National health authorities in North America, Europe, and increasingly across Asia Pacific are strengthening their recommendations for routine bone density testing in high-risk populations including postmenopausal women, elderly men, and individuals on long-term corticosteroid therapy. Healthcare systems are increasingly adopting a preventive approach to bone health management — emphasizing early detection of low bone mineral density and timely pharmacological intervention before fragility fractures occur rather than reactive management after fracture events. This preventive healthcare philosophy, combined with rising per-capita healthcare expenditure across middle-income countries, is progressively expanding the global installed base of bone densitometer devices and the annual volume of bone density scan procedures.

Market Outlook
With Global Osteoporosis Burden Rising Steadily, Emerging Market Healthcare Infrastructure Expanding Rapidly, and Next-Generation Portable and AI-Enhanced Densitometry Technologies Broadening Access, the Bone Densitometer Devices Market Outlook Is Stable and Well-Supported Through 2033
The long-term outlook for the Bone Densitometer Devices market is steadily positive, supported by the compounding effect of demographic aging, expanding clinical screening programs, and progressive healthcare infrastructure development in high-growth emerging markets. The forecast period from 2026 to 2033 will be characterized by continued procurement of central DXA systems in well-developed markets — driven by device replacement cycles and capacity expansion in diagnostic imaging centers — alongside meaningful first-time adoption of both axial and peripheral bone densitometry equipment in expanding emerging market healthcare facilities across Asia Pacific, Latin America, and the Middle East. India's government-initiated establishment of approximately 150000 Health and Wellness Centers is improving access to basic diagnostic services across rural and semi-urban populations, creating a new and scalable institutional channel for bone health screening device adoption in one of the world's most populous high-osteoporosis-burden countries.
Technological innovation is also playing an important supportive role in the Bone Densitometer Devices market outlook. Manufacturers are actively developing compact, portable, and handheld bone densitometer systems suitable for use in outpatient clinics, mobile health programs, and community screening initiatives — device formats that dramatically improve the accessibility of bone density assessment beyond traditional hospital and imaging center settings. The integration of AI-assisted analysis capabilities, cloud connectivity, and remote reporting features into next-generation densitometer platforms is enhancing device utility and clinical workflow efficiency — strengthening the investment case for device upgrades in established markets and making advanced bone health assessment capabilities accessible to a broader range of healthcare facility types globally.
Expert Speaks
-
Veronika Elsner, CEO, Hologic — "Advancing the clinical reach and diagnostic capabilities of bone densitometry is central to our mission of improving women's health outcomes globally. Our continued investment in AI-enhanced DXA platforms and body composition analysis reflects our commitment to ensuring that bone densitometer devices deliver ever-greater diagnostic value to clinicians and patients — particularly in the prevention and early management of osteoporosis."
-
Peter Arduini, CEO, GE HealthCare — "Integrating AI into medical imaging workflows — including bone density assessment — is one of the most impactful ways we can help clinicians make faster and more confident diagnostic decisions. GE HealthCare's investment in AI-powered densitometry analytics is focused on extracting greater clinical value from every scan, improving osteoporosis detection rates, and supporting better patient outcomes across our global customer base."
-
Bernd Montag, CEO, Siemens Healthineers — "The growing global burden of osteoporosis and bone-related disorders demands both better diagnostic tools and broader access to bone health screening across all healthcare settings. Siemens Healthineers is committed to developing imaging technologies — including advanced bone densitometry capabilities — that support early, accurate, and accessible bone mineral density assessment for aging populations worldwide."
Key Report Takeaways
-
North America dominates the Bone Densitometer Devices market, holding the largest global revenue share in 2025, supported by high healthcare spending, a well-developed medical imaging infrastructure, strong insurance and Medicare reimbursement frameworks for bone density testing, high osteoporosis prevalence among the aging U.S. population, and the presence of leading bone densitometer manufacturers including Hologic and GE HealthCare headquartered in the region.
