Anthrax Vaccine Market Overview
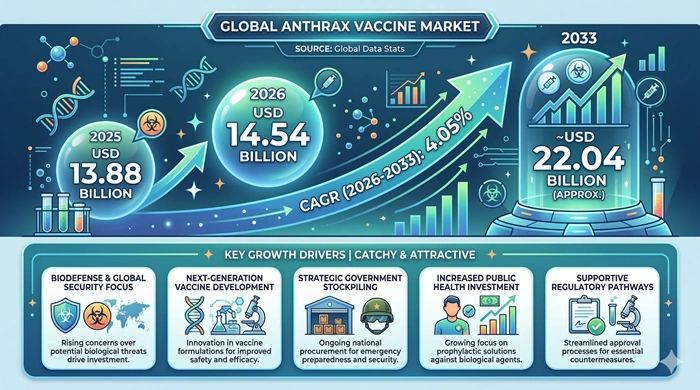
The global Anthrax Vaccine market size is valued at USD 13.88 billion in 2025 and is predicted to increase from USD 14.54 billion in 2026 to approximately USD 22.04 billion by 2033, growing at a CAGR of 4.05% from 2026 to 2033.
Anthrax vaccines are specialized immunological products developed to protect against infection by Bacillus anthracis — a highly dangerous spore-forming bacterium classified as a Category A bioterrorism agent by the U.S. CDC. These vaccines are primarily administered as pre-exposure and post-exposure prophylaxis to high-risk individuals including military personnel, laboratory researchers, veterinarians, and emergency response teams. The anthrax vaccine market is fundamentally shaped by national biodefense priorities, government stockpile mandates, and sustained military immunization program requirements — making it one of the most policy-driven segments within the broader vaccine and biodefense industries. Growing international investment in biological threat preparedness and advancing next-generation vaccine technologies are underpinning the market's steady expansion through 2033.
AI Impact on the Anthrax Vaccine Industry
Artificial Intelligence Is Accelerating Next-Generation Anthrax Vaccine Development, Strengthening Biodefense Biosurveillance, and Optimizing Strategic Stockpile Management to Meaningfully Advance Global Biological Threat Preparedness
Artificial intelligence is reshaping key dimensions of the Anthrax Vaccine market, most notably in the areas of vaccine antigen design and biological threat surveillance. AI-powered computational platforms — including protein structure prediction and molecular dynamics modeling tools — are enabling researchers to analyze the genetic architecture and antigenic components of Bacillus anthracis with unprecedented speed and precision, identifying novel protective antigen epitopes and designing next-generation recombinant vaccine candidates with enhanced immunogenicity and thermostability. These computational capabilities are compressing the preclinical discovery timeline significantly, allowing biodefense vaccine developers to advance promising new anthrax vaccine candidates toward clinical evaluation far more rapidly than traditional laboratory-based methods permit.
Beyond research and development, AI is also improving the practical management of government anthrax vaccine stockpiles and emergency distribution logistics. Machine learning algorithms applied to stockpile inventory systems can now optimize vaccine lot rotation schedules, predict expiry-related wastage, and model emergency deployment scenarios — ensuring that government reserves remain deployment-ready and cost-efficiently managed. AI-driven biosurveillance systems analyzing environmental sensor data, hospital admission patterns, and syndromic surveillance signals are simultaneously improving the early detection capabilities for potential anthrax release events, supporting faster and more targeted public health responses that would include emergency anthrax vaccine prophylaxis deployment. These cumulative AI applications are progressively elevating the operational readiness and scientific advancement of the anthrax vaccine industry.
