Biologics And Biosimilars Market Overview
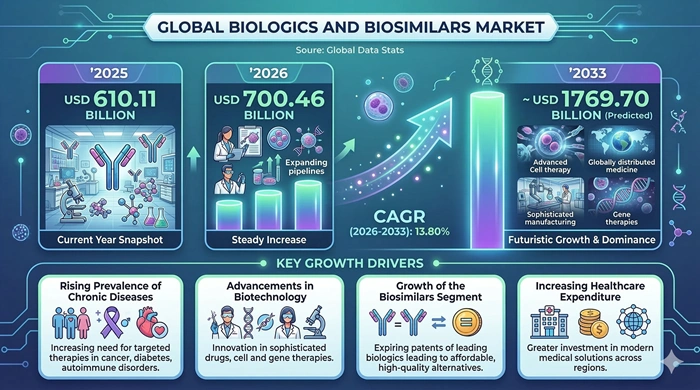
The global biologics and biosimilars market size is valued at USD 610.11 billion in 2025 and is predicted to increase from USD 700.46 billion in 2026 to approximately USD 1769.70 billion by 2033, growing at a CAGR of 13.80% from 2026 to 2033. Rising prevalence of chronic and autoimmune diseases, expanding biologic pipelines, and accelerating biosimilar approvals are reshaping treatment pathways and healthcare budgets worldwide. Payers and providers are increasingly focused on balancing access to innovative biologics with the cost-saving potential of biosimilar alternatives in key therapy areas such as oncology, immunology, and endocrinology.
AI-Powered Transformation Of Biologics And Biosimilars Supply, Access, And Innovation
Artificial intelligence is rapidly transforming how the biologics and biosimilars market discovers, develops, and delivers complex therapies. AI tools are used to optimize antibody design, predict protein folding, and screen thousands of candidates in silico, reducing early R&D time and cost. They also support smarter clinical trial design by identifying high‑probability responders, improving recruitment, and reducing protocol amendments.
Across manufacturing, AI-driven process control helps maintain consistent quality in cell culture, purification, and fill-finish steps, which is critical for regulatory compliance and patient safety. In commercial operations, machine-learning models forecast demand for biologics and biosimilars, inform pricing strategies, and guide market access decisions across different regions and payer systems. As more real‑world data flows from electronic health records and registries, AI will enable continuous safety monitoring and outcome tracking to support substitution and interchangeability of biosimilars.
Growth Factors
Multiple Demand, Access, And Technology Drivers Fueling Strong Multi-Year Expansion
Several structural trends are pushing robust growth in the biologics and biosimilars market through 2033. The global burden of cancer, autoimmune conditions, diabetes, and rare genetic diseases is increasing, and clinicians are shifting from small‑molecule drugs to targeted biologic therapies with superior efficacy and more personalized mechanisms of action. At the same time, numerous blockbuster biologics are losing patent protection, opening the door for biosimilars that can offer comparable clinical outcomes at lower cost.
Governments and private payers are actively promoting biosimilar uptake through tendering, reference pricing, and formulary incentives, particularly in North America and Europe. Regulatory frameworks from agencies such as the FDA and EMA now provide clearer pathways for biosimilar approvals and interchangeability designations, which improves physician confidence and speeds market entry. Advances in manufacturing platforms, including single‑use bioreactors and improved expression systems, also reduce production cost per unit and support competitive pricing strategies.

Market Outlook
Sustained Double-Digit Growth With Rising Share Of Biosimilars And Emerging Market Momentum
Between 2026 and 2033, the biologics and biosimilars market is expected to maintain a strong double‑digit growth trajectory, underpinned by a steady flow of new biologic launches and expanding biosimilar portfolios. Originator biologics will continue to generate the majority of revenue in the near term, but the share of biosimilars will rise as more reference products lose exclusivity in oncology, immunology, and endocrinology. This will gradually rebalance spending patterns, with biosimilars absorbing volume growth and freeing budgets for next‑generation biologics such as cell and gene therapies.
