Allergic Rhinitis Drugs Market Overview
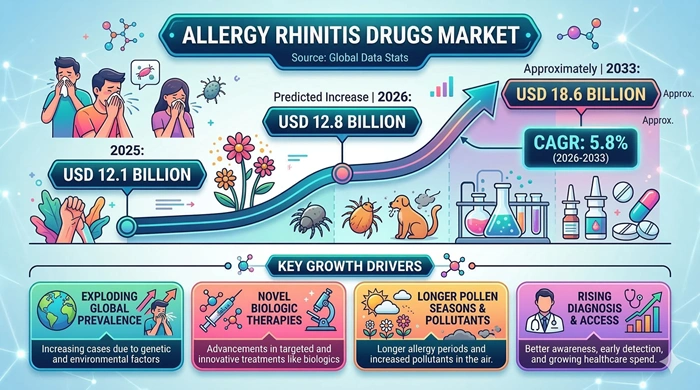
The global allergic rhinitis drugs market size is valued at USD 12.1 billion in 2025 and is predicted to increase from USD 12.8 billion in 2026 to approximately USD 18.6 billion by 2033, growing at a CAGR of 5.8% from 2026 to 2033.
Allergic rhinitis is one of the most prevalent chronic respiratory conditions worldwide, affecting hundreds of millions of people across all age groups and geographies. Characterized by nasal congestion, sneezing, runny nose, and itchy eyes triggered by allergens such as pollen, dust mites, and pet dander, the condition significantly impacts quality of life and workplace productivity. As environmental factors continue to worsen allergy burdens globally and pharmaceutical innovation delivers more effective and convenient treatment options, the allergic rhinitis drugs market is positioned for steady and sustained long-term growth.
AI Impact on the Allergic Rhinitis Drugs Industry
Artificial Intelligence Is Transforming Allergy Drug Discovery, Patient Stratification, and Personalized Treatment Planning, Accelerating Innovation Across the Entire Allergic Rhinitis Drugs Ecosystem
Artificial intelligence is beginning to reshape how allergic rhinitis is understood, diagnosed, and managed pharmacologically. AI-powered platforms are enabling pharmaceutical researchers to analyze large-scale genomic and immunological datasets to identify novel drug targets beyond conventional histamine and leukotriene pathways, opening the door to next-generation therapies with improved efficacy and reduced side effect profiles. Machine learning models are also being used in clinical trial design to identify patient subgroups most likely to respond to specific drug classes, improving trial efficiency and accelerating the regulatory approval timeline for new allergic rhinitis treatments.
Beyond R&D, AI is improving real-world patient management in the allergic rhinitis drugs space. Digital health platforms equipped with AI algorithms can analyze environmental allergen data, patient symptom logs, and medication adherence patterns to generate personalized treatment recommendations. These tools are helping allergists and primary care physicians optimize drug selection and dosing for individual patients, reducing the trial-and-error prescribing cycle that many rhinitis sufferers experience. As AI-driven digital therapeutics become more integrated with traditional pharmacotherapy, the overall standard of care for allergic rhinitis management is expected to improve significantly throughout the forecast period.
Growth Factors
Rising Global Allergy Prevalence, Expanding OTC Drug Availability, and Growing Demand for Disease-Modifying Immunotherapy Are the Core Growth Engines of the Allergic Rhinitis Drugs Market
The most fundamental driver of the allergic rhinitis drugs market is the relentless increase in allergy prevalence worldwide. Urbanization, climate change, increasing airborne pollen seasons, and rising indoor allergen exposure are collectively contributing to a growing global population of allergic rhinitis sufferers. The World Allergy Organization estimates that allergic rhinitis affects up to 40% of the global population, and this patient base continues to expand across both developed and developing markets. As more individuals seek pharmacological relief for increasingly severe and prolonged allergy seasons, demand for antihistamines, intranasal corticosteroids, and combination therapies is growing consistently year over year.
The expanding availability of over-the-counter antihistamines and nasal corticosteroids is simultaneously lowering the barrier for self-managed allergic rhinitis treatment, driving high-volume retail pharmacy sales globally. Second-generation antihistamines such as cetirizine, loratadine, and fexofenadine have become household staples in many countries, generating significant recurring revenue for pharmaceutical manufacturers. At the same time, growing awareness of allergen immunotherapy as a disease-modifying option — capable of reducing long-term symptom burden rather than simply managing symptoms — is driving increased adoption of both subcutaneous and sublingual immunotherapy products, adding a high-value segment to the overall allergic rhinitis drugs market.