Growth Factors
Persistent Bioterrorism Threat Environment, Mandatory Military Immunization Programs, Expanding Government Biodefense Stockpile Investment, and Advancing Recombinant Vaccine Technologies Are the Four Core Drivers of Anthrax Vaccine Market Growth
The primary and most stable driver of the Anthrax Vaccine market is the persistent and policy-embedded global threat of anthrax as a biological weapon and the resulting institutional imperative to maintain robust military and civilian biodefense immunization programs. The 2001 U.S. anthrax letter attacks provided a definitive real-world demonstration of anthrax's viability as a bioterrorism agent, and governments worldwide have since maintained uninterrupted investment in anthrax vaccine procurement and strategic stockpile maintenance as a core component of their biodefense preparedness infrastructure. Ongoing geopolitical tensions in multiple global regions — including sustained concerns about state-sponsored biological weapons programs — are reinforcing government commitment to anthrax medical countermeasure investment, creating a stable and policy-driven demand foundation that is structurally insulated from conventional healthcare market cycles.
The mandatory anthrax vaccination programs for military personnel in the United States and allied nations represent the single largest and most consistent demand channel in the anthrax vaccine market. U.S. Department of Defense contracts with Emergent BioSolutions for BioThrax supply — and more recently for the newer CYFENDUS anthrax vaccine — have been sustained through multi-year procurement agreements supported by BARDA funding, ensuring predictable and substantial revenue streams for licensed anthrax vaccine manufacturers. The expansion of biodefense preparedness planning in NATO member states and in strategically important countries across Asia Pacific and the Middle East is progressively broadening the geographic base of government anthrax vaccine procurement, creating new international revenue opportunities. Simultaneously, the active clinical development pipeline for next-generation recombinant anthrax vaccines — including GC Biopharma's Barythrax, which received approval in South Korea in 2025 — is expanding the long-term product and revenue landscape of the market.

Market Outlook
Sustained Government Biodefense Investment, Pipeline Maturation of Next-Generation Recombinant Vaccines, and Expanding International Biopreparedness Programs Create a Consistently Positive Anthrax Vaccine Market Outlook Through 2033
The long-term outlook for the Anthrax Vaccine market is solidly positive and structurally well-supported. The forecast period from 2026 to 2033 is expected to be defined by continued strong government procurement of existing anthrax vaccine products for strategic national stockpiles and military programs, supplemented by the progressive regulatory approval and commercial launch of next-generation recombinant anthrax vaccine candidates. These newer products — designed with improved dosing convenience, better tolerability profiles, and longer stockpile shelf lives — are expected to earn regulatory approvals in multiple markets during the forecast period, introducing new premium-priced revenue streams and potentially repositioning the competitive landscape of the licensed anthrax vaccine space.
The international dimension of the anthrax vaccine market is also expected to grow in commercial significance during the forecast period. Asia Pacific governments — particularly India, China, South Korea, and Australia — are investing in national biodefense stockpiles and expanding both human and veterinary anthrax vaccination programs in response to growing biological threat awareness and high livestock-sector anthrax risks. These investments are creating new procurement opportunities for both established and emerging anthrax vaccine manufacturers beyond the traditional North American and European market base. BARDA's continued funding of advanced development programs ensures that the U.S. biodefense vaccine pipeline remains robust, while international multilateral investments through organizations such as the Global Health Security Agenda are further broadening the scope of global anthrax preparedness investment.
Expert Speaks
-
Robert Kramer, CEO, Emergent BioSolutions — "Our commitment to biodefense preparedness is unwavering, and we are proud to be the primary supplier of anthrax vaccines to the U.S. Strategic National Stockpile and military forces. The continued government investment in anthrax vaccine stockpiling and the advancement of next-generation anthrax immunization technologies reflects the shared recognition that maintaining robust biodefense capabilities is a non-negotiable national security priority."
-
Albert Bourla, CEO, Pfizer — "Biodefense preparedness demands the same scientific rigor and manufacturing commitment as any other area of vaccine development. The pharmaceutical industry plays a critical role in ensuring that governments have reliable, high-quality anthrax vaccine supplies and medical countermeasures to protect both military and civilian populations against biological threat agents."
-
Joaquin Duato, CEO, Johnson & Johnson — "The development and reliable supply of biodefense vaccines — including those protecting against anthrax — requires sustained partnership between the public and private sectors. Without continued government investment in advanced development and procurement, the commercial incentive structures for maintaining and improving the anthrax vaccine supply are insufficient to meet national security requirements alone."