Regionally, North America is projected to retain the largest revenue share, while Asia Pacific will post the fastest CAGR as local manufacturers scale up development and export‑oriented strategies. By 2033, increased biosimilar penetration, broader insurance coverage, and improved healthcare infrastructure in emerging economies should significantly enlarge the treated patient pool. The overall market outlook remains positive, though pricing pressure, regulatory complexity, and manufacturing challenges will require continuous strategic adaptation.
Expert Speaks
-
CEOs of leading global pharma companies note that rising chronic disease prevalence and sustained innovation in monoclonal antibodies and targeted biologics are keeping demand in the biologics and biosimilars market well above historical averages. They emphasize that payers are demanding clear value evidence and real‑world outcomes data to justify premium pricing.
-
Leaders of major biotechnology firms highlight that biosimilars are now viewed as strategic growth engines, not just defensive plays, particularly in oncology and autoimmune therapy classes. They point to maturing regulatory guidance and greater physician familiarity as key enablers of faster biosimilar adoption.
-
Executives from diversified healthcare groups stress that manufacturing reliability, supply resilience, and proactive risk management across global sites are becoming as important as clinical efficacy in shaping competitive advantage. They also underline the growing role of digital and AI tools in optimizing end‑to‑end operations.
Key Report Takeaways
-
North America leads the market: North America currently accounts for the largest share of the global biologics and biosimilars market, supported by high healthcare spending, rapid adoption of innovative biologics, and well‑defined biosimilar regulatory pathways that encourage competition without compromising safety.
-
Asia Pacific is growing the fastest: Asia Pacific is projected to record the highest CAGR through 2033 as countries such as China, India, and South Korea expand biologic manufacturing capacity, streamline approval processes, and invest in broader access to cost‑effective biosimilars across public and private health systems.
-
Specialty care customers drive usage: Hospitals, specialty clinics, and oncology centers represent the most intensive users of biologics and biosimilars, as they manage complex conditions requiring targeted therapies, sophisticated administration, and close patient monitoring.
-
Oncology is the largest application: Oncology applications contribute the highest revenue share to the biologics and biosimilars market, reflecting the dominance of monoclonal antibodies, checkpoint inhibitors, and other targeted biologics in cancer treatment protocols and guidelines worldwide.
-
Monoclonal antibody platforms are most prominent: Among biologic modalities, monoclonal antibodies remain the most popular, owing to their well‑established clinical track record, scalable manufacturing processes, and broad applicability across oncology, autoimmune diseases, and inflammatory disorders.
-
Biosimilar oncology segment will grow quickly: Biosimilar oncology products are expected to gain share rapidly, with some estimates pointing to high‑teens CAGR as more reference cancer biologics face patent expiry, supporting an expanding pipeline and increasing physician and payer willingness to switch patients from reference brands.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 1769.70 billion |
| Market Size by 2025 | USD 610.11 billion |
| Market Size by 2026 | USD 700.46 billion |
| Market Growth Rate from 2026 to 2033 | CAGR of 13.80% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2033 |
| Segments Covered | Product Type, Molecule Type, Therapy Area, End User, Region |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Balanced Interplay Of Innovation, Cost Pressure, And Access Expansion Shaping Long-Term Growth
Driver Impact Analysis
Demand And Innovation Tailwinds Supporting Robust Expansion Across Major Therapy Areas
Key drivers in the biologics and biosimilars market include the rising incidence of cancer, autoimmune disorders, and metabolic diseases, which pushes demand for targeted and high‑efficacy therapies. Biologics often deliver superior clinical outcomes compared with conventional small molecules, encouraging their inclusion in treatment guidelines and reimbursement lists. Patent expiries of high‑revenue biologics continue to open space for biosimilar competitors, which can offer meaningful cost savings while preserving therapeutic value.