Market Outlook
The Allergic Rhinitis Drugs Market Maintains a Positive Long-Term Outlook Through 2033, Supported by Biologic Drug Entry, Expanding Immunotherapy Access, and Rising Healthcare Investment in Emerging Economies
The long-term outlook for the allergic rhinitis drugs market remains consistently positive across the forecast period. The entry of biologic therapies targeting key allergy pathway molecules — such as dupilumab, which has shown clinical benefit in allergic rhinitis patients with comorbid conditions — is opening a premium treatment segment that commands significantly higher per-patient revenue compared to conventional pharmacotherapy. As biologic agents gain broader regulatory approvals and reimbursement coverage for allergic rhinitis indications, they are expected to contribute meaningfully to overall market value growth through 2033.
Emerging markets are expected to be an important source of incremental growth for the allergic rhinitis drugs market over the forecast period. In Asia Pacific, Latin America, and the Middle East, rising urbanization, worsening air quality, and growing middle-class populations with increased healthcare access are expanding both the diagnosed patient population and the willingness to invest in quality allergy treatment. Pharmaceutical companies are actively tailoring their product strategies for these markets, including launching affordable generic antihistamine and corticosteroid combinations to capture high-volume, price-sensitive patient segments while also introducing premium branded therapies for urban consumers with higher purchasing power.
Expert Speaks
-
AstraZeneca CEO Pascal Soriot noted that respiratory and allergy conditions represent one of the most important therapy areas for the company, stating that the convergence of biologic innovation and digital health is creating unprecedented opportunities to deliver more effective, personalized treatment solutions for patients living with allergic rhinitis and related airway disorders globally.
-
Sanofi CEO Paul Hudson emphasized that allergy immunotherapy represents the only currently available disease-modifying approach for allergic rhinitis, and that Sanofi's investment in next-generation sublingual immunotherapy products reflects the company's belief that shifting treatment from symptom suppression to long-term tolerance induction is the future of the allergic rhinitis drugs market.
-
Johnson & Johnson CEO Joaquin Duato highlighted the growing significance of combination allergy therapies, noting that patients with moderate to severe allergic rhinitis increasingly require multi-mechanism treatment approaches, and that J&J's pipeline investments are focused on delivering convenient, efficacious combination products that address the full symptom burden of this highly prevalent condition.
Key Report Takeaways
-
North America leads the allergic rhinitis drugs market, holding the largest revenue share driven by high allergy prevalence, strong OTC pharmaceutical infrastructure, widespread physician awareness of immunotherapy options, and robust health insurance reimbursement frameworks.
-
Asia Pacific is the fastest-growing region, propelled by rapidly worsening urban air quality, rising pollen seasons, a large and underserved allergy patient population across China, India, and Japan, and growing healthcare expenditure in key emerging economies.
-
Retail and online pharmacy channels are the dominant distribution segment, reflecting the widespread OTC availability of antihistamines and nasal corticosteroids, which drive the highest prescription and self-medication volumes across all major markets.
-
Intranasal corticosteroids contribute the largest share of market revenue by drug class, recognized as the most clinically effective first-line pharmacological treatment for moderate to severe allergic rhinitis symptoms by major allergy clinical guidelines globally.
-
Second-generation antihistamines are the most widely used drug category overall, owing to their favorable safety profile, non-sedating properties, and broad OTC availability, making them the first choice for self-managed seasonal and perennial allergic rhinitis treatment worldwide.
-
Allergen immunotherapy is the fastest-growing treatment segment, expected to grow at a CAGR above the overall market average through 2033 with approximately 17% market share, as sublingual immunotherapy tablets gain broader regulatory approval and patient preference for disease-modifying rather than purely symptomatic treatment continues to strengthen.
