Biological Indicators Market Overview
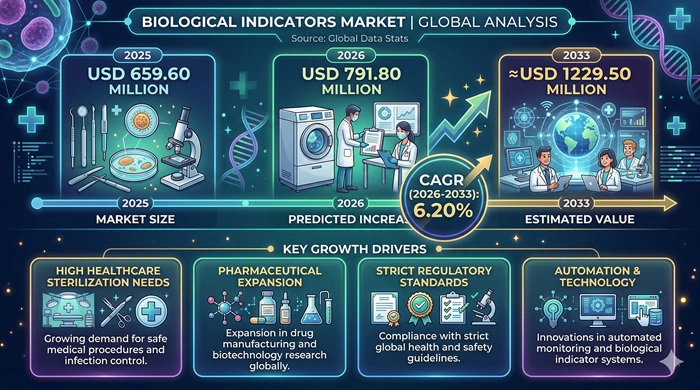
The global Biological Indicators market size is valued at USD 659.60 million in 2025 and is predicted to increase from USD 791.80 million in 2026 to approximately USD 1229.50 million by 2033, growing at a CAGR of 6.20% from 2026 to 2033.
Biological indicators are the most scientifically validated and internationally recognized tools used to verify the effectiveness of sterilization processes across healthcare, pharmaceutical manufacturing, medical device production, and food processing industries — working as living microbial challenge systems that confirm directly whether a sterilization cycle has successfully achieved the required level of microbial kill. These indicators typically contain known populations of highly resistant bacterial spores — most commonly Geobacillus stearothermophilus for steam and hydrogen peroxide sterilization, and Bacillus atrophaeus for ethylene oxide and dry heat sterilization — that must be fully inactivated for a sterilization process to be validated as effective. The Biological Indicators market is growing steadily, driven by rising healthcare-associated infection prevention mandates, expanding pharmaceutical and medical device manufacturing, increasingly strict regulatory sterilization validation requirements, and the growing adoption of self-contained rapid-readout biological indicator systems that significantly reduce sterilization cycle release time in clinical and industrial settings.
AI Impact on the Biological Indicators Industry
Artificial Intelligence Is Transforming Sterilization Process Monitoring, Rapid Biological Indicator Result Interpretation, Predictive Cycle Failure Detection, and Integrated Infection Control Quality Assurance in Ways That Are Meaningfully Improving Patient Safety, Operational Efficiency, and Regulatory Compliance Across the Global Sterilization Monitoring Ecosystem*
Artificial intelligence is delivering commercially meaningful contributions to the Biological Indicators market through the integration of machine learning algorithms into digital rapid-readout biological indicator reader systems — where AI-powered fluorescence detection and optical signal interpretation platforms analyze biological indicator incubation data in real time to identify sterilization failure signals earlier and with greater sensitivity than conventional threshold-based detection systems. These AI-enhanced reader platforms from leading suppliers are capable of learning from historical incubation data patterns to distinguish genuine sterilization failure signals from instrument noise, environmental variation, and biological indicator lot-to-lot performance differences — progressively reducing both false-positive and false-negative result rates that carry costly and clinically serious consequences in healthcare and pharmaceutical sterilization operations. The integration of AI interpretation intelligence into biological indicator readers is also enabling automated sterilization cycle release workflows — where validated AI-powered reader platforms connect directly to sterilization cycle management software to generate compliant documentation and accelerate sterile load release without manual result interpretation delays.
AI is simultaneously advancing the biological indicators ecosystem through predictive analytics applications integrated into hospital and pharmaceutical central sterile supply department management platforms — where machine learning models analyze combinations of biological indicator result histories, sterilizer maintenance records, cycle parameter logs, and equipment sensor data to identify emerging sterilization process risks before they produce failed indicator results or process deviations. By enabling proactive quality management — shifting from reactive investigation of individual failures to predictive identification of systemic process risk factors — these AI-powered platforms are delivering meaningful improvements in sterile supply reliability, regulatory inspection readiness, and overall infection prevention program quality that healthcare organizations and pharmaceutical manufacturers are increasingly valuing. The growing integration of biological indicator monitoring data into AI-powered healthcare quality management platforms is progressively elevating the strategic role of biological indicator programs from basic regulatory compliance activities toward core components of comprehensive, data-driven sterile processing quality systems.