In parallel, advances in biotechnology, cell line engineering, and process intensification are boosting yields and lowering unit costs, making both biologics and biosimilars more commercially attractive. Regulatory bodies have also refined biosimilar approval criteria, giving manufacturers clearer expectations for demonstrating similarity, immunogenicity, and interchangeability. Together, these drivers increase patient access, support higher treatment volumes, and underpin sustained market growth.
| Drivers | ≈ % Impact On CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising chronic disease burden | High | Global | Short to long term |
| Biologic innovation and pipeline expansion | High | North America, Europe, Asia Pacific | Medium to long term |
| Patent expiries and biosimilar entry | High | Global | Short to medium term |
| Supportive reimbursement and access policies | Medium | North America, Europe | Medium term |

Restraints Impact Analysis
Cost, Complexity, And Regulatory Nuances Temper The Pace Of Market Penetration
Despite strong drivers, several restraints limit the full potential of the biologics and biosimilars market. Biologics are complex to manufacture, requiring specialized facilities, strict quality systems, and significant capital expenditure, which raises entry barriers for smaller firms. Pricing pressures from payers, combined with intense competition among originators and biosimilars, can compress margins and challenge long‑term profitability.
Physician and patient hesitancy toward switching from originator biologics to biosimilars can slow uptake, especially in sensitive indications where clinical familiarity and brand trust are deeply entrenched. Additionally, diverse regulatory and interchangeability standards across regions create fragmentation and increase the cost and complexity of global launches. Supply chain vulnerabilities, such as cold‑chain disruptions, also pose risks to consistent product availability.
| Restraints | ≈ % Impact On CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High manufacturing and development costs | Medium | Global | Medium to long term |
| Pricing and reimbursement pressure | Medium to high | North America, Europe | Short to medium term |
| Physician and patient switching hesitancy | Medium | Global | Short to medium term |
| Regulatory complexity and variability | Medium | Global | Medium term |
Opportunities Impact Analysis
New Modalities, Emerging Markets, And Expanded Indications Create Upside Potential
Significant opportunities exist to expand the biologics and biosimilars market over the forecast horizon. Emerging markets in Asia Pacific, Latin America, and the Middle East are investing in healthcare infrastructure and reimbursement schemes that support broader access to biologics and biosimilars. Local manufacturers in these regions are building capabilities to supply domestic and export markets, often with cost advantages.
There is also growing scope to develop biosimilars for more complex biologics and newer modalities as regulatory and technological experience increases. Expansion into additional indications, including earlier lines of therapy and combination regimens, will broaden the treated population. Partnerships between global innovators and regional players, along with contract manufacturing and co‑development models, can accelerate portfolio expansion and geographic reach.
| Opportunities | ≈ % Impact On CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Expansion in emerging markets | High | Asia Pacific, Latin America, MEA | Medium to long term |
| Next‑wave biosimilars and complex biologics | Medium to high | Global | Medium to long term |
| Strategic collaborations and outsourcing | Medium | Global | Short to medium term |

Segment Analysis
Concentrated Revenue In Originator Biologics With Rapidly Scaling Biosimilars Contribution
By Molecule Type – Originator Biologics
Established Biologic Portfolios Anchor Revenue With Broad Indication Coverage And Strong Brand Equity
Originator biologics currently command the majority share of the biologics and biosimilars market, with estimates indicating that original biologics contributed more than four‑fifths of total revenue in the mid‑2020s. This segment benefits from strong clinical evidence, entrenched prescribing patterns, and extensive commercialization infrastructure built over many years. Originator products are widely used in oncology, autoimmune diseases, and metabolic disorders, often occupying first‑line or key subsequent‑line positions in treatment algorithms.
Although growth in this segment is gradually moderating as more products face patent expiry, it still delivers a healthy single‑ to high‑single‑digit CAGR globally. North America and Europe remain the core revenue centers due to high biologic adoption and premium pricing environments. Large multinational companies dominate this space, leveraging broad pipelines, lifecycle management strategies, and ongoing indication expansions. Continued innovation in mechanisms of action and delivery formats will help sustain momentum.