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size by 2033 | USD 18.6 Billion |
| Market Size by 2025 | USD 12.1 Billion |
| Market Size by 2026 | USD 12.8 Billion |
| Market Growth Rate (2026–2033) | CAGR of 5.8% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2033 |
| Segments Covered | Drug Class, Distribution Channel, Disease Type, End User |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Market Dynamics
Drivers Impact Analysis
Surging Global Allergy Burden, OTC Drug Expansion, and Biologic Therapy Entry Are the Most Powerful Forces Driving Sustained Revenue Growth Across the Allergic Rhinitis Drugs Market
| Driver | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising global allergic rhinitis prevalence | ~30% | Global | Near to Long-term |
| Expanding OTC antihistamine and corticosteroid availability | ~25% | Global | Near to Mid-term |
| Growing adoption of allergen immunotherapy | ~22% | North America, Europe | Mid to Long-term |
| Entry of biologic therapies for severe allergic rhinitis | ~15% | North America, Europe | Mid to Long-term |
| Increasing healthcare spending in emerging markets | ~8% | Asia Pacific, Latin America | Long-term |
The global escalation in allergic rhinitis prevalence is the most consistent and durable driver of the allergic rhinitis drugs market. Climate change is extending pollen seasons, increasing allergen concentrations, and introducing new allergenic plant species into regions previously unaffected, driving a measurable increase in both seasonal and perennial rhinitis cases. Urbanization is compounding this trend by increasing exposure to traffic-related pollutants that sensitize airways and amplify allergic responses, particularly in rapidly developing Asian and Latin American cities. These environmental shifts are expanding the patient population continuously, creating a growing base of consumers seeking pharmacological relief.
The commercialization of biologic therapies targeting IgE and type 2 inflammatory pathways represents a transformative growth driver for the premium end of the allergic rhinitis drugs market. Dupilumab, originally approved for atopic dermatitis and asthma, has demonstrated significant clinical benefits in patients with comorbid allergic rhinitis, and pharmaceutical companies including Sanofi and Regeneron are actively pursuing dedicated rhinitis indications. These high-value biologic agents carry annual treatment costs orders of magnitude higher than generic antihistamines, meaning even relatively modest patient volumes in the biologic segment can generate substantial market revenue, significantly lifting overall market value projections through 2033.

Restraints Impact Analysis
Generic Drug Competition, Patent Expiries of Key Branded Products, and Price Sensitivity in Emerging Markets Are the Most Significant Restraints Constraining Revenue Growth in This Market
| Restraint | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Extensive generic competition in antihistamine segment | ~35% | Global | Ongoing |
| Patent expiries of branded intranasal corticosteroids | ~28% | North America, Europe | Near to Mid-term |
| Price sensitivity and limited insurance in emerging markets | ~22% | Asia Pacific, Latin America, MEA | Near-term |
| Self-medication trends reducing prescription drug sales | ~15% | Global | Ongoing |
Generic drug competition remains the most persistent structural restraint on revenue growth in the allergic rhinitis drugs market. The majority of widely used antihistamines and intranasal corticosteroids have long since lost patent protection, creating intensely competitive generic markets where price erosion is severe and branded products struggle to maintain premium positioning. Companies that previously derived significant revenue from branded cetirizine, loratadine, or fluticasone propionate formulations have seen dramatic revenue compression as generics captured the bulk of market volume at substantially lower price points, limiting the overall average selling price growth in the largest drug class segments.
The widespread self-medication behavior among allergic rhinitis patients further constrains the prescription drug market. Because second-generation antihistamines and many nasal corticosteroid sprays are available without prescription in major markets, a large proportion of patients manage their symptoms independently without physician consultation, bypassing the prescription channel entirely. This dynamic reduces the commercial opportunity for higher-priced prescription-only products and makes it challenging for pharmaceutical companies to differentiate their branded offerings in a market where patient decision-making is largely driven by convenience and price rather than clinical differentiation.
Opportunities Impact Analysis
Sublingual Immunotherapy Tablets, Biologic Drug Approvals, and Digital Health-Integrated Allergy Management Present the Most Compelling Growth Opportunities in the Allergic Rhinitis Drugs Market
| Opportunity | ≈ % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Sublingual immunotherapy tablet market expansion | ~33% | North America, Europe, Asia Pacific | Mid to Long-term |
| Biologic therapy approvals for allergic rhinitis | ~28% | North America, Europe | Mid to Long-term |
| Digital health and AI-driven allergy management platforms | ~22% | Global | Mid-term |
| Generic market expansion in emerging economies | ~17% | Asia Pacific, Latin America | Near to Mid-term |
Sublingual immunotherapy tablets represent one of the most commercially exciting opportunities in the allergic rhinitis drugs market. Unlike traditional subcutaneous immunotherapy — which requires regular clinic visits for injections — sublingual tablets can be self-administered at home, significantly improving patient convenience and adherence. Products such as ALK's GRAZAX and Stallergenes Greer's ORALAIR have already demonstrated strong commercial traction in European markets, and ongoing regulatory submissions in North America and Asia Pacific are expected to expand market access considerably. The combination of disease-modifying efficacy, patient-friendly administration, and premium pricing positions sublingual immunotherapy as a major long-term revenue growth driver.