-
Asia Pacific is the fastest-growing regional market for bone densitometer devices, driven by a rapidly aging population — particularly across China, Japan, India, and South Korea — rising per-capita income, ongoing improvements in healthcare infrastructure, growing government investment in preventive bone health screening programs, and the enormous and largely underdiagnosed osteoporosis burden across the region's vast elderly population.
-
Hospitals are the dominant end-user segment, holding the largest share of bone densitometer device installations in 2025, as hospitals provide access to multidisciplinary care teams — including orthopedic physicians, radiologists, and endocrinologists — combined with advanced diagnostic infrastructure, enabling comprehensive bone mineral density evaluation, integrated fracture risk management, and high device utilization rates.
-
Diagnostic centers are the fastest-growing end-user segment, expected to expand at the highest CAGR through 2033, driven by growing investments in advanced DXA, pDXA, and QUS equipment by diagnostic imaging centers, their referral network collaborations with physicians and specialists, and supportive regulatory and preventive healthcare initiatives that are expanding osteoporosis screening access through outpatient diagnostic settings.
-
Dual Energy X-Ray Absorptiometry (DXA/DEXA) is the dominant technology segment, contributing the largest market revenue share in 2025, as it represents the globally recognized gold standard for bone mineral density measurement — valued for its high precision, strong reproducibility, low radiation dose, and ability to deliver comprehensive diagnostic information including fracture risk stratification, vertebral fracture analysis, and body composition measurement.
-
Quantitative Ultrasound (QUS) is the fastest-growing technology segment, projected to expand at a significant CAGR through 2033, driven by its radiation-free operation, compact and portable device format, lower cost per test, ease of use without dedicated technical staff, and suitability for deployment in diverse clinical settings — particularly in community screening programs and emerging market healthcare facilities where DXA infrastructure is limited or unavailable.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 550.03 Million |
| Market Size by 2025 | USD 369.70 Million |
| Market Size by 2026 | USD 384.69 Million |
| Market Growth Rate (2026–2033) | CAGR of 3.45% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Segments Covered | Product, Technology, End User |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Rising Global Osteoporosis Prevalence, Aging Demographics, Expanding DXA Screening Mandates, and AI-Integrated Bone Densitometry Innovation Are Together Creating a Stable and Growing Commercial Demand Environment
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rapidly aging global population driving osteoporosis and low BMD prevalence | ~34% | Global — especially North America, Europe, Asia Pacific | Short to Long-term |
| Expanding clinical guidelines for routine bone density screening in high-risk groups | ~28% | North America, Europe | Short to Medium-term |
| Growing adoption of preventive healthcare frameworks and fracture liaison services | ~24% | North America, Europe, Asia Pacific | Medium-term |
| AI-enhanced DXA platforms expanding clinical utility and diagnostic value | ~14% | North America, Europe | Medium to Long-term |
The most durable and structurally embedded driver of the Bone Densitometer Devices market is the relentless global aging trend, which is continuously expanding the at-risk population for osteoporosis, low bone mineral density, and fragility fractures at a scale that existing diagnostic infrastructure is often unable to fully serve. As the proportion of people aged 65 and older grows across both developed and emerging economies, the clinical imperative for proactive bone density screening expands proportionally — creating a demand trajectory that is fundamentally demographic in nature and therefore exceptionally stable across economic cycles. National health authorities in North America and Europe have responded to this growing burden by strengthening routine DXA screening recommendations for postmenopausal women and older men, directly converting demographic aging into expanding institutional demand for bone densitometer devices across hospitals, diagnostic centers, and orthopedic clinics.
The progressive adoption of fracture liaison services as a standard component of hospital orthopedic care — now well established across North America, the United Kingdom, Scandinavia, and Australia — is creating a reliable and growing institutional demand channel for bone densitometer equipment within hospital departments. These systematic post-fracture secondary prevention programs require dependable, high-throughput bone density testing infrastructure to evaluate every fragility fracture patient for underlying osteoporosis and to monitor treatment response over time, generating consistent device utilization and institutional procurement volumes. The demonstrated cost-effectiveness of fracture liaison services in reducing re-fracture rates and long-term fracture-related healthcare costs is motivating their adoption by an expanding number of hospital systems globally, progressively widening the institutional demand base for bone densitometer devices throughout the forecast period.