Growth Factors
Rising Healthcare-Associated Infection Prevention Priorities, Expanding Pharmaceutical and Medical Device Manufacturing Sterilization Compliance, Stringent Regulatory Validation Requirements, and Self-Contained Rapid-Readout System Adoption Are the Core Growth Engines of the Biological Indicators Market*
The most foundational and commercially durable growth driver of the Biological Indicators market is the globally rising priority of healthcare-associated infection (HAI) prevention — where regulatory authorities, hospital accreditation bodies, infection control programs, and patient safety organizations are collectively demanding increasingly rigorous sterilization process validation documentation and biological indicator monitoring compliance. Healthcare-associated infections affect hundreds of millions of patients globally each year — generating significant mortality, prolonged hospitalization, and substantial healthcare costs — with inadequate sterilization of surgical instruments, medical devices, and pharmaceutical products being one of the most preventable contributing factors that biological indicator monitoring programs directly address. The combination of growing HAI regulatory scrutiny, expanding hospital accreditation requirements mandating biological indicator monitoring, and rising institutional accountability for sterilization process compliance is creating strong and structurally growing demand for biological indicators across hospital central sterile supply departments, operating room sterile processing programs, and outpatient surgical center sterilization operations globally.
The extraordinary growth of the global pharmaceutical and biopharmaceutical manufacturing industry — driven by the expanding biologics pipeline, growing biosimilar production, new small-molecule drug manufacturing investments in emerging markets, and rising generic drug output requiring validated sterilization processes for parenteral and sterile pharmaceutical products — is generating substantial incremental biological indicator demand. FDA, EMA, and PMDA pharmaceutical manufacturing regulations require comprehensive sterilization validation documentation — including biological indicator challenge testing that demonstrates the specified sterility assurance level for each sterilization process — with inspection deficiencies related to inadequate sterilization validation among the most consequential and frequently cited pharmaceutical manufacturing compliance findings. This regulatory sterilization validation imperative creates recurring, non-discretionary biological indicator procurement demand from pharmaceutical manufacturers that sustains predictable and commercially resilient market revenue growth regardless of broader pharmaceutical industry capital spending fluctuations.

Market Outlook
With Self-Contained Rapid-Readout Systems Achieving Healthcare Mainstream Adoption, Pharmaceutical Sterilization Validation Requirements Expanding, Emerging Market Healthcare Infrastructure Growing Rapidly, and Next-Generation Rapid Platforms Entering Commercialization, the Biological Indicators Market Outlook Remains Robustly Positive Through 2033*
The long-term outlook for the Biological Indicators market is firmly positive, supported by a compelling combination of regulatory, clinical, technological, and demographic growth catalysts expected to sustain consistent revenue expansion throughout the 2026–2033 forecast period. The most commercially transformative technology trend accelerating adoption within the biological indicator product landscape is the progressive displacement of conventional 24–48 hour incubation biological indicators by self-contained rapid-readout systems — including fluorescence-based platforms that deliver verified sterilization efficacy results within one to three hours — that dramatically reduce sterilization cycle release time, improve sterile supply chain efficiency, and enable same-day instrument reprocessing workflows in high-volume surgical and endoscopy programs. As self-contained rapid-readout biological indicator systems from leading manufacturers continue to improve result speed and reduce reader system costs, the adoption trajectory of rapid biological indicators is expected to accelerate progressively throughout the forecast period — shifting the product mix toward higher-value rapid systems and improving overall market revenue quality.
Emerging market healthcare infrastructure development represents one of the most commercially significant long-term growth opportunities in the Biological Indicators market — with rapidly growing hospital networks in China, India, Southeast Asia, Latin America, and Africa investing in central sterile supply department infrastructure, sterilization equipment, and infection control program development that creates large cohorts of first-time biological indicator program adopters. Government healthcare quality improvement programs, hospital accreditation initiatives, and international health organization infection prevention support programs across these regions are systematically establishing biological indicator monitoring as a standard sterilization compliance requirement — generating durable new market revenue from geographies that are currently underrepresented in global biological indicator sales but carry large and rapidly growing unmet demand for reliable sterilization verification tools. This emerging market adoption wave is expected to contribute meaningfully to accelerating global market growth during the latter years of the 2026–2033 forecast period.
Expert Speaks
-
Russ Hochman, CEO, Mesa Labs — "The biological indicator market is experiencing meaningful commercial momentum driven by the healthcare industry's growing investment in patient safety, the accelerating adoption of rapid-readout sterilization monitoring systems, and the expanding regulatory requirements for sterilization process validation documentation in both healthcare and pharmaceutical manufacturing settings. Mesa Labs is investing in next-generation rapid biological indicator platform development — including faster fluorescence-based systems and integrated digital monitoring solutions — to serve the growing demand for more efficient, reliable, and connected sterilization monitoring programs across hospital and pharmaceutical manufacturing customers globally."
-
J.H. Mikael Bratt, CEO, Getinge AB — "Sterilization process quality and verification — including biological indicator monitoring programs — sits at the core of patient safety and infection prevention in every clinical and pharmaceutical manufacturing environment we serve, and we see growing investment from healthcare and life science customers in more comprehensive and technology-enabled sterilization monitoring solutions that deliver faster results, better documentation, and stronger regulatory compliance. The integration of rapid biological indicator systems with digital sterile processing management platforms is one of the most important innovation areas we are pursuing to help our customers achieve the highest levels of sterilization process reliability."