Key Report Takeaways
-
North America dominates the Anthrax Vaccine market, holding approximately 44.70% of global revenue share in 2025, driven by the United States' large-scale military anthrax vaccination program, the world's most extensively funded Strategic National Stockpile, sustained BARDA investment in anthrax medical countermeasure development and procurement, and the presence of the primary licensed anthrax vaccine manufacturer — Emergent BioSolutions — in the U.S.
-
Asia Pacific is the fastest-growing regional market for anthrax vaccines, driven by growing government recognition of biological and zoonotic disease threats, expanding national biodefense stockpiling programs in South Korea, India, and China, high livestock population density creating sustained veterinary anthrax vaccine demand, and the recent landmark approval of GC Biopharma's Barythrax as the world's first licensed recombinant anthrax vaccine.
-
Government stockpiles and public health agencies are the dominant end-user segment, holding approximately 44.60% of market share in 2025, as governments worldwide maintain large strategic reserves of anthrax vaccines for military immunization programs and emergency civilian response — generating the highest consistent procurement volumes in the market through direct government-to-manufacturer contract channels.
-
Pre-exposure prophylaxis is the leading indication segment, accounting for approximately 58.70% of global market revenue in 2025, driven by the large-scale mandatory pre-exposure vaccination programs for military personnel, laboratory workers, veterinarians, and other high-risk occupational groups — representing the most consistent and highest-volume anthrax vaccine demand channel globally.
-
Cell-free protective antigen vaccine adsorbed (BioThrax/AVA) is the dominant vaccine platform segment, holding approximately 63.40% of market share in 2025, as the longest-established and only historically FDA-licensed anthrax vaccine with decades of clinical use, established manufacturing infrastructure, and deep institutional familiarity among military and government procurement authorities.
-
Recombinant protective antigen subunit vaccine is the fastest-growing platform segment, expected to grow at a CAGR of approximately 9.60% through 2033, driven by the 2025 approval of GC Biopharma's Barythrax in South Korea — the world's first recombinant anthrax vaccine — and by the advanced development of multiple additional recombinant candidates supported by BARDA funding, offering improved safety, tolerability, and dosing convenience over conventional vaccine formulations.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 22.04 Billion |
| Market Size by 2025 | USD 13.88 Billion |
| Market Size by 2026 | USD 14.54 Billion |
| Market Growth Rate (2026–2033) | CAGR of 4.05% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Segments Covered | Vaccine Platform, Indication, End User, Procurement Channel |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Persistent Biological Threat Environment, Military Immunization Mandates, BARDA-Funded Stockpile Investment, and Next-Generation Recombinant Vaccine Approvals Are the Four Pillars Sustaining Anthrax Vaccine Market Demand
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Persistent bioterrorism threat awareness driving biodefense investment | ~34% | Global — especially North America, Europe | Short to Long-term |
| Mandatory military anthrax vaccination programs in U.S. and allied nations | ~30% | North America, Europe, Asia Pacific | Short to Long-term |
| Government strategic national stockpile procurement and maintenance | ~24% | North America, Europe | Short to Medium-term |
| Next-generation recombinant vaccine approvals expanding product pipeline | ~12% | North America, Asia Pacific | Medium to Long-term |
The foundational and most durable demand driver in the Anthrax Vaccine market is the sustained, policy-mandated requirement for anthrax immunization in military populations combined with the maintenance of government-funded strategic national stockpiles. Anthrax's classification as a Category A bioterrorism agent by the U.S. CDC has driven decades of uninterrupted government investment in anthrax vaccine procurement, development funding, and stockpile maintenance — investment that is renewed through recurring multi-year contract cycles between government agencies and licensed vaccine manufacturers. Recent contract extensions — including Emergent BioSolutions' USD 250 million BARDA contract extension for CYFENDUS supply to the Strategic National Stockpile announced in July 2024 — demonstrate the continued strength and predictability of this government-driven demand channel.