Emerging markets offer a parallel high-volume opportunity driven primarily by the expansion of affordable generic antihistamine and corticosteroid products. In countries such as India, Brazil, Indonesia, and Mexico, a large and growing allergy patient population currently has limited access to quality pharmacological treatment due to cost and healthcare access barriers. As domestic generic manufacturers expand their allergic rhinitis product portfolios and retail pharmacy networks improve drug distribution, this patient population represents a substantial incremental revenue opportunity for companies positioned to compete effectively in price-sensitive, high-volume market segments.

Segment Analysis
By Drug Class
Intranasal Corticosteroids Lead the Allergic Rhinitis Drugs Market as the Most Clinically Preferred First-Line Therapy, While Allergen Immunotherapy Emerges as the Fastest-Growing and Highest-Value Segment
Intranasal corticosteroids account for approximately 34% of the total allergic rhinitis drugs market revenue and hold the top position among all drug classes due to their recognition by international allergy guidelines as the most effective pharmacological treatment for moderate to severe allergic rhinitis. Agents such as fluticasone propionate, mometasone furoate, and budesonide deliver comprehensive symptom control across all nasal symptom domains, including congestion, sneezing, and rhinorrhea, with a favorable local safety profile that supports long-term use. The segment is expected to grow at a CAGR of approximately 5.4% through 2033, driven by new formulation innovations including combination nasal sprays pairing corticosteroids with antihistamines in a single delivery device. North America leads intranasal corticosteroid adoption, with companies such as GlaxoSmithKline, Sanofi, and Bayer maintaining strong branded and generic positions in this segment through extensive retail pharmacy distribution networks.
Asia Pacific is rapidly emerging as a growth market for intranasal corticosteroids within the broader allergic rhinitis drugs landscape, driven by rising allergy diagnosis rates and growing physician awareness of guideline-recommended treatment approaches. In markets such as China and Japan, where antihistamines have historically dominated allergic rhinitis prescribing patterns, increasing medical education initiatives and the expanding availability of branded intranasal corticosteroid products are gradually shifting prescribing behavior toward more comprehensive symptom management. Companies including Takeda Pharmaceutical and local Chinese manufacturers are actively expanding their intranasal corticosteroid portfolios to capture this market shift, contributing to strong projected segment growth in Asia Pacific through 2033.
By Disease Type
Seasonal Allergic Rhinitis Dominates Overall Market Revenue, While Perennial Allergic Rhinitis Represents a Growing and Commercially Significant Disease Segment in the Allergic Rhinitis Drugs Market
Seasonal allergic rhinitis accounts for approximately 58% of the total allergic rhinitis drugs market revenue, driven by the large patient population affected by pollen-triggered allergy symptoms during spring and summer months. This condition is particularly prevalent in North America, Europe, and parts of Asia where high-allergen plant species generate significant airborne pollen loads during seasonal peak periods. The segment is expected to grow at a CAGR of approximately 5.5% through 2033, sustained by lengthening pollen seasons due to climate change and the growing market penetration of both OTC and prescription allergy therapies. Key companies generating significant revenue from seasonal allergic rhinitis treatments include Sanofi, ALK-Abelló, Stallergenes Greer, and GlaxoSmithKline, each offering a range of antihistamines, corticosteroid sprays, and immunotherapy products targeting this large patient population.
Perennial allergic rhinitis — triggered year-round by indoor allergens such as dust mites, mold, and pet dander — represents a growing and commercially valuable disease type segment within the allergic rhinitis drugs market, holding approximately 42% of total market share and expected to grow at a slightly faster CAGR than seasonal rhinitis through 2033. The chronic, continuous nature of perennial rhinitis means affected patients require sustained pharmacological management rather than seasonal treatment, generating higher per-patient annual drug expenditure and greater long-term revenue per treated individual. Sublingual immunotherapy products specifically formulated for house dust mite allergy — including ALK's ACARIZAX tablet — are gaining strong commercial traction in Europe and are advancing through regulatory pathways in Asia Pacific, positioning this as a particularly dynamic sub-segment for growth within the perennial disease category.

Regional Insights
North America
North America Dominates the Global Allergic Rhinitis Drugs Market, Backed by the Highest Per-Capita Allergy Treatment Spending, Widespread OTC Availability, and a Rapidly Expanding Immunotherapy Adoption Landscape
North America holds approximately 38% of the global allergic rhinitis drugs market share and is expected to maintain its leading position through 2033, growing at a CAGR of approximately 5.4%. The United States is the dominant contributor to regional revenue, driven by extremely high seasonal allergy prevalence — with an estimated 60 million Americans affected by allergic rhinitis — and a well-developed pharmaceutical distribution ecosystem that makes both prescription and OTC allergy medications widely accessible. Strong health insurance reimbursement frameworks support physician-prescribed immunotherapy programs, and growing consumer awareness of sublingual immunotherapy tablets is generating incremental demand in the premium treatment segment. Key companies with major North American commercial presence include Sanofi (France/U.S.), ALK-Abelló (Denmark), GlaxoSmithKline (U.K.), and Johnson & Johnson (U.S.).