Restraints Impact Analysis
High Capital Cost of Central DXA Systems, Limited Reimbursement in Emerging Markets, Radiation Exposure Concerns, and Cross-Device Standardization Gaps Restrain Faster Bone Densitometer Devices Market Penetration
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High capital acquisition cost of central DXA systems | ~33% | Latin America, MEA, parts of Asia Pacific | Short to Medium-term |
| Limited insurance and government reimbursement for DXA testing in emerging markets | ~29% | Asia Pacific, Latin America, MEA | Short to Medium-term |
| Radiation exposure concerns associated with X-ray-based bone densitometry | ~22% | Global | Short-term |
| Lack of cross-device standardization complicating serial BMD monitoring | ~16% | Global | Medium-term |
The high capital cost of central DXA systems — typically ranging from USD 70000 to over USD 150000 per unit depending on configuration and software capabilities — represents the most significant barrier to first-time adoption of bone densitometer devices in lower-income healthcare settings, community clinics, and emerging market hospitals where large medical equipment budgets are constrained by competing clinical investment priorities. This cost barrier is particularly acute in Latin America, sub-Saharan Africa, and parts of Southeast Asia, where the significant capital commitment required for a central DXA installation competes with more immediately visible clinical priorities such as surgical equipment and basic imaging infrastructure. While lower-cost peripheral and portable QUS devices offer a more affordable alternative, their clinical limitations relative to central DXA — particularly their inability to measure BMD at the hip and lumbar spine, the two most clinically important fracture risk sites — prevent them from substituting fully for DXA in evidence-based osteoporosis diagnosis and management.
The absence of comprehensive reimbursement frameworks for routine bone density testing in many emerging and middle-income markets creates a fundamental structural impediment to Bone Densitometer Devices market penetration beyond established healthcare systems. In markets where DXA scanning lacks clear insurance or government health program reimbursement — including large patient populations across China, India, Brazil, and much of the Middle East — the economic incentive for healthcare facilities to invest in bone densitometer devices is significantly reduced, suppressing device adoption despite growing clinical need. Additionally, the lack of cross-device measurement standardization across different DXA manufacturers complicates the serial BMD monitoring that is essential for effective long-term osteoporosis management, creating clinical uncertainty when patients transition between scanning facilities with different device brands and software platforms.
Opportunities Impact Analysis
Emerging Market Healthcare Infrastructure Expansion, Portable Point-of-Care Densitometry Innovation, AI-Powered Fracture Risk Analysis, and Opportunistic Osteoporosis Screening From Routine Imaging Create Compelling Growth Opportunities
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Healthcare infrastructure expansion in Asia Pacific, Latin America, and MEA | ~34% | Asia Pacific, Latin America, MEA | Medium to Long-term |
| Portable and point-of-care bone densitometry enabling community screening | ~28% | Global — especially emerging markets | Short to Medium-term |
| AI-powered opportunistic osteoporosis screening from routine X-ray imaging | ~26% | North America, Europe, Asia Pacific | Medium to Long-term |
| DXA body composition analysis expanding into metabolic and sports medicine | ~12% | North America, Europe | Short to Medium-term |
The rapid expansion of healthcare infrastructure across Asia Pacific, Latin America, and the Middle East represents the most strategically important long-term growth opportunity for the Bone Densitometer Devices market. India's government-initiated establishment of approximately 150000 Health and Wellness Centers is expanding diagnostic service access across rural and semi-urban populations at an unprecedented scale, creating a new institutional demand channel for accessible bone health screening equipment in one of the world's most populous high-osteoporosis-burden countries. As hospital networks expand and diagnostic imaging capabilities are upgraded across these high-population regions, the installed base of bone density testing equipment will grow substantially — driven by first-time device adoption rather than replacement cycles, creating genuinely new commercial volume for device manufacturers.