The expansion of mandatory military anthrax vaccination requirements beyond the United States to allied and partner nation militaries is progressively broadening the institutional demand base for the anthrax vaccine market. NATO member states and strategic partners in Asia Pacific are investing in anthrax vaccine procurement for their defense forces as part of broader biological threat preparedness upgrades, creating new international government procurement opportunities for licensed anthrax vaccine manufacturers. The April 2025 South Korean government approval of Barythrax — the world's first recombinant anthrax vaccine, developed by GC Biopharma in partnership with the Korea Disease Control and Prevention Agency — marks a significant milestone in expanding the licensed anthrax vaccine landscape beyond the United States and signaling the beginning of a new international regulatory approval cycle for next-generation anthrax vaccine products.

Restraints Impact Analysis
Highly Concentrated Demand Dependent on Government Procurement Cycles, Limited Commercial Market Beyond Defense, Complex Regulatory Requirements, and Adverse Effect Concerns Restrain Faster Anthrax Vaccine Market Growth
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Near-total dependence on government procurement cycles and budgets | ~36% | Global — especially North America | Short to Long-term |
| Limited commercial civilian market outside defense and occupational use | ~28% | Global | Short to Long-term |
| Complex regulatory requirements for new anthrax vaccine approvals | ~24% | North America, Europe | Medium-term |
| Historical adverse effect profile concerns among military vaccine recipients | ~12% | North America, Europe | Short to Medium-term |
The Anthrax Vaccine market's most significant structural restraint is its near-total dependence on government procurement decisions — particularly from the U.S. Department of Defense and BARDA — for the majority of its commercial revenue. This concentration of demand in a single or small number of large government buyers creates inherent revenue volatility, as contract award timing, budget allocation decisions, and policy shifts can cause significant fluctuations in annual procurement volumes. Unlike commercial pharmaceutical markets where demand is distributed across millions of individual prescriptions, the anthrax vaccine market's revenue is heavily dependent on periodic, large-volume government contract awards that can create boom-and-bust procurement cycles for manufacturers. This structural characteristic limits the market's ability to achieve the smooth, consistent growth trajectory typical of broader vaccine markets.
The limited addressable civilian market for anthrax vaccines represents a second fundamental commercial constraint. Outside of occupational risk groups — including laboratory workers handling Bacillus anthracis, veterinarians in endemic regions, and certain agricultural workers — there is no meaningful civilian consumer demand for anthrax vaccination. This narrow end-user base outside the defense sector means that the total addressable market is structurally capped at a far lower level than most other vaccine categories. Historical reports of injection-site reactions and systemic adverse effects associated with the conventional BioThrax vaccine have also generated some resistance to anthrax vaccination among military personnel, contributing to compliance challenges that affect total vaccine consumption volumes in the military end-user segment.
Opportunities Impact Analysis
Next-Generation Recombinant Vaccine Commercialization, International Biodefense Stockpile Expansion, Veterinary Anthrax Vaccination Growth, and Animal Sector Endemic Region Programs Create Meaningful Untapped Market Opportunities
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Commercial launch and international approval of recombinant anthrax vaccines | ~36% | North America, Asia Pacific, Europe | Medium to Long-term |
| International biodefense stockpile expansion in Asia Pacific and Middle East | ~28% | Asia Pacific, Middle East | Short to Medium-term |
| Growth of veterinary anthrax vaccination programs in endemic regions | ~24% | Asia Pacific, Latin America, Africa | Short to Medium-term |
| Hospitals and clinics segment expansion for post-exposure preparedness | ~12% | Global | Medium-term |
The commercial launch and international regulatory approval of next-generation recombinant anthrax vaccines represents the most strategically significant long-term opportunity in the Anthrax Vaccine market. Products like GC Biopharma's Barythrax — which received South Korean regulatory approval in April 2025 — and multiple BARDA-funded recombinant candidates in advanced U.S. clinical development offer substantially improved profiles over the conventional BioThrax vaccine: fewer required doses, better tolerability, longer stockpile shelf life, and stronger immunogenicity. Successful international regulatory approvals for these products would meaningfully expand both the licensed vaccine choice set for government procurement authorities and the total market revenue potential, potentially displacing portions of conventional anthrax vaccine procurement with higher-value recombinant product contracts.