-
Dan Carestio, CEO, Steris plc — "Biological indicators and sterilization process monitoring represent a critical and non-discretionary component of every sterile processing program in healthcare and pharmaceutical manufacturing — and the growing regulatory scrutiny of sterilization validation documentation, combined with the healthcare system's intensifying focus on HAI prevention, is creating strong and sustained demand for advanced biological indicator monitoring solutions. Steris is committed to continuous innovation in biological indicator product performance, reader technology, and digital integration that enables our customers to operate the most reliable and compliant sterilization monitoring programs possible."
Key Report Takeaways
-
North America dominates the Biological Indicators market, holding approximately 41.75% of global market share in 2025, driven by the highest healthcare infrastructure density, the most stringent FDA and CDC sterilization process regulatory requirements, the highest hospital accreditation compliance standards mandating biological indicator monitoring programs, the strongest pharmaceutical and medical device manufacturing sterilization validation compliance infrastructure, and the concentration of leading global biological indicator manufacturers including Solventum, Mesa Labs, Steris, and Cantel Medical with dominant North American commercial positions.
-
Asia Pacific is the fastest-growing regional market for biological indicators, projected to expand at a CAGR of approximately 8.60% from 2026 to 2033, driven by China and India's rapidly expanding hospital infrastructure and pharmaceutical manufacturing capacity, progressive adoption of international infection control standards and hospital accreditation requirements, growing medical device manufacturing sterilization validation compliance investment, and government healthcare quality improvement programs across Southeast Asia establishing biological indicator monitoring as a standard sterile processing requirement.
-
Self-contained biological indicators are the dominant product type, accounting for approximately 54.20% of total market revenue in 2025, driven by the strong clinical adoption of rapid-readout self-contained systems — particularly fluorescence-based platforms delivering results within one to three hours — that dramatically improve sterilization cycle release efficiency compared to conventional 24–48 hour incubation systems, with healthcare organizations progressively standardizing on self-contained rapid systems as the preferred biological indicator format.
-
Steam sterilization is the dominant sterilization method segment, contributing approximately 46.80% of total market revenue in 2025, reflecting autoclaving's status as the most widely used sterilization method in hospital central sterile supply departments, surgical instrument reprocessing programs, and pharmaceutical terminal sterilization operations — generating the largest volume of biological indicator use globally across both self-contained rapid and conventional spore strip formats.
-
Hospitals and clinics represent the largest end-user segment, contributing approximately 44.60% of total market revenue in 2025, driven by mandatory biological indicator monitoring requirements of hospital accreditation standards that collectively require routine biological indicator testing of all steam, ethylene oxide, and low-temperature sterilization cycles used in surgical instrument reprocessing and medical device sterilization operations.
-
Hydrogen peroxide sterilization is the fastest-growing sterilization method segment, expected to expand at a CAGR of approximately 9.80% through 2033, driven by the progressive adoption of hydrogen peroxide vapor and plasma sterilization systems for heat-sensitive and moisture-sensitive surgical instruments, flexible endoscopes, and electronic medical devices that cannot withstand steam sterilization temperatures — generating growing demand for Geobacillus stearothermophilus-based biological indicators specifically validated for hydrogen peroxide sterilization cycle monitoring.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 1229.50 Million |
| Market Size by 2025 | USD 659.60 Million |
| Market Size by 2026 | USD 791.80 Million |
| Market Growth Rate (2026–2033) | CAGR of 6.20% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Segments Covered | Product Type, Sterilization Method, End User |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Rising HAI Prevention Priorities, Pharmaceutical Sterilization Validation Mandates, Rapid-Readout Adoption, and Emerging Market Healthcare Infrastructure Growth Are the Four Pillars Powering Biological Indicators Market Growth*
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising healthcare-associated infection prevention priorities and hospital accreditation requirements | ~35% | Global | Short to Long-term |
| Expanding pharmaceutical and medical device manufacturing sterilization validation compliance | ~27% | North America, Europe, Asia Pacific | Short to Long-term |
| Self-contained rapid-readout biological indicator adoption displacing conventional systems | ~25% | North America, Europe | Short to Medium-term |
| Emerging market healthcare infrastructure and sterile processing program development | ~13% | Asia Pacific, Latin America, MEA | Medium to Long-term |
The most enduring structural driver of the Biological Indicators market is the healthcare system's non-negotiable commitment to sterilization process efficacy verification — where hospital accreditation bodies including The Joint Commission, DNV GL Healthcare, and equivalent national accreditation organizations worldwide mandate documented biological indicator monitoring programs as a fundamental patient safety and infection control requirement. Every hospital and ambulatory surgical center operating sterilization equipment must demonstrate and document biological indicator monitoring compliance during accreditation surveys and ongoing quality assurance audits — creating a large, stable, and recurring biological indicator procurement demand base that is structurally immune to the discretionary spending pressures affecting most medical products. The progressive intensification of healthcare accreditation standards — with recent framework revisions in multiple major markets expanding biological indicator monitoring frequency requirements, increasing documentation standards, and introducing new requirements for reader system validation — is systematically increasing per-facility biological indicator consumption volume.