The development and growing clinical validation of AI-powered opportunistic osteoporosis screening tools — which can estimate bone mineral density from routine X-ray, CT, or MRI images obtained for entirely different clinical indications — represents a transformative opportunity to dramatically expand the population screened for low bone density without requiring dedicated densitometer appointments. These AI tools effectively convert millions of routine imaging studies performed annually into opportunistic bone health assessments, potentially identifying large numbers of undiagnosed osteoporosis patients who would then proceed to formal DXA confirmation scanning — generating meaningful incremental demand for bone densitometer devices globally. As these AI screening tools receive regulatory clearance and achieve clinical integration in major healthcare systems, they are expected to meaningfully expand the bone densitometry testing funnel and support sustained market growth throughout the 2026–2033 forecast period.

Segment Analysis
By Technology
DXA Technology Dominates the Bone Densitometer Devices Market as the Uncontested Clinical Gold Standard, While Quantitative Ultrasound Rapidly Emerges as the Fastest-Growing Modality for Accessible Community and Emerging Market Screening
Dual Energy X-Ray Absorptiometry holds a commanding dominant position within the technology segment of the Bone Densitometer Devices market, accounting for approximately 62.80% of total global market revenue in 2025. DXA's clinical supremacy is rooted in its global recognition as the gold standard for bone mineral density measurement — endorsed by the WHO, the International Society for Clinical Densitometry, and national osteoporosis guidelines across virtually every major healthcare market worldwide. Its capacity to precisely measure BMD at the clinically critical hip and lumbar spine sites, combined with low radiation exposure, excellent measurement precision, and the additional ability to perform vertebral fracture analysis and whole-body composition measurements within a single examination session, makes DXA the most diagnostically comprehensive and clinically versatile bone density assessment tool available. North America accounts for the largest DXA technology revenue share globally, where high screening rates, strong Medicare and private insurance reimbursement, and dense device installations across hospitals and imaging centers generate the highest DXA scan procedure volumes. Hologic — with its Horizon DXA system family — and GE HealthCare with its Lunar Prodigy and iDXA platforms are the two dominant manufacturers in the global DXA segment, together commanding the largest combined market share.
Quantitative Ultrasound represents the fastest-growing technology segment in the Bone Densitometer Devices market, projected to expand at a CAGR of approximately 5.80% from 2026 to 2033, driven by its compelling practical advantages for point-of-care and community-based bone health screening. QUS devices are radiation-free, compact, portable, and substantially less expensive than central DXA systems, making them highly practical for deployment in primary care offices, mobile health screening programs, and healthcare facilities in emerging markets where dedicated DXA infrastructure is unavailable. Asia Pacific is the fastest-growing region for QUS device adoption, driven by the enormous underserved rural and semi-urban population across India, China, and Southeast Asia that lacks access to hospital-based DXA facilities. Key companies active in the QUS segment include Echolight S.p.A., Sunlight Medical Ltd. (BeamMed), and Osteometer MediTech, all of which are advancing portable and point-of-care QUS platforms targeting emerging market and community screening applications.
By Product
Axial Bone Densitometers Lead the Market With the Highest Clinical Accuracy and Revenue Contribution, While Peripheral Bone Densitometers Grow Fastest as Accessible Screening Tools in Outpatient and Community Settings
Axial bone densitometers — primarily central DXA systems measuring bone mineral density at the lumbar spine and proximal femur — dominate the product segment of the Bone Densitometer Devices market, accounting for approximately 67.40% of total global market revenue in 2025. Their dominance reflects the clinical gold-standard status of central DXA for osteoporosis diagnosis and fracture risk stratification, with established clinical guidelines across North America and Europe universally specifying axial DXA measurements at the hip and spine as the definitive diagnostic standard for osteoporosis. Hospitals and high-volume diagnostic imaging centers in North America and Europe represent the core buyer segment for axial bone densitometers, generating the largest institutional procurement volumes and the highest per-device revenue contributions in the market. Hologic and GE HealthCare dominate the global axial bone densitometer product segment, with their respective DXA platforms holding leading positions across hospital and diagnostic center installations in the United States, Canada, Western Europe, and Japan.