The growth of veterinary anthrax vaccination programs in endemic regions — particularly across sub-Saharan Africa, Central Asia, and parts of South America where anthrax outbreaks in livestock remain a recurring economic and zoonotic health challenge — represents an important and growing commercial opportunity that broadens the anthrax vaccine market beyond its traditional biodefense core. Governments and international agricultural health agencies in these regions are investing in expanded livestock anthrax vaccination campaigns to protect agricultural economies and reduce the risk of zoonotic transmission to human populations. This veterinary segment is served by several specialized animal health companies including Zoetis, Bayer Animal Health, and regional manufacturers, and its growth is expected to accelerate as agricultural health infrastructure strengthens in key endemic-region markets during the 2026–2033 forecast period.

Segment Analysis
By Vaccine Platform
Cell-Free Protective Antigen Vaccine Adsorbed Leads the Anthrax Vaccine Market as the Established Clinical Gold Standard, While Recombinant Subunit Vaccines Rapidly Emerge as the Highest-Growth Next-Generation Platform
The cell-free protective antigen vaccine adsorbed segment — represented primarily by BioThrax (AVA) and the newer CYFENDUS (AV7909) from Emergent BioSolutions — dominates the vaccine platform segment, accounting for approximately 63.40% of total Anthrax Vaccine market revenue in 2025. This segment's overwhelming market leadership reflects its status as the only historically licensed anthrax vaccine in the United States, with decades of regulatory approval, established manufacturing infrastructure, extensive military clinical use data, and deep institutional familiarity among government procurement authorities. The mandatory U.S. military anthrax vaccination program — which has been running continuously for decades — generates the largest consistent procurement volumes for this vaccine platform, and multi-year government supply contracts provide highly predictable revenue for the segment's primary manufacturer, Emergent BioSolutions. North America accounts for the dominant share of this segment's global revenue, driven by U.S. DoD and SNS procurement contracts, with the U.S. market generating approximately USD 4.79 billion in anthrax vaccine revenue in 2025 alone.
The recombinant protective antigen subunit vaccine segment is the fastest-growing platform in the Anthrax Vaccine market, projected to expand at a CAGR of approximately 9.60% from 2026 to 2033, driven by the landmark 2025 South Korean regulatory approval of GC Biopharma's Barythrax and the advanced development of multiple BARDA-funded recombinant candidates approaching regulatory submission in the United States. These next-generation recombinant vaccines use genetically engineered protective antigen proteins to elicit strong and durable immune responses with significantly improved safety profiles, fewer required doses, and better manufacturing scalability compared to conventional cell-free protective antigen formulations. Asia Pacific is the fastest-growing region for recombinant anthrax vaccine adoption, driven by South Korea's pioneering regulatory milestone with Barythrax and by growing government interest across the region in next-generation biodefense vaccine technologies that offer practical advantages for military immunization and stockpile management.
By Indication
Pre-Exposure Prophylaxis Anchors the Anthrax Vaccine Market With the Largest Institutional Demand Base, While Post-Exposure Prophylaxis Emerges as the Fastest-Growing Indication Driven by Emergency Preparedness Investment
Pre-exposure prophylaxis (PrEP) is the dominant indication segment in the Anthrax Vaccine market, accounting for approximately 58.70% of total global market revenue in 2025. The PrEP segment's dominance reflects the large-scale mandatory pre-exposure vaccination programs administered to military personnel, laboratory researchers, veterinarians, and other occupational high-risk groups across North America and allied nations — populations for whom regular anthrax booster vaccination is a standing institutional requirement. Government defense agencies and public health organizations strongly prioritize pre-exposure immunization programs because they provide long-term immunological protection before any potential Bacillus anthracis exposure event, making them the most proactive and cost-effective biodefense strategy available. North America holds the largest PrEP anthrax vaccine market share, driven entirely by the scale of the U.S. military vaccination program and BARDA-funded SNS procurement, with Emergent BioSolutions holding dominant market position through its BioThrax and CYFENDUS product franchises.
