Automated Endoscope Reprocessors Market Overview
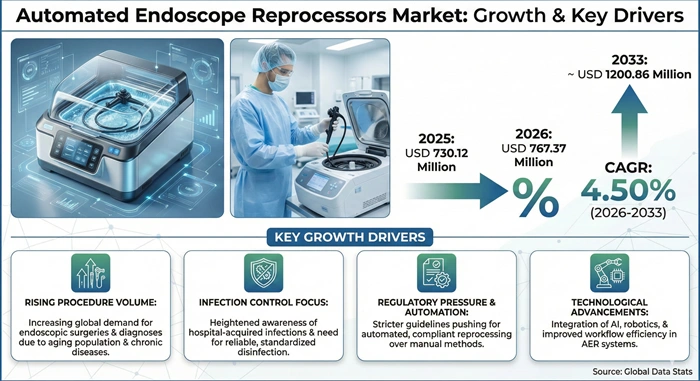
The global automated endoscope reprocessors market size is valued at USD 730.12 million in 2025 and is predicted to increase from USD 767.37 million in 2026 to approximately USD 1200.86 million by 2033, growing at a CAGR of 4.50% from 2026 to 2033.
The automated endoscope reprocessors market focuses on advanced medical devices that clean, disinfect, and reprocess endoscopes using standardized automated cycles to ensure infection control and patient safety in healthcare facilities. These systems replace manual cleaning processes with programmable workflows that include leak testing, cleaning, rinsing, disinfection, and drying cycles. The market encompasses various reprocessor types including flexible endoscope reprocessors, rigid endoscope reprocessors, and integrated systems that combine multiple functions. Growing concerns about hospital-acquired infections, increasing endoscopic procedures, and stringent regulatory requirements drive adoption of automated endoscope reprocessors across hospitals, ambulatory surgical centers, and diagnostic clinics worldwide.
The transformation toward automated endoscope reprocessing represents a fundamental shift in how healthcare facilities manage infection control. Modern reprocessors incorporate digital monitoring systems, data logging capabilities, barcode scanning, and connectivity features enabling comprehensive documentation and audit trails. These technological advancements enhance compliance with infection prevention guidelines while reducing human error in reprocessing workflows. Growing pressure from regulatory bodies, accreditation organizations, and healthcare administrators has accelerated adoption of automated systems across both established healthcare providers and emerging medical facilities.
AI Impact on the Automated Endoscope Reprocessors Industry
Artificial Intelligence Enhances Infection Control and Workflow Optimization
The automated endoscope reprocessors market experiences rapid adoption of artificial intelligence-assisted monitoring technologies that enable sophisticated tracking capabilities, real-time alerts, and predictive maintenance across endoscope reprocessing operations. AI technologies analyze extensive volumes of operational data generated from sensors, cycle logs, and performance metrics to identify process deviations, generate maintenance predictions, and ensure consistent reprocessing quality. Machine learning algorithms optimize reprocessing parameters including cycle times, chemical concentrations, and temperature profiles to maximize efficiency while maintaining infection control standards.
Predictive analytics capabilities enable healthcare facilities to anticipate equipment failures, schedule preventive maintenance, and minimize downtime in endoscope reprocessing operations. AI-enabled digital monitoring systems provide opportunities to conduct virtual simulations of reprocessing workflows, accelerate optimization processes, and continuously fulfill infection control goals without requiring extensive physical testing. The progressive shift toward intelligent automation positions the automated endoscope reprocessors market at the forefront of scalable, efficient operations that align patient safety with operational efficiency across healthcare sectors.
Growth Factors
Rising Endoscopic Procedures and Infection Control Awareness Drive Market Expansion
The automated endoscope reprocessors market experiences substantial growth momentum driven by increasing global demand for endoscopic procedures including gastrointestinal endoscopy, bronchoscopy, cystoscopy, and minimally invasive surgeries. Healthcare facilities worldwide perform millions of endoscopic procedures annually requiring reliable reprocessing solutions to ensure patient safety and prevent cross-contamination. Aging populations, rising chronic disease prevalence, and growing awareness of early disease detection through endoscopic screening programs intensify demand for endoscope reprocessing equipment across hospitals and outpatient facilities.