Pharmaceutical and biopharmaceutical manufacturing sterilization validation requirements under FDA 21 CFR and EU GMP frameworks represent an equally important and commercially resilient growth driver — where mandatory biological indicator challenge testing required for initial sterilization process validation, periodic revalidation, and post-maintenance performance qualification generates substantial and predictable biological indicator procurement volumes from pharmaceutical manufacturers. The extraordinary growth of the global biopharmaceutical manufacturing industry — including monoclonal antibody production, vaccine manufacturing, gene therapy, and cell therapy — is generating new categories of pharmaceutical sterilization validation biological indicator demand as manufacturers validate novel sterilization processes for complex, heat-sensitive biological drug substances. FDA inspection findings related to inadequate sterilization validation documentation represent some of the most commercially consequential warning letter citations for pharmaceutical manufacturers — creating strong and sustained compliance investment motivation that ensures non-discretionary biological indicator procurement regardless of pharmaceutical company cost reduction initiatives.

Restraints Impact Analysis
High Cost of Rapid-Readout Reader Systems, Stringent Storage Requirements, Concentrated Supplier Base, and Long Regulatory Validation Timelines Restrain Faster Biological Indicators Market Expansion*
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High capital cost of rapid-readout reader systems limiting adoption in budget-constrained settings | ~34% | Asia Pacific, Latin America, MEA | Short to Long-term |
| Stringent cold-chain storage and handling requirements increasing operational complexity | ~28% | Global — especially emerging markets | Short to Long-term |
| Limited supplier base creating dependency and concentrated pricing leverage | ~26% | Global | Short to Long-term |
| Long regulatory validation timelines for new biological indicator products slowing innovation | ~12% | Global | Short to Medium-term |
The most broadly experienced commercial barrier constraining faster adoption of advanced biological indicators — particularly self-contained rapid-readout systems — is the significant capital investment required for reader system infrastructure that accompanies the transition from conventional incubation-based biological indicators to fluorescence-based rapid systems. Hospital central sterile supply departments and pharmaceutical manufacturing facilities seeking to transition to rapid biological indicator programs must invest in validated reader systems that create meaningful capital investment barriers for budget-constrained healthcare organizations in emerging markets and smaller hospitals in developed markets operating under tight operating expense constraints. The combination of reader capital cost, ongoing calibration and maintenance requirements, and staff training investment required for reliable rapid biological indicator program operation creates a total cost of adoption barrier that limits penetration of rapid-readout systems in market segments where operational efficiency benefits do not clearly justify the investment.
The stringent cold-chain storage and handling requirements of biological indicators — which contain living bacterial spores that must be maintained under controlled temperature conditions throughout the supply chain and storage period to ensure specified resistance performance at time of use — create operational complexity and supply chain cost challenges that are particularly significant for healthcare facilities and pharmaceutical manufacturers in emerging markets with less reliable cold-chain infrastructure. Biological indicator lot-to-lot performance variability — despite rigorous manufacturer quality control testing — creates ongoing quality assurance challenges for facilities conducting sterilization validation programs that require demonstrated performance equivalence across different biological indicator lot numbers, adding testing burden and cost to pharmaceutical validation programs. The highly concentrated supplier landscape in the biological indicator market — where a small number of large manufacturers command the majority of global revenue — creates customer dependency concerns and limits the competitive pricing dynamics that broader supplier competition would generate.
Opportunities Impact Analysis
Next-Generation Rapid Platform Innovation, Digital Connected Sterilization Monitoring Integration, Emerging Market Healthcare Program Establishment, and Pharmaceutical Biomanufacturing Expansion Create Major Biological Indicators Market Opportunities*
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Next-generation rapid biological indicator systems with faster readout and improved digital connectivity | ~34% | North America, Europe | Short to Medium-term |
| Digital integrated sterilization monitoring platform adoption connecting biological indicator data | ~27% | North America, Europe, Japan | Short to Medium-term |
| Emerging market first-time biological indicator program establishment | ~27% | Asia Pacific, Latin America, MEA | Medium to Long-term |
| Pharmaceutical biomanufacturing and cell therapy sterilization validation new application growth | ~12% | North America, Europe | Medium to Long-term |
The most immediately commercially compelling opportunity in the Biological Indicators market is the development and commercialization of next-generation rapid biological indicator platforms — including systems targeting sub-30-minute sterilization efficacy results for steam sterilization and novel molecular detection approaches that eliminate incubation entirely by detecting enzymatic or genetic markers of spore kill immediately following sterilization cycle completion. These ultra-rapid platforms — currently in active commercial development by leading suppliers — would represent a transformative advancement in sterilization workflow efficiency, enabling fully validated same-cycle biological indicator clearance that eliminates the remaining operational inefficiency of even current-generation rapid fluorescence systems and dramatically improves surgical instrument reprocessing and sterile supply throughput in high-volume clinical settings. Companies that successfully commercialize validated ultra-rapid biological indicator platforms with robust clinical performance data and regulatory clearance will capture substantial market share from existing rapid and conventional system users — making this the highest-value product innovation opportunity within the biological indicator market during the forecast period.