Post-exposure prophylaxis (PEP) represents the fastest-growing indication segment in the anthrax vaccine market, projected to expand at a CAGR of approximately 6.30% from 2026 to 2033, driven by growing government investment in emergency response capabilities for anthrax exposure events and by the clinical advancement of next-generation anthrax vaccines with improved speed of immune response induction. The PEP segment is administered following confirmed or suspected exposure to anthrax spores — typically in combination with antibiotic therapy — to prevent disease onset in exposed individuals. Healthcare institutions and government emergency response agencies are investing in expanding PEP vaccine stockpiles and establishing efficient distribution protocols to ensure rapid deployment following potential bioterrorism incidents or industrial accidents. North America and Europe are the primary growth markets for PEP anthrax vaccine procurement, where detailed emergency response plans for potential anthrax release scenarios include specific vaccine deployment logistics aligned with national biodefense preparedness frameworks.

Regional Insights
North America
North America Leads the Global Anthrax Vaccine Market With the Dominant Revenue Share, Largest Government Biodefense Investment, and Most Comprehensive Strategic Stockpile and Military Immunization Infrastructure
North America holds the dominant position in the global Anthrax Vaccine market, accounting for approximately 44.70% of total global revenue in 2025, and is projected to maintain its regional leadership at a CAGR of approximately 3.76% from 2026 to 2033. The United States is the overwhelmingly dominant national market within the region, where the U.S. Department of Defense's mandatory military anthrax vaccination program, the world's most extensively funded Strategic National Stockpile, and BARDA's continuous investment in anthrax medical countermeasure development and procurement collectively generate the largest anthrax vaccine procurement volumes globally. The U.S. anthrax vaccine market alone was valued at approximately USD 4.79 billion in 2025, with the majority of this revenue generated through large government supply contracts with Emergent BioSolutions — the manufacturer of both BioThrax and CYFENDUS, the two most commercially significant anthrax vaccine products in the North American market.
Canada contributes to regional market activity through its own biodefense stockpile maintenance and military immunization programs, though at a substantially smaller scale than the U.S. The region benefits from exceptional pharmaceutical research infrastructure, advanced biomanufacturing capabilities, and strong regulatory frameworks that support continued innovation in anthrax vaccine development. BARDA's sustained funding commitments — including the USD 250 million contract extension for CYFENDUS announced in July 2024 — provide high revenue visibility for North American anthrax vaccine manufacturers through the forecast period, ensuring the region's continued dominance in global anthrax vaccine procurement and market revenue.
Asia Pacific
Asia Pacific Is the Fastest-Growing Anthrax Vaccine Market, Driven by the World's First Recombinant Anthrax Vaccine Approval in South Korea, Expanding National Biodefense Programs, and High Veterinary Anthrax Demand
Asia Pacific is the fastest-growing regional segment in the Anthrax Vaccine market, projected to expand at a CAGR of approximately 5.80% from 2026 to 2033 — the highest of any region. South Korea has emerged as the most strategically significant growth driver within the region following the April 2025 approval of Barythrax by GC Biopharma — the world's first licensed recombinant anthrax vaccine, developed in partnership with the Korea Disease Control and Prevention Agency. This landmark approval positions South Korea as an international leader in next-generation biodefense vaccine development and creates a new domestic and export market for recombinant anthrax vaccine technology that is expected to expand significantly during the forecast period. India, China, and Australia are also growing their national biodefense stockpile investments and military immunization capabilities, while the region's enormous livestock populations across South and Southeast Asia generate substantial and growing veterinary anthrax vaccine demand that is a distinct and important contributor to regional market growth.