Stringent infection control regulations imposed by governmental agencies and accreditation organizations compel healthcare providers to implement standardized reprocessing protocols that minimize human error and ensure consistent disinfection quality. Regulatory frameworks established by agencies including the United States Food and Drug Administration, Centers for Disease Control and Prevention, and European regulatory bodies mandate compliance with infection prevention guidelines while encouraging adoption of automated reprocessing systems. Hospitals face mounting pressure to demonstrate infection control compliance through implementation of automated endoscope reprocessors with comprehensive documentation and audit trail capabilities. Technological innovations such as digital monitoring, barcode scanning, and connectivity features optimize reprocessing workflows while substantially reducing infection risks, positioning automated endoscope reprocessors as essential components of modern healthcare infrastructure.

Market Outlook
Industry Transitions Toward Standardized, Data-Driven Reprocessing Practices
The automated endoscope reprocessors market outlook reflects an industry undergoing fundamental transformation as healthcare facilities embrace standardized, data-driven reprocessing methodologies in response to infection control objectives and regulatory requirements. Demand for endoscopic procedures, minimally invasive surgeries, and diagnostic imaging combined with infection control regulations drives robust market expansion across developed and emerging economies. Integrated reprocessing systems, digital monitoring platforms, and advanced disinfection technologies are gaining mainstream acceptance as essential components of modern endoscopy departments. Companies are deploying comprehensive digital ecosystems that integrate reprocessing operations with hospital information systems to support data-driven decision-making and enhance collaboration throughout healthcare operations.
Regional expansion patterns demonstrate significant investment activity across North America, Europe, and Asia Pacific as hospitals, ambulatory surgical centers, and diagnostic clinics prioritize infection control-focused innovations. Government initiatives, healthcare accreditation requirements, and collaborative partnerships between medical device manufacturers and healthcare providers accelerate commercialization of automated endoscope reprocessors. The startup ecosystem contributes innovative solutions including compact reprocessing units, portable systems, and novel disinfection technologies that push industry boundaries. Strategic alliances between established medical device manufacturers and emerging technology providers facilitate rapid deployment of automated endoscope reprocessors at commercial scale. The convergence of regulatory requirements, patient safety expectations, and technological capabilities creates powerful incentives for healthcare facilities worldwide to adopt automated endoscope reprocessors as standard operating procedures rather than optional enhancements.
Expert Speaks
-
James L. Herbert II, CEO of STERIS Corporation, stated that the company remains committed to advancing infection prevention solutions and continues investing significantly in technologies that help healthcare providers reduce infection risks while maintaining operational efficiency across endoscope reprocessing operations worldwide.
-
Shuichi Hiramatsu, President and CEO of Olympus Corporation, emphasized that patient safety represents a core component of the company's mission, highlighting ongoing efforts to develop innovative endoscope reprocessing systems that substantially reduce infection risks and enhance workflow efficiency throughout endoscopic procedures.
-
Mattias Perjos, President and CEO of Getinge AB, noted that implementing standardized reprocessing practices throughout healthcare facilities has become essential for infection control, with the company focusing on advanced automated reprocessors that minimize human error and contribute to lower infection rates.
Key Report Takeaways
-
North America leads the automated endoscope reprocessors market with the largest revenue share of 42.3% in 2025 due to robust healthcare infrastructure, advanced infection control standards, and early adoption of automated reprocessing technologies across hospitals and ambulatory surgical centers
-
Asia Pacific emerges as the fastest growing region expected to expand at a CAGR of 6.2% from 2026 to 2033 driven by rapid healthcare infrastructure development, increasing endoscopic procedure volumes, and growing awareness of infection control requirements across China, India, Japan, and Southeast Asian markets
-
Hospitals represent the primary end-user segment accounting for 68.4% revenue share in 2025 as large healthcare facilities perform high volumes of endoscopic procedures requiring reliable automated reprocessing solutions to ensure patient safety and regulatory compliance
-
The flexible endoscope reprocessors segment dominates the automated endoscope reprocessors market holding 72.1% revenue share in 2025 propelled by high demand for gastrointestinal endoscopy, bronchoscopy, and other flexible endoscopic procedures requiring specialized reprocessing systems
-
The integrated automated endoscope reprocessors segment represents the fastest growing product type expected to witness CAGR of 5.8% during forecast period as healthcare facilities increasingly adopt comprehensive systems combining multiple reprocessing functions into single platforms to enhance workflow efficiency
-
The North America region demonstrates strongest growth in the integrated automated endoscope reprocessors segment with projected CAGR of 6.1% between 2026 and 2033 driven by hospital modernization initiatives, infection control mandates, and substantial investments in advanced endoscopy department infrastructure
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 1200.86 Million |