The progressive integration of biological indicator monitoring into comprehensive digital sterile processing management platforms — where biological indicator results, chemical indicator data, sterilizer cycle parameters, and instrument tracking information are unified in connected quality management software generating automated compliance documentation — is simultaneously reshaping the competitive dynamics of the biological indicator market by elevating the importance of digital connectivity and software ecosystem compatibility in product selection decisions. Healthcare systems are progressively evaluating biological indicator suppliers not merely on spore-based performance specifications but on the quality, connectivity, and analytical capability of their associated reader systems and software integration offerings — creating important technology differentiation advantages for suppliers that invest in comprehensive connected sterile processing ecosystem development. This digital integration trend is progressively expanding the revenue opportunity in biological indicator monitoring from product sales alone toward recurring software subscription and data services revenue — meaningfully improving the commercial model quality of leading digital biological indicator ecosystem providers.

Segment Analysis
By Product Type
Self-Contained Biological Indicators Lead Market Revenue While Driving the Fastest Product Innovation and Clinical Adoption Growth Across the Biological Indicators Market*
Self-contained biological indicators hold the dominant position in the Biological Indicators market, accounting for approximately 54.20% of total global market revenue in 2025, driven by the strong and accelerating clinical adoption of rapid-readout self-contained systems — particularly fluorescence-based platforms — that deliver sterilization efficacy verification within one to three hours compared to the 24–48 hour incubation period required by conventional spore strip biological indicators. The self-contained format integrates all components required for biological indicator incubation and result reading within a single device — eliminating separate incubation equipment, growth media inoculation steps, and subjective visual result interpretation required by conventional formats — delivering operational simplicity, reduced contamination risk, and standardized result documentation that are highly valued by busy central sterile supply department staff. North America leads self-contained biological indicator revenue globally — driven by the highest rapid-readout system adoption rates supported by healthcare operational efficiency priorities and the strongest concentration of facilities with established rapid biological indicator programs — while Europe is the second-largest market. Leading North American suppliers including Solventum, Steris, and Mesa Labs maintain dominant positions through comprehensive product portfolios, validated reader system ecosystems, and deep hospital sterile processing program customer relationships built over decades of commercial engagement.
Conventional biological indicators — comprising spore strips, spore suspensions, and ampoules used with separate incubation equipment — account for approximately 45.80% of the Biological Indicators market revenue in 2025 and remain commercially relevant in cost-sensitive healthcare markets, food sterilization applications, pharmaceutical validation programs, and research settings where the lower per-unit product cost and flexibility of conventional formats justifies their use despite slower result generation. Conventional spore strips for steam and ethylene oxide sterilization monitoring remain the dominant conventional product format — used extensively in emerging market hospital sterilization programs, veterinary sterilization, dental office sterilization, and food industry sterilization validation where rapid-readout reader systems have not yet been widely adopted. Asia Pacific is the primary and fastest-growing conventional biological indicator market — driven by the large volume of budget-constrained healthcare facilities across India, China, Southeast Asia, and other developing markets that rely on conventional indicators for cost-effective sterilization monitoring program compliance. Key suppliers including Getinge, Cantel Medical, and a range of regional Asian manufacturers serve conventional biological indicator demand across Asia Pacific markets at competitive price points suited to emerging market healthcare budgets.
By Sterilization Method
Steam Sterilization Leads the Biological Indicators Market While Hydrogen Peroxide Sterilization Registers the Fastest Adoption Growth Across All Sterilization Method Segments*
Steam sterilization represents the dominant sterilization method segment in the Biological Indicators market, accounting for approximately 46.80% of total global market revenue in 2025, reflecting autoclaving's status as the most widely used, most cost-efficient, and most broadly applicable sterilization method in hospital central sterile supply departments, surgical instrument reprocessing programs, pharmaceutical terminal sterilization, and food processing sterilization — generating the largest volume of biological indicator consumption across both routine cycle monitoring and periodic process validation applications. Geobacillus stearothermophilus spore-based biological indicators used to verify steam sterilization cycle efficacy represent the single highest-volume biological indicator product category globally, with every hospital sterilizer requiring periodic biological indicator testing under accreditation standards that generate consistent and recurring procurement demand. North America and Europe are the largest steam sterilization biological indicator markets — driven by the highest hospital infrastructure density and most comprehensive steam sterilizer biological indicator monitoring regulatory requirements — while Asia Pacific is the fastest-growing steam sterilization biological indicator region at approximately 8.60% CAGR through 2033. Solventum, Steris, and Mesa Labs are the dominant steam sterilization biological indicator suppliers in North America and Europe, maintaining strong market positions through comprehensive product portfolios and validated performance documentation meeting accreditation and regulatory requirements.