By Molecule Type – Biosimilars
High-Growth Segment Leveraging Patent Cliffs And Cost Pressures To Expand Global Access
The biosimilars segment is evolving into the fastest‑growing component of the biologics and biosimilars market, posting a robust double‑digit CAGR as more reference biologics lose exclusivity. Biosimilars are particularly attractive in budget‑constrained health systems, where they can generate substantial savings while maintaining similar safety and efficacy profiles. Oncology, immunology, and supportive care therapies are leading areas for biosimilar penetration, with multiple molecules already launched or in late‑stage development.
Europe initially led in biosimilar adoption, but North America and Asia Pacific are now catching up as regulatory policies mature and payer incentives strengthen. The segment’s market share is set to rise steadily through 2033, especially in hospital and specialty clinic settings that can aggregate demand through tenders. A mix of global pharmaceutical companies and regional manufacturers are active here, pursuing strategies such as portfolio breadth, competitive pricing, and value‑added services to differentiate offerings.

Regional Insights
North America – Dominating Region
High Biologic Adoption, Strong Pricing Power, And Structured Biosimilar Pathways Underpin Leadership
North America holds the largest share of the global biologics and biosimilars market, supported by advanced healthcare infrastructure, high per‑capita spending, and rapid uptake of innovative therapies. The United States, in particular, dominates regional revenue and benefits from a large population of patients with cancer, autoimmune conditions, and chronic metabolic diseases. An increasing number of biosimilars are being approved and launched under well‑defined regulatory pathways, gradually reshaping competitive dynamics.
Key players in this region include major multinational pharmaceutical and biotechnology companies with extensive biologics portfolios and expanding biosimilar lines. North America is projected to maintain a substantial market share through 2033, with a strong double‑digit CAGR in selected therapy areas where biosimilar competition intensifies and new biologics enter the market. Ongoing policy discussions around pricing, interchangeability, and formulary management will critically influence future adoption and revenue patterns.
Asia Pacific – Fastest Growing Region
Local Manufacturing Scale-Up And Policy Support Driving Accelerated Growth From A Lower Base
Asia Pacific is expected to record the fastest growth rate in the biologics and biosimilars market over the forecast period, albeit from a smaller base compared with North America and Europe. Rising healthcare expenditure, expanding insurance coverage, and growing awareness of advanced therapies are driving higher demand across major countries such as China, India, South Korea, and Japan. Governments are encouraging domestic biologic and biosimilar manufacturing to improve affordability and reduce reliance on imports.
Regional players are increasingly active in developing biosimilars for both local and export markets, often forming alliances with global companies to access technology and regulatory expertise. Asia Pacific is projected to deliver a high‑teens CAGR through 2033, with oncology, autoimmune disease, and diabetes care as particularly dynamic therapy areas. The combination of local cost advantages and improving quality standards positions the region as both a major consumption and production hub for biologics and biosimilars.
Top Key Players
-
Roche Holding AG (Switzerland)
-
Amgen Inc (United States)
-
Pfizer Inc (United States)
-
Novartis AG (Switzerland)
-
Johnson & Johnson (United States)
-
Merck & Co Inc (United States)
-
AbbVie Inc (United States)
-
Samsung Biologics (South Korea)
-
Celltrion Healthcare (South Korea)
-
Biocon Biologics (India)
Recent Developments
-
2025 – Roche Holding AG (Switzerland): The company expanded its oncology biologics portfolio with additional indication approvals and advanced lifecycle management strategies to defend share against emerging biosimilars. It also increased investment in real‑world evidence programs to support outcomes‑based discussions with payers.
-
2025 – Amgen Inc (United States): Amgen launched new biosimilars in immunology and oncology across North America and Europe, strengthening its position as a key participant in both originator and biosimilar segments of the biologics and biosimilars market. The firm also invested in next‑generation manufacturing technologies.