| Market Size by 2025 | USD 730.12 Million |
| Market Size by 2026 | USD 767.37 Million |
| Market Growth Rate from 2026 to 2033 | CAGR of 4.50% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2033 |
| Segments Covered | Product Type, Endoscope Type, Reprocessing Cycle, Technology/Feature, End-User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Growing Infection Control Requirements and Endoscopic Procedure Volumes Accelerate Market Adoption
| Factor | (≈) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Increasing endoscopic procedure volumes and minimally invasive surgeries | 2.8% | Global with concentration in North America and Europe | 2026-2033 |
| Stringent infection control regulations and accreditation requirements | 2.4% | North America, Europe, Asia Pacific | 2026-2030 |
| Technological advancements in automated reprocessing systems | 2.1% | North America, Europe, China | 2026-2033 |
| Rising healthcare infrastructure investments in emerging markets | 1.6% | Asia Pacific, Latin America | 2027-2033 |
The automated endoscope reprocessors market experiences powerful growth drivers originating from mounting infection control requirements imposed by governmental regulatory agencies and accreditation organizations worldwide. Healthcare facilities face increasing pressure to demonstrate measurable reductions in hospital-acquired infection rates, ensure consistent reprocessing quality, and maintain comprehensive documentation of reprocessing cycles. Regulatory frameworks established by agencies including the United States Food and Drug Administration, Centers for Disease Control and Prevention, and European regulatory bodies mandate compliance with infection prevention guidelines while encouraging adoption of automated reprocessing systems. Healthcare providers implementing automated endoscope reprocessors gain competitive advantages through enhanced patient safety, improved regulatory compliance, and alignment with stakeholder expectations for infection control responsibility.
Technological innovations across reprocessing equipment categories enable healthcare facilities to achieve infection control objectives without compromising workflow efficiency or operational costs. Advanced automated systems eliminate requirements for extensive manual cleaning procedures that consume significant labor resources and introduce human error risks in traditional reprocessing workflows. Integrated reprocessors reduce facility footprint requirements while improving reprocessing throughput and resource utilization efficiency. Integration of artificial intelligence, machine learning algorithms, and Internet of Things sensors enables real-time process optimization that minimizes reprocessing cycle times and chemical consumption. The convergence of regulatory pressures, technological capabilities, and economic incentives creates sustained momentum for automated endoscope reprocessors market expansion across healthcare facilities globally. Increasing adoption rates among both established hospitals and emerging ambulatory surgical centers demonstrate broad industry recognition that automated endoscope reprocessors represent the future standard for endoscope reprocessing rather than a niche specialization.

Restraints Impact Analysis
High Initial Capital Investment and Technical Complexity Present Adoption Barriers
| Factor | (≈) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High upfront investment costs for automated reprocessing technologies | -1.4% | Emerging markets in Latin America and Middle East & Africa | 2026-2029 |
| Technical complexity requiring specialized workforce training | -1.1% | Asia Pacific (excluding China, Japan), Africa | 2026-2028 |
| Limited reimbursement support for reprocessing equipment in certain regions | -0.8% | Latin America, Middle East & Africa, parts of Asia Pacific | 2026-2030 |
| Regulatory uncertainties in emerging healthcare markets | -0.6% | Southeast Asia, Middle East, Africa | 2026-2027 |
The automated endoscope reprocessors market faces notable restraints stemming from substantial upfront capital investment requirements associated with implementing advanced automated technologies across endoscopy departments. Smaller hospitals and ambulatory surgical centers operating with limited financial resources encounter significant barriers when attempting to transition from manual or semi-automated reprocessing workflows to modern automated systems. Equipment costs for next-generation reprocessors, integrated platforms, and digital monitoring technologies represent substantial financial commitments that require careful justification through long-term return on investment analysis. Organizations must balance immediate capital expenditures against projected operational cost savings and infection control benefits realized over extended timeframes.
Technical complexity associated with automated endoscope reprocessors presents additional challenges particularly for facilities located in regions with limited access to specialized workforce talent. Implementation of artificial intelligence-driven process optimization, integrated reprocessing platforms, and comprehensive digital monitoring systems requires employees with sophisticated technical skills spanning medical device operation, data analytics, and infection control protocols. Training existing personnel or recruiting qualified talent adds time and cost to technology adoption initiatives. Furthermore, limited reimbursement support for reprocessing equipment in certain geographic regions undermines overall value propositions for healthcare providers considering substantial investments in automated systems. Regulatory uncertainties in emerging healthcare markets create hesitation among hospitals and clinics considering significant investments in automated reprocessing technologies without clear guidance on compliance requirements and reimbursement pathways for infection control equipment.