Hydrogen peroxide sterilization is the fastest-growing sterilization method segment in the Biological Indicators market, projected to expand at a CAGR of approximately 9.80% from 2026 to 2033, driven by the accelerating adoption of hydrogen peroxide vapor and plasma low-temperature sterilization systems for reprocessing heat-sensitive and moisture-sensitive instruments that cannot tolerate steam autoclaving. The growing clinical use of flexible endoscopes, robotic surgical instruments, electronic medical devices, and implantable electronic components that require validated low-temperature sterilization is generating rapidly expanding demand for hydrogen peroxide sterilization biological indicators validated for Steris V-PRO, Stryker Sterrad, and equivalent platform performance verification. North America leads hydrogen peroxide sterilization biological indicator adoption — driven by the highest concentration of advanced surgical programs using heat-sensitive robotic and electronic instruments — while Asia Pacific is the fastest-growing region as leading hospitals in China, India, and Southeast Asia progressively adopt hydrogen peroxide sterilization systems for their growing robotic surgery and advanced endoscopy programs. Solventum and Steris dominate the hydrogen peroxide sterilization biological indicator segment globally through products specifically validated for their respective low-temperature sterilization system platforms.

Regional Insights
North America
North America Commands the Global Biological Indicators Market With the Highest Hospital Sterilization Compliance Requirements, Leading Pharmaceutical Sterilization Validation Infrastructure, and the World's Strongest Concentration of Biological Indicator Technology Innovation and Manufacturing*
North America holds the dominant position in the global Biological Indicators market, accounting for approximately 41.75% of total global market revenue in 2025, and is projected to sustain steady market leadership at a CAGR of approximately 5.80% from 2026 to 2033. The United States drives the overwhelming majority of North American revenue — where the combination of The Joint Commission and CMS hospital accreditation biological indicator monitoring mandates, FDA pharmaceutical manufacturing sterilization validation requirements, and the highest per-capita concentration of hospitals, ambulatory surgical centers, and pharmaceutical manufacturing facilities collectively create the world's most commercially developed and compliance-intensive biological indicator demand environment. Leading companies commanding the North American Biological Indicators market include Solventum (formerly 3M Health Care), Steris plc, Mesa Labs, Cantel Medical, and Getinge — each maintaining dominant positions across their respective product and sterilization method segments through decades of customer relationship development and comprehensive product portfolio coverage. Canada contributes meaningfully to regional market revenue through its well-developed hospital accreditation requirements and established pharmaceutical manufacturing sterilization compliance infrastructure across Ontario, Quebec, and Alberta manufacturing centers.
The North American biological indicator market is characterized by the highest rapid-readout self-contained system adoption rates globally — with the majority of large hospital systems and pharmaceutical manufacturers now operating primarily on rapid fluorescence biological indicator platforms that deliver one to three hour results — and the most active commercial pipeline of next-generation ultra-rapid and digitally connected biological indicator product development. The market is progressively evolving toward integrated digital sterile processing management ecosystems — where biological indicator monitoring data connects with sterilizer cycle management, instrument tracking, and compliance reporting software in unified quality platforms — creating growing demand for connected biological indicator reader systems that command premium pricing and support stronger customer retention. North America's combination of regulatory stringency, technology leadership, and high procedure volumes positions it to maintain market leadership throughout the 2026–2033 forecast period.
Asia Pacific
Asia Pacific Is the Fastest-Growing Biological Indicators Market, Driven by China and India's Rapidly Expanding Hospital Infrastructure, Growing Pharmaceutical Manufacturing Compliance, and Progressive Adoption of International Infection Control Standards*
Asia Pacific is the fastest-growing regional segment in the Biological Indicators market, projected to expand at a CAGR of approximately 8.60% from 2026 to 2033 — the highest of any region globally. China is the most commercially significant and rapidly growing Asia Pacific biological indicator market — where the government's massive hospital infrastructure expansion program, the progressive implementation of hospital accreditation requirements mandating sterilization monitoring compliance, and the extraordinary growth of pharmaceutical and medical device manufacturing requiring validated sterilization processes are collectively generating the region's largest and most rapidly growing biological indicator procurement demand. India represents the second-largest and equally dynamic Asia Pacific biological indicator market — driven by the country's extraordinary hospital network expansion, rapidly growing generic pharmaceutical manufacturing sterilization validation requirements, and the progressive adoption of international infection control standards across public and private healthcare systems. Key companies serving Asia Pacific Biological Indicators market growth include Solventum, Steris, Getinge, and Mesa Labs alongside regional Asian biological indicator manufacturers serving domestic healthcare demand at cost-competitive price points that match emerging market healthcare budgets.