Peripheral bone densitometers — including pDXA, QUS, and radiographic absorptiometry systems measuring BMD at peripheral skeletal sites such as the heel, wrist, and finger — are the fastest-growing product segment in the Bone Densitometer Devices market, projected to expand at a CAGR of approximately 5.20% from 2026 to 2033. Their growth is driven by growing adoption in outpatient clinics, primary care settings, and community health programs where compact, affordable, and radiation-free or low-radiation peripheral devices provide a practical and accessible means of initial bone health screening. Diagnostic centers represent the fastest-growing end-user segment for peripheral bone densitometers, particularly across Asia Pacific and Latin America where these devices are being adopted as cost-effective entry-point tools for opportunistic osteoporosis screening in settings where full central DXA installation is not yet feasible. Companies including Echolight S.p.A., BeamMed, and Osteometer MediTech are among the primary providers of peripheral bone densitometer systems targeting these fast-growing outpatient and emerging market applications.

Regional Insights
North America
North America Leads the Global Bone Densitometer Devices Market With the Highest DXA Scan Utilization, Strongest Clinical Screening Infrastructure, and Most Advanced Osteoporosis Diagnosis and Management Ecosystem
North America holds the dominant position in the global Bone Densitometer Devices market, accounting for approximately 38.20% of total global revenue in 2025, and is projected to maintain its regional leadership at a CAGR of approximately 3.20% from 2026 to 2033. The United States is the primary revenue driver within the region, where well-established USPSTF and National Osteoporosis Foundation guidelines for routine DXA screening in postmenopausal women and older adults, strong Medicare and private insurance reimbursement for bone density testing, a highly developed network of diagnostic imaging centers and hospital-based bone health programs, and the presence of leading bone densitometer device manufacturers — including Hologic and GE HealthCare — collectively create the world's most commercially developed Bone Densitometer Devices market. The region benefits from high physician and patient awareness of osteoporosis as a preventable fracture risk condition, widespread adoption of fracture liaison services in major hospital systems, and growing utilization of advanced DXA body composition analysis across metabolic disease and sports medicine clinical applications.
Canada contributes meaningfully to North American market revenue through its provincial healthcare systems, which include DXA testing coverage for eligible high-risk populations, and through a growing private diagnostic imaging sector serving patients seeking routine preventive bone health screening. The strong institutional presence of Osteoporosis Canada — which advocates for evidence-based bone density screening access and supports clinical guideline adherence — reinforces consistently high testing volumes and device utilization rates across the Canadian market. North America's market leadership is expected to remain secure through 2033, supported by continuous device innovation from Hologic and GE HealthCare, progressive integration of AI-assisted bone health analytics, and growing clinical momentum around fracture prevention program expansion.
Asia Pacific
Asia Pacific Is the Fastest-Growing Bone Densitometer Devices Market, Powered by the World's Largest Aging Population, Expanding Healthcare Infrastructure, and Growing Government Investment in Osteoporosis Screening Programs
Asia Pacific is the fastest-growing regional segment in the Bone Densitometer Devices market, projected to expand at a CAGR of approximately 4.60% from 2026 to 2033 — the highest of any region globally. The region is home to the world's largest and most rapidly aging population, with China, Japan, India, and South Korea collectively accounting for an enormous and growing osteoporosis burden that represents the most significant untapped commercial opportunity in global bone densitometry. Japan — with the world's oldest population by proportion — is the most mature and largest bone densitometer device market within Asia Pacific, characterized by high DXA installation density, well-developed osteoporosis screening guidelines, and strong clinical infrastructure for bone health management. China and India represent the region's most significant growth frontiers, where rapid healthcare facility expansion, fast-growing elderly populations, and rising government and clinical awareness of osteoporosis are driving accelerating procurement of bone density testing equipment. Key companies with strong Asia Pacific presence in the Bone Densitometer Devices market include Hologic, GE HealthCare, Furuno Electric Co., Osteometer MediTech, and Echolight S.p.A.