Japan and Australia represent more mature anthrax vaccine sub-markets within Asia Pacific, where established biodefense preparedness frameworks and growing recognition of biological threat risks are driving measured but consistent government procurement activity. Indian Immunologicals Limited is a key regional anthrax vaccine manufacturer serving both domestic and export markets across Asia and Africa, particularly for veterinary anthrax vaccination applications where demand remains strong in endemic-region markets. The combination of the world's most rapidly advancing national biodefense programs, significant livestock-sector anthrax vaccine demand, and the region's first recombinant anthrax vaccine approval makes Asia Pacific the most commercially dynamic and fastest-growing regional market in the global Anthrax Vaccine market through 2033.
Top Key Players
-
Emergent BioSolutions Inc. (United States)
-
GC Biopharma (South Korea)
-
Indian Immunologicals Limited (India)
-
Porton Biopharma (United Kingdom)
-
Valneva SE (France)
-
DynPort Vaccine Company LLC (United States)
-
Altimmune, Inc. (United States)
-
Colorado Serum Company (United States)
-
Merck & Co., Inc. (United States)
-
Zoetis Inc. (United States)
-
Bayer AG — Animal Health Division (Germany)
-
Botswana Vaccine Institute (Botswana)
-
VECOL S.A. (Colombia)
-
Biogénesis Bagó (Argentina)
Recent Developments
-
GC Biopharma (April 2025) — Received landmark Ministry of Food and Drug Safety approval in South Korea for Barythrax (GC-1109) — the world's first licensed recombinant anthrax vaccine — co-developed with the Korea Disease Control and Prevention Agency. This historic approval marks a pivotal milestone in next-generation biodefense vaccine development and significantly enhances South Korea's biological threat preparedness capabilities while establishing GC Biopharma as a global leader in recombinant anthrax vaccine technology.
-
Emergent BioSolutions (July 2024) — Secured a USD 250 million contract extension from BARDA for continued supply of CYFENDUS (anthrax vaccine adsorbed, adjuvanted) to the U.S. Strategic National Stockpile, reinforcing its position as the primary anthrax vaccine supplier to the U.S. government and ensuring continued high-value revenue from long-term government procurement commitments through the forecast period.
-
Emergent BioSolutions (January 2024) — Announced an indefinite delivery, indefinite quantity (IDIQ) contract award for the supply of BioThrax as a pre-exposure prophylaxis vaccine to all branches of the United States Armed Forces, securing a significant multi-year military procurement commitment that strengthens the company's revenue visibility and reinforces the BioThrax franchise's continued role in the U.S. military anthrax immunization program.
-
Altimmune, Inc. (2025) — Advanced its next-generation intranasal anthrax vaccine candidate through continued BARDA-supported development, reporting encouraging preclinical immunogenicity and protective efficacy data that supports the program's potential as a mucosal delivery alternative to conventional intramuscular anthrax vaccine administration — a format that could significantly improve military vaccination compliance and convenience.
-
Porton Biopharma (2024) — Expanded its anthrax vaccine manufacturing capacity at the Porton Down facility in the United Kingdom as part of the UK Government's broader biodefense stockpile enhancement program, increasing production capabilities for anthrax vaccine products designated for UK strategic national stockpile reserves and allied nation supply commitments.
Market Trends
The Global Transition Toward Next-Generation Recombinant Anthrax Vaccines and the Expansion of International Biodefense Stockpiling Programs Are the Two Most Consequential Trends Reshaping the Anthrax Vaccine Market
The most transformative trend in the Anthrax Vaccine market is the progressive global transition from conventional cell-free protective antigen adsorbed vaccines toward next-generation recombinant and adjuvanted anthrax vaccine platforms. The April 2025 approval of GC Biopharma's Barythrax in South Korea — the world's first licensed recombinant anthrax vaccine — marked the beginning of a new product generation cycle in the anthrax vaccine space that is expected to accelerate during the 2026–2033 forecast period as additional recombinant candidates advance through regulatory submission in the U.S. and other major markets. These next-generation products offer compelling advantages over the established BioThrax formulation — including improved tolerability, fewer required doses, longer stockpile shelf lives, and potentially more rapid induction of protective immunity — attributes that are highly valued by military immunization program managers and government stockpile authorities seeking more efficient and operationally practical vaccine options.