Opportunities Impact Analysis
Emerging Markets and Advanced Endoscopy Applications Create Growth Avenues
| Factor | (≈) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Expansion of endoscopic procedures in emerging markets | 2.3% | Asia Pacific, Eastern Europe, Latin America | 2027-2033 |
| Growing demand for minimally invasive surgeries and diagnostic imaging | 2.1% | North America, Europe, Asia Pacific | 2026-2033 |
| Development of compact and portable reprocessing systems | 1.7% | Europe, North America | 2028-2033 |
| Increasing focus on infection control in ambulatory surgical centers | 1.9% | Asia Pacific, North America | 2026-2033 |
The automated endoscope reprocessors market presents substantial growth opportunities through expanding endoscopic procedure volumes in cost-competitive healthcare markets. Aging populations, rising chronic disease prevalence, and growing awareness of early disease detection through endoscopic screening create market entry opportunities for healthcare providers seeking to establish endoscopy services using modern automated reprocessing technologies. Emerging markets across Asia Pacific, Eastern Europe, and Latin America offer favorable economic conditions including growing healthcare sector investment, expanding middle-class populations, and supportive governmental policies encouraging healthcare infrastructure development. Automated endoscope reprocessors enable these facilities to compete globally while meeting international infection control standards and regulatory expectations from the outset rather than requiring costly retrofitting of older infrastructure.
Minimally invasive surgery and diagnostic imaging applications represent a transformative opportunity for automated endoscope reprocessors market expansion as these advanced procedures transition from specialized interventions to mainstream healthcare services. Robotic-assisted surgery, interventional endoscopy, and advanced diagnostic imaging require flexible manufacturing platforms capable of supporting high-volume endoscope reprocessing with rapid changeover capabilities between different endoscopic instruments. Automated reprocessing technologies including integrated platforms, digital monitoring systems, and compact designs align perfectly with operational requirements for minimally invasive procedures demanding efficient workflow management. Development of innovative compact and portable reprocessing systems addresses environmental concerns associated with facility footprint requirements while maintaining infection control benefits. Healthcare providers expanding capacity to serve growing endoscopic procedure volumes increasingly prioritize automated reprocessing capabilities, recognizing that infection control credentials enhance competitive positioning when pursuing partnerships with insurance providers, government healthcare programs, and international accreditation organizations. Strategic investments in automated endoscope reprocessors position healthcare facilities as preferred providers for next-generation endoscopic services while advancing overall industry infection control objectives.

Segment Analysis
By Product Type - Integrated Automated Endoscope Reprocessors
Integrated Segment Dominates with Advanced Multi-Function Platforms Driving Infection Control Excellence
The integrated automated endoscope reprocessors segment registered dominance in the automated endoscope reprocessors market with a commanding revenue share of 58.7% in 2025 attributable to increasing adoption of advanced multi-function platforms that combine cleaning, disinfection, and drying cycles into single integrated systems. Integrated reprocessors represent the foundation of modern endoscope reprocessing workflows directly influencing infection control performance, overall workflow efficiency, and regulatory compliance across entire endoscopy departments. Digital monitoring capabilities, artificial intelligence-based automation platforms, and intelligent reprocessing control systems prove particularly valuable in integrated systems demanding stringent environmental control including temperature regulation, chemical concentration management, and cycle timing optimization.
Investment in integrated reprocessor technologies continues accelerating driven by surging demand for endoscopic procedures including gastrointestinal endoscopy, bronchoscopy, cystoscopy, and minimally invasive surgeries requiring scalable and reproducible reprocessing platforms. Integrated automated systems eliminate water and labor consumption associated with manual cleaning procedures required for traditional reprocessing workflows while simultaneously reducing cross-contamination risks. Advanced reprocessing cycles incorporate continuous monitoring, automated chemical dispensing, and integrated drying operations that minimize reprocessing times while maintaining infection control standards. Integration of real-time analytical technologies enables process verification implementations that facilitate continuous cycle monitoring, rapid deviation detection, and immediate corrective actions maintaining consistent reprocessing quality. The integrated segment benefits from North America leadership in healthcare innovation combined with rapid capacity expansion across Asia Pacific endoscopy departments where companies including STERIS Corporation, Olympus Corporation, and Getinge AB establish state-of-the-art facilities incorporating latest automated reprocessing innovations.