Japan and South Korea represent the most technically sophisticated and commercially mature Asia Pacific biological indicator markets — with well-established pharmaceutical and medical device manufacturing sterilization validation compliance cultures, strong hospital accreditation requirements, and high rapid-readout biological indicator system adoption rates comparable to North American standards. Southeast Asian markets — including Singapore, Thailand, Malaysia, and Vietnam — are experiencing accelerating biological indicator adoption as hospital accreditation programs progressively expand coverage and government healthcare quality improvement initiatives establish sterilization monitoring compliance as a standard institutional requirement. The demographic scale, rapid healthcare infrastructure growth, and expanding pharmaceutical manufacturing compliance investment across Asia Pacific collectively position the region as the most commercially dynamic and fastest-growing biological indicator market globally through 2033.
Top Key Players
-
Solventum Corporation — formerly 3M Health Care (United States)
-
Steris plc (United States / Ireland)
-
Mesa Labs Inc. (United States)
-
Getinge AB (Sweden)
-
Cantel Medical LLC — Medivators (United States)
-
Crosstex International Inc. (United States)
-
Terragene SA (Argentina)
-
Sychem Ltd. (United Kingdom)
-
Hu-Friedy Mfg. Co. LLC (United States)
-
Ritter Medical (Germany)
-
Propper Manufacturing Co. Inc. (United States)
-
GKE GmbH (Germany)
Recent Developments
-
Solventum Corporation (2025) — Following its spin-off from 3M as an independent healthcare technology company in 2024, Solventum announced continued investment in its Attest biological indicator product portfolio — including next-generation rapid-readout Attest Super Rapid Readout Biological Indicator systems with enhanced fluorescence detection technology delivering faster result confirmation for steam sterilization monitoring — while advancing its integrated digital sterile processing management platform connecting Attest biological indicator reader data with comprehensive sterilization cycle documentation and compliance reporting workflows for hospital central sterile supply departments.
-
Steris plc (2025) — Announced expansion of its biological indicator portfolio with new VERICEL hydrogen peroxide biological indicators specifically validated for its V-PRO Max sterilization system — addressing growing clinical demand for validated low-temperature sterilization monitoring solutions for heat-sensitive surgical instruments and electronic medical devices — while concurrently advancing digital integration capabilities through enhanced connectivity between its biological indicator reader systems and hospital sterile processing management software platforms.
-
Mesa Labs (2024) — Completed integration of its Bios International biological indicator product line following previous acquisition — consolidating manufacturing operations and expanding product portfolio breadth across steam, ethylene oxide, hydrogen peroxide, and dry heat sterilization biological indicator categories — while reporting strong commercial revenue growth driven by pharmaceutical sterilization validation biological indicator demand growth and progressive hospital adoption of its rapid-readout biological indicator reader systems in North American and European markets.
-
Getinge AB (2024) — Announced strategic investments in its sterilization monitoring product portfolio including biological indicators compatible with its AMSCO steam sterilization systems — and launched enhanced digital connectivity features for its biological indicator incubation and reading equipment integrating with Getinge's Torin sterile processing management software platform — supporting hospital customers in achieving comprehensive digital sterile processing documentation and compliance reporting workflows meeting evolving Joint Commission and CMS regulatory documentation standards.
-
Terragene SA (2025) — Announced significant commercial expansion into Asian and Middle Eastern healthcare markets — establishing regional distribution partnerships in India, Southeast Asia, and the Gulf Cooperation Council countries — supported by international regulatory clearance of its BIONOVA rapid-readout self-contained biological indicator systems for steam, ethylene oxide, and hydrogen peroxide sterilization monitoring, positioning Terragene as a competitive cost-effective alternative to North American and European branded biological indicator suppliers in rapidly growing emerging healthcare markets.