-
2025 – Pfizer Inc (United States): Pfizer broadened access to its biosimilar portfolio in emerging markets through strategic partnerships and expanded distribution agreements. It focused on physician education and pharmacovigilance to support switching confidence.
-
2024 – Novartis AG (Switzerland): Novartis advanced several pipeline biosimilars into late‑stage development and leveraged its Sandoz business to strengthen global reach, particularly in Europe and Asia Pacific. The company also emphasized sustainability and supply resilience across its biologics network.
-
2024 – Biocon Biologics (India): Biocon scaled up its contract manufacturing and co‑development services while expanding biosimilar launches in North America and Europe. This helped reinforce India’s role as an important production base for global biologics and biosimilars.
Market Trends
Evolving Competitive Models, Access Strategies, And Technology Adoption Across The Value Chain
Key trends in the biologics and biosimilars market include rising emphasis on interchangeability and switching studies, as stakeholders seek stronger evidence to support broader substitution at the pharmacy and payer levels. There is growing use of value‑based contracts and outcomes‑linked reimbursement for high‑cost biologics, which encourages manufacturers to generate robust real‑world data and patient support programs. Digital tools and AI are increasingly used to monitor adherence, manage side effects, and optimize dosing patterns.
Another important trend is the shift toward more integrated supply networks and risk‑sharing arrangements between originators, biosimilar manufacturers, and healthcare systems. Companies are diversifying manufacturing footprints to reduce supply chain risk and exploring modular, flexible facilities to respond quickly to demand changes. Sustainability considerations, such as energy efficiency and waste reduction in biologic production, are gaining prominence and may influence long‑term investment decisions.
Segments Covered In The Report
-
By Product Type
-
Monoclonal Antibodies
-
Vaccines
-
Recombinant Hormones
-
Cytokines And Growth Factors
-
Fusion Proteins
-
Others
-
-
By Molecule Type
-
Originator Biologics
-
Biosimilars
-
-
By Therapy Area
-
Oncology
-
Autoimmune And Inflammatory Diseases
-
Diabetes And Metabolic Disorders
-
Blood Disorders
-
Infectious Diseases
-
Others
-
-
By End User
-
Hospitals
-
Specialty Clinics
-
Retail And Specialty Pharmacies
-
Others
-
-
By Region
-
North America (United States, Canada)
-
Europe (Germany, United Kingdom, France, Rest of Europe)
-
Asia Pacific (China, India, Japan, South Korea, Rest of Asia Pacific)
-
Latin America (Brazil, Mexico, Rest of Latin America)
-
Middle East & Africa (GCC Countries, South Africa, Rest of MEA)
-
Frequently Asked Questions
Question 1: What is driving growth in the biologics and biosimilars market?
Answer: Growth in the biologics and biosimilars market is driven by rising chronic disease burden and strong demand for targeted therapies. Loss of exclusivity for major biologics and supportive regulatory frameworks further accelerate biosimilar uptake.
Question 2: How fast will the biologics and biosimilars market grow from 2026 to 2033?
Answer: The biologics and biosimilars market is expected to grow at a CAGR of about 13.80 percent between 2026 and 2033. This reflects sustained innovation in biologics and rapid expansion of biosimilar portfolios globally.
Question 3: Which region leads the biologics and biosimilars market today?
Answer: North America currently leads the biologics and biosimilars market in terms of revenue, supported by high healthcare spending and broad adoption of advanced therapies. Structured biosimilar approval and reimbursement pathways also contribute to regional leadership.
Question 4: What role do biosimilars play in the biologics and biosimilars market?
Answer: Biosimilars help expand access and reduce treatment costs within the biologics and biosimilars market by offering similar efficacy and safety at lower prices. They also encourage competition and create budget space for next‑generation biologics.
Question 5: How is AI influencing the biologics and biosimilars market?
Answer: AI supports faster discovery, smarter trial design, and more efficient manufacturing across the biologics and biosimilars market. It also enhances pharmacovigilance and real‑world outcome tracking, giving payers and regulators better evidence for decision‑making.