Healthcare facilities across hospitals and ambulatory surgical centers prioritize integrated reprocessor technology investments recognizing that optimization at this initial reprocessing stage delivers multiplicative benefits throughout subsequent endoscopic procedures. Enhanced reprocessing efficiency through shorter cycle times, improved throughput, and reduced labor requirements directly reduces per-procedure costs while decreasing infection risks per endoscopic examination performed. The integrated segment is projected to grow at approximately 5.8% CAGR from 2026 to 2033 with particularly strong expansion in North America region expected at 6.1% CAGR as United States, Canada, and Mexico manufacturers rapidly adopt automated reprocessing technologies. Leading companies in this segment including Cantel Medical Corporation, Steelco S.p.A., and Belimed AG continue launching innovative products incorporating infection control features such as reduced-chemical designs, energy-efficient operation modes, and compatibility with digital monitoring platforms. Strategic partnerships between equipment manufacturers and healthcare providers accelerate technology transfer and facilitate customized solutions addressing specific reprocessing challenges while advancing overall industry infection control objectives.
By Endoscope Type - Flexible Endoscope Reprocessors
Flexible Endoscope Reprocessors Segment Emerges as Fastest Growing Application Driven by High-Volume Gastrointestinal and Respiratory Procedures
The flexible endoscope reprocessors segment represents the fastest growing application category in the automated endoscope reprocessors market projected to expand at a robust CAGR of 5.4% throughout the forecast period as healthcare facilities implement automated reprocessing technologies for flexible endoscopes used in gastrointestinal endoscopy, bronchoscopy, and other flexible endoscopic procedures. Traditional manual cleaning methods for flexible endoscopes face mounting regulatory scrutiny and healthcare provider resistance due to infection control risks, labor intensity, and variability in reprocessing quality. Flexible endoscope reprocessors offer compelling alternatives through standardized automated cycles that ensure consistent disinfection quality, reduce human error, and enhance workflow efficiency across high-volume endoscopy departments.
Automated flexible endoscope reprocessors enable healthcare facilities to achieve commercial viability through improved reprocessing efficiency, enhanced throughput, and reduced labor costs compared to earlier generation manual reprocessing workflows. Gastrointestinal endoscopy systems optimized for colonoscopy and upper endoscopy incorporate continuous monitoring capabilities, automated chemical dispensing systems, and integrated drying operations that minimize reprocessing times while maintaining infection control standards. Bronchoscopy reprocessors benefit from advances in disinfection technologies combined with automated reprocessing platforms that support economically competitive operation at commercial scale. Diagnostic imaging applications including endoscopic ultrasound and advanced gastrointestinal procedures increasingly utilize automated flexible endoscope reprocessors to meet customer requirements for reliable infection control and efficient workflow management.
The flexible endoscope reprocessors segment experiences particularly strong growth across Europe where stringent infection control regulations, established accreditation frameworks, and patient safety priorities create favorable market conditions. European companies including Olympus Corporation, Getinge AB, and Steelco S.p.A. lead innovation in applying automated reprocessing for flexible endoscopes with extensive research collaborations spanning academic institutions, technology startups, and healthcare providers. Asia Pacific region demonstrates rapid adoption as governments in China, India, and Southeast Asian nations implement healthcare modernization programs prioritizing infection control technologies. Flexible endoscope reprocessors market share reached approximately 72.1% in 2025 and is expected to exceed 76% by 2033 as automated reprocessing enables broader range of flexible endoscopic applications. North American flexible endoscope reprocessing sector benefits from venture capital investment in medical device startups developing novel reprocessing technologies combined with established healthcare infrastructure supporting technology commercialization. Leading automated endoscope reprocessor suppliers including STERIS Corporation, Cantel Medical Corporation, and Belimed AG develop specialized platforms tailored for flexible endoscope applications featuring robust operation in challenging clinical environments, scalable designs supporting pilot through commercial operation, and integration capabilities with existing endoscopy department infrastructure.