Market Trends
The Accelerating Transition to Ultra-Rapid Biological Indicator Platforms and the Progressive Integration of Connected Digital Sterile Processing Monitoring Systems Are the Two Most Commercially Significant Trends Reshaping the Biological Indicators Market Through 2033*
The most commercially transformative trend in the Biological Indicators market is the progressive displacement of conventional 24–48 hour incubation biological indicators by self-contained rapid-readout fluorescence systems — and the active commercial development of next-generation ultra-rapid platforms targeting sub-30-minute sterilization efficacy verification. Healthcare operational efficiency pressures — including growing surgical procedure volumes, staff shortage challenges in central sterile supply departments, and demand for same-day instrument reprocessing in high-turnover surgical and endoscopy programs — are creating strong institutional motivation for biological indicator result time reduction that is driving both accelerating adoption of current-generation rapid systems and strong commercial interest in even faster next-generation platforms from leading biological indicator manufacturers. As result time improvements progressively eliminate the remaining operational barriers to complete biological indicator-based sterilization release workflows, the clinical and operational value proposition of rapid biological indicators is strengthening — accelerating the product mix shift toward premium rapid systems throughout the biological indicator product landscape.
The progressive integration of biological indicator monitoring into comprehensive digital sterile processing management platforms — where biological indicator results, chemical indicator data, sterilizer cycle parameters, and instrument tracking information are unified in connected quality management software — is simultaneously reshaping the competitive dynamics of the biological indicator market by elevating the importance of digital connectivity and software ecosystem compatibility in biological indicator product selection decisions. Healthcare systems are progressively evaluating biological indicator suppliers not merely on spore-based performance specifications but on the quality, connectivity, and analytical capability of their associated reader systems and software integration offerings — creating important technology differentiation advantages for suppliers that invest in comprehensive connected sterile processing ecosystem development. This digital integration trend is progressively expanding the revenue opportunity in biological indicator monitoring from product sales alone toward recurring software subscription and data services revenue — meaningfully improving the commercial model quality of leading digital biological indicator ecosystem providers.
Segments Covered in the Report
By Product Type:
-
Self-Contained Biological Indicators
-
Conventional Biological Indicators
-
Spore Strips
-
Spore Suspensions
-
Ampoules
By Sterilization Method:
-
Steam Sterilization
-
Ethylene Oxide Sterilization
-
Dry Heat Sterilization
-
Radiation Sterilization
-
Hydrogen Peroxide Sterilization
-
Others
By End User:
-
Hospitals and Clinics
-
Pharmaceutical and Biotechnology Companies
-
Medical Device Manufacturers
-
Food and Beverage Industry
-
Research and Academic Institutes
-
Others
By Region:
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the Biological Indicators market and what is its projected value by 2033?
Answer: The Biological Indicators market was valued at USD 659.60 million in 2025 and is projected to reach USD 1229.50 million by 2033, growing at a CAGR of 6.20% from 2026 to 2033. This growth is driven by rising HAI prevention priorities, expanding pharmaceutical sterilization validation requirements, accelerating self-contained rapid-readout system adoption, and emerging market healthcare infrastructure investment.
Question 2: What are biological indicators used for in sterilization monitoring?
Answer: Biological indicators are used to directly verify the efficacy of sterilization processes — containing known populations of highly resistant bacterial spores that must be fully inactivated for a sterilization cycle to be validated as effective across steam, ethylene oxide, hydrogen peroxide, and dry heat sterilization methods. The Biological Indicators market serves healthcare facilities, pharmaceutical manufacturers, medical device companies, and food processors that require documented sterilization efficacy verification for regulatory compliance, accreditation, and patient safety.
Question 3: Which region leads the Biological Indicators market and which is growing fastest?
Answer: North America leads the Biological Indicators market with approximately 41.75% of global revenue in 2025 — driven by the highest hospital sterilization accreditation compliance requirements, leading pharmaceutical sterilization validation infrastructure, and the strongest concentration of biological indicator technology manufacturers. Asia Pacific is the fastest-growing region at approximately 8.60% CAGR through 2033 — driven by China and India's expanding hospital networks, growing pharmaceutical manufacturing sterilization compliance investment, and progressive adoption of international infection control standards.
Question 4: What is the difference between self-contained and conventional biological indicators?
Answer: Self-contained biological indicators integrate all components for incubation and result reading within a single device — delivering results in one to three hours with minimal handling — representing the dominant and fastest-growing product type in the Biological Indicators market at approximately 54.20% of revenue. Conventional biological indicators including spore strips and suspensions require separate incubation equipment and 24–48 hour result periods — remaining cost-effective options for budget-constrained settings and applications where rapid result turnaround is not operationally critical.
Question 5: How are regulatory requirements driving growth in the Biological Indicators market?
Answer: FDA, EMA, Joint Commission, and equivalent regulatory and accreditation frameworks mandate documented biological indicator monitoring for sterilization process validation and routine cycle verification in healthcare and pharmaceutical manufacturing settings — creating non-discretionary procurement demand that sustains consistent Biological Indicators market revenue regardless of broader healthcare spending pressures. Progressive international expansion of sterilization validation and hospital accreditation requirements — particularly across Asia Pacific and Latin American markets — is systematically expanding the biological indicator program compliance requirement to new healthcare and manufacturing facilities that represent growing incremental market demand throughout the 2026–2033 forecast period.