South Korea and Australia are also important contributors to Asia Pacific market revenue, where established government-funded DXA screening programs and high physician awareness of fracture risk generate consistent device utilization and procurement activity. India's government-led expansion of Health and Wellness Centers — targeting approximately 150000 facilities nationwide — is progressively creating a new accessible institutional channel for bone health screening device adoption across India's vast and largely underserved rural and semi-urban population base. This government-driven healthcare access expansion, combined with the rapid growth of private hospital networks and diagnostic imaging chains across Tier 1 and Tier 2 Indian cities, positions Asia Pacific as the most commercially dynamic and fastest-growing regional market in global bone densitometry through 2033.
Top Key Players
-
Hologic, Inc. (United States)
-
GE HealthCare Technologies Inc. (United States)
-
Siemens Healthineers AG (Germany)
-
Philips Healthcare (Netherlands)
-
Fujifilm Holdings Corporation (Japan)
-
Osteometer MediTech A/S (Denmark)
-
Echolight S.p.A. (Italy)
-
Sunlight Medical Ltd. — BeamMed (Israel)
-
Swissray International AG (Switzerland)
-
Demetech AB (Sweden)
-
Scanflex Healthcare AB (Sweden)
-
Medlink Imaging (France)
Recent Developments
-
Hologic, Inc. (2025) — Launched an enhanced version of its Horizon DXA system platform incorporating updated AI-assisted vertebral fracture analysis (VFA) capabilities and expanded body composition analytics, enabling clinicians to extract richer diagnostic insights from routine bone density examinations and strengthening Hologic's position as the leading global provider of advanced DXA-based bone health assessment solutions.
-
GE HealthCare (2024) — Announced the integration of advanced AI-powered bone mineral density analysis tools into its Lunar DXA system software suite, designed to improve measurement reproducibility, automate report generation, and enhance fracture risk classification — reinforcing GE HealthCare's commitment to elevating the clinical utility of its bone densitometer platforms through intelligent imaging analytics.
-
Echolight S.p.A. (2025) — Expanded the international market presence of its ECHOSTATION radiation-free bone densitometry platform — which uses quantitative ultrasound imaging to assess bone quality without ionizing radiation — securing new distribution agreements across Asia Pacific and Middle Eastern markets where demand for radiation-free, portable bone assessment devices is growing among primary care and community health settings.
-
Siemens Healthineers (2024) — Advanced its musculoskeletal imaging portfolio with new AI-assisted bone density assessment capabilities integrated into its CT and X-ray platforms, enabling opportunistic osteoporosis screening from routine imaging examinations — a development aligned with the growing clinical momentum toward broader population-level bone health identification beyond dedicated DXA appointments.
-
BeamMed (Sunlight Medical) (2025) — Released an upgraded version of its Sunlight Omnisense portable quantitative ultrasound bone densitometer featuring enhanced measurement accuracy, improved software interface, and expanded connectivity for cloud-based reporting — targeting primary care, orthopedic clinic, and mobile health screening applications in emerging markets where accessible and affordable bone density assessment tools are in strong demand.
Market Trends
AI-Driven Opportunistic Osteoporosis Screening From Routine Imaging and the Growing Clinical Adoption of Portable Bone Densitometry for Community Health Programs Are the Two Defining Trends Reshaping the Bone Densitometer Devices Market
The most transformative trend in the Bone Densitometer Devices market is the rapid development and progressive clinical integration of AI-powered tools capable of detecting low bone mineral density opportunistically from routine X-ray, CT, and MRI examinations performed for entirely different clinical indications. These AI systems — trained on large imaging datasets with corresponding DXA BMD measurements — can automatically flag patients with signs of low bone density or vertebral fractures from standard imaging studies, enabling a fundamentally new and scalable model for population-level osteoporosis identification that does not depend on dedicated densitometer appointments. As these AI screening tools receive regulatory clearance and are integrated into hospital radiology and electronic health record workflows, they are expected to generate meaningful new DXA referral volumes — expanding the bone densitometry testing funnel and driving incremental demand for bone densitometer devices globally.