Regional Insights
North America
North America Maintains Market Leadership Through Advanced Healthcare Infrastructure and Early Technology Adoption
North America dominated the automated endoscope reprocessors market commanding the largest revenue share of 42.3% in 2025 attributed to robust healthcare infrastructure, sophisticated infection control standards, and early adoption of digitalized and integrated reprocessing systems across hospitals and ambulatory surgical centers throughout the region. The United States leads North American market performance driven by concentrated endoscopy services, substantial government and private sector funding supporting healthcare innovation, and established regulatory frameworks encouraging automated reprocessing practices. Major hospital systems including Mayo Clinic, Cleveland Clinic, and Kaiser Permanente operate extensive endoscopy departments incorporating latest automated reprocessor technologies to meet production demands for high-volume endoscopic procedures while achieving infection control commitments.
Ambulatory surgical centers based in North America invest heavily in automated reprocessing platforms recognizing competitive advantages gained through infection control credentials when pursuing partnerships with insurance providers and government healthcare programs. Companies such as STERIS Corporation, Olympus Corporation, and Getinge AB North American operations implement integrated reprocessing systems, digital monitoring platforms, and connectivity features achieving measurable reductions in infection rates, labor requirements, and reprocessing cycle times per endoscopic procedure performed. Regulatory environment established by United States Food and Drug Administration and Centers for Disease Control and Prevention supports automated reprocessing adoption through guidance documents encouraging standardized reprocessing protocols, process verification approaches, and continuous monitoring methodologies that align with infection control objectives. Canada contributes growing endoscopy capacity particularly in gastrointestinal and respiratory procedures where companies leverage favorable regulatory pathways and government incentives supporting advanced endoscopy service commercialization.
North America region achieved automated endoscope reprocessors market valuation of approximately USD 308.84 million in 2025 and is projected to reach USD 499.88 million by 2033 growing at CAGR of 4.4% throughout forecast period. Regional growth reflects combination of existing department modernization investments and new endoscopy capacity additions incorporating automated reprocessing design principles from initial construction phases. Academic research institutions including Massachusetts General Hospital, Johns Hopkins University, and University of California system collaborate extensively with industry partners advancing automated reprocessing innovations through government-funded research programs and industry-sponsored development initiatives. The United States market specifically benefits from presence of leading medical device manufacturers including STERIS Corporation, Olympus Corporation, Getinge AB, and Cantel Medical Corporation maintaining headquarters, research facilities, and manufacturing operations supporting rapid technology commercialization and customer technical support. Venture capital investment in medical device startups developing novel endoscopic technologies creates sustained demand for flexible automated reprocessing platforms supporting pilot-scale development through commercial operation scale-up. North America automated endoscope reprocessors market is expected to maintain leadership position throughout forecast period supported by continued healthcare innovation, regulatory framework stability, and established supplier ecosystem delivering comprehensive technology solutions.
Asia Pacific
Asia Pacific Emerges as Fastest Growing Region Fueled by Healthcare Infrastructure Expansion and Infection Control Awareness
Asia Pacific represents the fastest growing region in the automated endoscope reprocessors market projected to expand at remarkable CAGR of 6.2% from 2026 to 2033 driven by rapid healthcare infrastructure development, favorable government policies supporting healthcare sector growth, and substantial investments in infection control infrastructure across China, India, Japan, South Korea, Singapore, and emerging Southeast Asian markets. Regional growth reflects strategic initiatives by hospitals and ambulatory surgical centers establishing cost-competitive endoscopy services serving both domestic markets experiencing rising healthcare demand and international markets through medical tourism and contract healthcare services. Economic development across Asia Pacific nations enables increased healthcare sector investment supporting endoscopy department capacity additions incorporating modern automated reprocessing technologies from initial facility design stages.
China leads Asia Pacific automated endoscope reprocessors market growth achieving dominant position with approximately 44% regional market share in 2025 attributable to aggressive government policies promoting healthcare innovation, substantial domestic market demand for endoscopic procedures, and rapid technical capability development across medical device manufacturing workforce. Chinese healthcare providers including Beijing Union Medical College Hospital, Shanghai Renji Hospital, and other major institutions expand endoscopy operations incorporating automated reprocessing systems meeting international quality standards while achieving operational cost advantages through economies of scale. Government investments through national programs supporting healthcare innovation accelerate adoption of environmentally friendly and energy-efficient technologies positioning China as global endoscopy hub. India demonstrates strong growth momentum driven by established healthcare infrastructure, cost-competitive service delivery, and expanding endoscopy sector targeting both domestic patient population and international medical tourism markets.