The second defining trend is the deliberate and growing expansion of international government investment in anthrax biodefense stockpiles beyond the traditional North American and European core markets. Governments across Asia Pacific — particularly South Korea, India, Australia, and increasingly China — are recognizing biological threat preparedness as an integral component of national security strategy and are investing in anthrax vaccine procurement programs for military forces and strategic national stockpiles. This international stockpile expansion trend is creating new government procurement opportunities for both established manufacturers and emerging next-generation vaccine developers, progressively diversifying the anthrax vaccine market's geographic revenue base and reducing its historical dependence on U.S. government procurement cycles.
Segments Covered in the Report
By Vaccine Platform:
-
Cell-Free Protective Antigen Vaccine Adsorbed
-
Adjuvanted Protective Antigen Vaccine Adsorbed
-
Recombinant Protective Antigen Subunit Vaccine
-
Live Attenuated Spore Vaccine for Human Use
-
Veterinary Live Spore Vaccine
By Indication:
-
Pre-Exposure Prophylaxis
-
Post-Exposure Prophylaxis
By End User:
-
Government Stockpiles and Public Health Agencies
-
Military and Defense Organizations
-
Occupational Risk Programs (Laboratories, First Responders, Animal Handlers)
-
Hospitals and Clinics
By Procurement Channel:
-
Direct Government Procurement
-
Defense Medical Supply Procurement
-
Hospital and Institutional Procurement
-
Specialty Distributors
By Region:
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the Anthrax Vaccine market and what is its projected value by 2033?
Answer: The Anthrax Vaccine market was valued at USD 13.88 billion in 2025 and is projected to reach USD 22.04 billion by 2033. It is expected to grow at a CAGR of 4.05% from 2026 to 2033, driven by sustained government biodefense stockpile investment, military immunization mandates, and the commercial advancement of next-generation recombinant anthrax vaccine products.
Question 2: What are the primary growth drivers of the Anthrax Vaccine market?
Answer: Persistent bioterrorism threat awareness sustaining government biodefense investment and mandatory military anthrax vaccination programs in the U.S. and allied nations are the two most powerful drivers of the Anthrax Vaccine market. The progressive approval and commercialization of next-generation recombinant anthrax vaccines and expanding international biodefense stockpile programs are also meaningfully accelerating market growth globally.
Question 3: Which region leads the Anthrax Vaccine market and which is the fastest growing?
Answer: North America holds the dominant position in the Anthrax Vaccine market with approximately 44.70% of global revenue in 2025, driven primarily by large-scale U.S. government procurement. Asia Pacific is the fastest-growing region, expanding at approximately 5.80% CAGR from 2026 to 2033, driven by South Korea's landmark recombinant anthrax vaccine approval and expanding national biodefense programs across the region.
Question 4: Which vaccine platform and indication segment lead the Anthrax Vaccine market?
Answer: Cell-free protective antigen vaccine adsorbed — represented by BioThrax and CYFENDUS — leads the vaccine platform segment with approximately 63.40% of total Anthrax Vaccine market revenue in 2025. Pre-exposure prophylaxis is the dominant indication, contributing approximately 58.70% of global market revenue, driven by large-scale mandatory military and occupational health vaccination programs.
Question 5: What is the most significant recent development shaping the future of the Anthrax Vaccine market?
Answer: The April 2025 South Korean regulatory approval of GC Biopharma's Barythrax — the world's first licensed recombinant anthrax vaccine — is the most consequential recent development in the Anthrax Vaccine market. This landmark approval signals the beginning of a new next-generation product cycle that is expected to expand the licensed vaccine landscape, improve upon the conventional BioThrax formulation's limitations, and progressively reshape government procurement preferences during the 2026–2033 forecast period.