The second major trend reshaping the bone densitometer devices market is the growing clinical and commercial momentum behind portable, compact, and radiation-free bone densitometry technologies — particularly QUS and peripheral DXA devices — as practical tools for osteoporosis risk screening in primary care, community health programs, and emerging market healthcare settings. The simplicity of operation, low maintenance requirements, absence of radiation, and substantially lower acquisition cost of portable bone densitometry platforms make them ideal for deployment across a wide range of settings where central DXA is not available or economically viable. As healthcare access improves in Asia Pacific, Latin America, and sub-Saharan Africa, these portable screening devices are increasingly being adopted as first-line bone health assessment tools that can identify at-risk individuals for referral to definitive DXA evaluation — extending the reach of osteoporosis screening programs and supporting market growth in geographies that are currently underserved by conventional densitometry infrastructure.
Segments Covered in the Report
By Product:
-
Peripheral Bone Densitometer
-
Axial Bone Densitometer
By Technology:
-
Dual Energy X-Ray Absorptiometry (DXA/DEXA)
-
Peripheral Dual Energy X-Ray Absorptiometry (pDXA)
-
Quantitative Ultrasound (QUS)
-
Quantitative Computed Tomography (QCT)
-
Other Technologies
By End User:
-
Hospitals
-
Orthopedic Clinics
-
Diagnostic Centers
-
Academic and Research Institutions
By Region:
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the Bone Densitometer Devices market and what is its projected value by 2033?
Answer: The Bone Densitometer Devices market was valued at USD 369.70 million in 2025 and is projected to reach USD 550.03 million by 2033. It is expected to grow at a CAGR of 3.45% from 2026 to 2033, driven by rising global osteoporosis prevalence, expanding clinical screening guidelines, and growing healthcare infrastructure investment across emerging markets.
Question 2: What are the primary growth drivers of the Bone Densitometer Devices market?
Answer: The rapidly aging global population driving higher osteoporosis prevalence and expanding clinical guidelines for routine DXA screening in high-risk groups are the two most powerful drivers of the Bone Densitometer Devices market. Growing adoption of fracture liaison services in hospital systems and the integration of AI-enhanced analytics into modern densitometry platforms are also meaningfully supporting market growth.
Question 3: Which region leads the Bone Densitometer Devices market and which is the fastest growing?
Answer: North America holds the dominant position in the Bone Densitometer Devices market with approximately 38.20% of global revenue in 2025, driven by strong reimbursement frameworks and high DXA utilization. Asia Pacific is the fastest-growing region, expanding at approximately 4.60% CAGR from 2026 to 2033, powered by a massive aging population and rapidly expanding healthcare infrastructure.
Question 4: Which technology and product segment dominate the Bone Densitometer Devices market?
Answer: Dual Energy X-Ray Absorptiometry dominates the technology segment with approximately 62.80% of total Bone Densitometer Devices market revenue in 2025 as the globally recognized clinical gold standard for BMD measurement. Axial bone densitometers lead the product segment with approximately 67.40% market share, while Quantitative Ultrasound and peripheral densitometers represent the fastest-growing segments respectively.
Question 5: How is AI shaping the future of the Bone Densitometer Devices market?
Answer: AI is transforming the Bone Densitometer Devices market by enabling automated fracture detection, opportunistic osteoporosis screening from routine imaging, and AI-assisted BMD analysis that reduces inter-operator variability and improves diagnostic consistency. Leading manufacturers including Hologic and GE HealthCare are actively integrating AI-powered analytics into their next-generation DXA platforms to deliver enhanced diagnostic value and improved clinical workflow efficiency.