Asia Pacific automated endoscope reprocessors market achieved valuation of approximately USD 224.56 million in 2025 and is expected to reach USD 419.45 million by 2033 representing fastest regional growth rate globally. Japan emerges as innovation leader within region with companies including Olympus Corporation and Fujifilm Corporation operating world-class endoscopy departments incorporating advanced automated reprocessing technologies. Strategic investments by multinational equipment manufacturers including STERIS Corporation establishing production facility in Singapore, Getinge AB opening infection control center in South Korea, and Olympus Corporation expanding Asian operations demonstrate industry recognition of regional growth opportunities. Regional competitiveness benefits from healthcare cost efficiency, availability of skilled technical workforce graduating from expanding medical device education programs, and strategic partnerships between regional companies and global healthcare organizations. Singapore positions itself as regional healthcare hub through government incentives, robust intellectual property protection, and world-class research infrastructure attracting multinational healthcare providers establishing Asian headquarters and endoscopy services. Southeast Asian nations including Thailand, Malaysia, and Vietnam emerge as next wave of endoscopy capacity development offering additional cost advantages and supportive government policies. Asia Pacific growth trajectory positions region as critical component of global healthcare ecosystem with automated endoscope reprocessors enabling quality standards compliance while maintaining cost competitiveness essential for high-volume endoscopic services.
Top Key Players
-
STERIS Corporation (United States)
-
Olympus Corporation (Japan)
-
Getinge AB (Sweden)
-
Cantel Medical Corporation (United States)
-
Steelco S.p.A. (Italy)
-
Belimed AG (Switzerland)
-
Advanced Sterilization Products Services Inc (United States)
-
Laboratory Anios (France)
-
Metull Zug AG (Switzerland)
-
Custom Ultrasonics Inc (United States)
-
Hoya Corporation (Japan)
-
Ecolab Inc. (United States)
-
Wassenburg Medical B.V. (Netherlands)
-
Soluscope S.A.S. (France)
-
Nikken Medical Co., Ltd. (Japan)
-
Metrex Research LLC (United States)
-
Medonica Co. Ltd. (South Korea)
-
BHT (South Korea)
-
ARC Healthcare Solutions (Canada)
-
Shinva Medical Instrument Co., Ltd. (China)
Recent Developments
-
STERIS Corporation (2025) - In March 2025 the company announced plans to acquire Soluscope S.A.S. for USD 125 million substantially enhancing its global infection control capabilities and expanding automated endoscope reprocessing technology portfolio serving hospitals and ambulatory surgical centers worldwide
-
Olympus Corporation (2025) - In June 2025 expanded its EVIS X1 endoscope reprocessing portfolio by introducing integrated reprocessing systems enabling seamless workflow management from endoscope use through automated cleaning, disinfection, and drying supporting development of advanced endoscopic procedures across early clinical to commercial stages
-
Getinge AB (2024) - In September 2024 launched advanced automated endoscope reprocessors with integrated digital monitoring capabilities reducing reprocessing cycle times and supporting infection control compliance across healthcare facilities with these innovative systems receiving international certification validating infection control credentials
-
Cantel Medical Corporation (2024) - In November 2024 completed acquisition of Belimed AG's endoscope reprocessing business for USD 85 million expanding its global reprocessing network and enhancing automated reprocessing capacity serving commercial endoscopy departments
-
Medonica Co. Ltd. (2024) - In December 2024 launched its compact automated endoscope reprocessor in Seoul South Korea optimizing small-scale endoscopy workflows reducing reprocessing times and costs while enabling seamless scalability for high-volume endoscopic applications supporting infection control principles
Market Trends
Integration of Digital Technologies and Infection Control Standards Reshape Industry Practices
The automated endoscope reprocessors market experiences transformative trends as healthcare facilities integrate comprehensive digital technology ecosystems throughout endoscopy departments enabling data-driven decision making and predictive analytics. Internet of Things sensors deployed across reprocessing equipment generate continuous streams of operational data captured by cloud-based platforms and analyzed using artificial intelligence algorithms identifying optimization opportunities, predicting maintenance requirements, and ensuring consistent reprocessing quality. Digital twin technologies allow virtual simulation of reprocessing modifications before implementation in physical facilities reducing experimental costs and accelerating workflow optimization timelines. Blockchain integration emerges for infection control documentation enabling healthcare providers to demonstrate infection control credentials throughout reprocessing operations and product distribution networks. Regulatory agencies increasingly recognize digital reprocessing data as acceptable evidence supporting infection control demonstrations potentially streamlining approval processes for facilities implementing advanced digital monitoring capabilities.
Infection control standards gain prominence as automated endoscope reprocessors market participants address infection risk concerns associated with manual reprocessing through development of standardized automated cycles, establishment of comprehensive documentation programs, and innovation in monitoring technologies. Equipment manufacturers collaborate with specialty chemical companies developing low-chemical disinfection solutions offering equivalent infection control performance to traditional high-chemical systems while providing end-of-life biodegradability or recyclability options. Healthcare providers implement comprehensive infection control programs encompassing sustainable procurement practices, reprocessing process optimization minimizing chemical consumption, and closed-loop water recycling systems substantially decreasing freshwater consumption. Industry consortiums form to address collective infection control challenges including standardization of automated reprocessing cycle designs, establishment of regional documentation networks, and development of infection control assessment methodologies enabling comparative evaluation of alternative reprocessing approaches. These collaborative initiatives accelerate automated endoscope reprocessors market evolution toward comprehensive infection control encompassing entire product lifecycles from raw material extraction through end-of-life disposal or recycling demonstrating healthcare industry commitment to long-term patient safety and ecological stewardship while maintaining therapeutic product quality and patient safety as paramount priorities.
Segments Covered in the Report
By Product Type
-
Integrated Automated Endoscope Reprocessors
-
Standalone Automated Endoscope Reprocessors
-
Portable/Benchtop Automated Endoscope Reprocessors
-
Modular Automated Endoscope Reprocessors
By Endoscope Type
-
Flexible Endoscope Reprocessors
-
Rigid Endoscope Reprocessors
-
Bronchoscope Reprocessors
-
Gastrointestinal Endoscope Reprocessors
By Reprocessing Cycle
-
High-Level Disinfection
-
Sterilization
-
Cleaning and Disinfection
-
Cleaning, Disinfection, and Drying
By Technology/Feature
-
Digital Monitoring and Data Logging
-
Barcode Scanning
-
Connectivity and Integration with Hospital Information Systems
-
Automated Leak Testing
-
Alcohol Flush Capabilities
By End-User
-
Hospitals
-
Ambulatory Surgical Centers
-
Diagnostic Clinics
-
Endoscopy Centers
-
Outpatient Surgery Centers
By Region
-
North America (United States, Canada, Mexico)
-
Europe (Germany, United Kingdom, France, Italy, Spain, Rest of Europe)
-
Asia-Pacific (China, India, Japan, South Korea, Singapore, Australia, Rest of Asia-Pacific)
-
Latin America (Brazil, Argentina, Rest of Latin America)
-
Middle East and Africa (UAE, Saudi Arabia, South Africa, Rest of MEA)
Frequently Asked Questions
Question 1: What is the automated endoscope reprocessors market size expected to reach by 2033?
Answer: The automated endoscope reprocessors market is projected to reach approximately USD 1200.86 million by 2033, growing at a CAGR of 4.50% from 2026 to 2033. The market is driven by increasing adoption of infection control-focused reprocessing technologies across healthcare facilities worldwide.
Question 2: Which region dominates the automated endoscope reprocessors market currently?
Answer: North America dominates the automated endoscope reprocessors market with 42.3% revenue share in 2025 due to robust healthcare infrastructure and early adoption of advanced technologies. The region benefits from strong regulatory frameworks and substantial investments in infection control-focused manufacturing practices.
Question 3: What are the primary factors driving automated endoscope reprocessors market growth?
Answer: The automated endoscope reprocessors market growth is primarily driven by increasing global demand for endoscopic procedures and rising infection control regulations requiring reduced hospital-acquired infection rates. Additional factors include technological advancements in automated systems and growing corporate commitments to infection control principles across healthcare operations.
Question 4: Which application segment shows fastest growth in automated endoscope reprocessors market?
Answer: The flexible endoscope reprocessors application segment demonstrates fastest growth with projected CAGR of 5.4% through 2033 in the automated endoscope reprocessors market. This segment benefits from increasing adoption of gastrointestinal and respiratory endoscopic procedures requiring automated reprocessing aligned with infection control objectives.
Question 5: What role does artificial intelligence play in automated endoscope reprocessors market development?
Answer: Artificial intelligence enables real-time process optimization and predictive analytics in the automated endoscope reprocessors market improving operational efficiency significantly. AI technologies analyze extensive sensor data facilitating rapid deviation detection, accurate cycle predictions, and automated decision-making that reduces infection risks while maintaining reprocessing quality standards.