Canada represents a steadily growing contribution to the North American allergic rhinitis drugs market, with rising allergy diagnosis rates and a public healthcare system that is progressively incorporating evidence-based allergy treatment guidelines into provincial formularies. The Canadian allergy immunotherapy market is expanding, supported by growing allergist specialty networks and increasing patient awareness of the long-term benefits of disease-modifying therapy. Together, the U.S. and Canada form the world's most commercially mature and revenue-intensive regional market for allergic rhinitis drugs, with continued innovation in biologic therapies and immunotherapy products expected to sustain above-average revenue growth within this region through the end of the forecast period.
Asia Pacific
Asia Pacific Is the Fastest-Growing Region in the Allergic Rhinitis Drugs Market, Driven by Worsening Urban Air Quality, a Massive Underserved Patient Population, and Rapidly Expanding Pharmaceutical Distribution Infrastructure
Asia Pacific is the fastest-growing region in the allergic rhinitis drugs market, expected to register a CAGR of approximately 7.1% through 2033, significantly exceeding the global market average. China is the primary growth engine, with hundreds of millions of urban residents exposed to some of the world's highest levels of traffic-related air pollutants and particulate matter — environmental conditions that are strongly associated with sensitization and allergic airway disease. Rising household incomes, expanding health insurance coverage under national healthcare reform initiatives, and the rapid growth of retail and online pharmacy channels are collectively improving access to allergy medications at scale. Key companies building strong Asia Pacific commercial positions include Takeda Pharmaceutical (Japan), Hisamitsu Pharmaceutical (Japan), and global players including Sanofi and Bayer that are actively localizing their allergic rhinitis drug portfolios for Asian markets.
India and Southeast Asia represent compelling longer-term growth opportunities within the Asia Pacific allergic rhinitis drugs market. Both regions have large, rapidly urbanizing populations with rising allergy burdens and historically low rates of pharmacological allergic rhinitis management due to limited healthcare access and low disease awareness. Expanding primary care physician networks, growing consumer health awareness, and the rapid proliferation of e-commerce pharmacy platforms are gradually closing this access gap. As allergy education initiatives reach more patients and prescribers in these markets, and as affordable generic antihistamine and corticosteroid products become more widely distributed, Asia Pacific's share of global allergic rhinitis drug revenue is expected to increase meaningfully through 2033.
Top Key Players
-
Sanofi S.A. (France)
-
ALK-Abelló A/S (Denmark)
-
GlaxoSmithKline plc (United Kingdom)
-
Johnson & Johnson (United States)
-
Bayer AG (Germany)
-
AstraZeneca plc (United Kingdom)
-
Stallergenes Greer (France / United States)
-
Takeda Pharmaceutical Company Ltd. (Japan)
-
Regeneron Pharmaceuticals Inc. (United States)
-
Novartis AG (Switzerland)
-
Teva Pharmaceutical Industries Ltd. (Israel)
-
Mylan N.V. (Viatris Inc.) (United States)
Recent Developments
-
Sanofi & Regeneron (2025): Sanofi and Regeneron announced the submission of a supplemental Biologics License Application to the FDA for dupilumab in adults with moderate to severe allergic rhinitis inadequately controlled by standard pharmacotherapy, potentially opening a major new biologic indication within the allergic rhinitis drugs market and significantly expanding the addressable patient population for this high-value therapy.
-
ALK-Abelló (2024): ALK-Abelló reported strong commercial growth for its ACARIZAX house dust mite sublingual immunotherapy tablet across key European markets and announced the initiation of regulatory submissions in multiple new Asian markets, continuing to build the global commercial footprint for its disease-modifying allergy treatment portfolio.
-
GlaxoSmithKline (2024–2025): GlaxoSmithKline expanded its consumer healthcare allergy portfolio through the launch of new combination antihistamine and decongestant formulations targeting convenience-driven OTC consumers, and continued to invest in digital allergy management tools integrated with its branded allergy product line to improve patient engagement and adherence.
-
Stallergenes Greer (2025): Stallergenes Greer received a new European CE mark extension for an expanded range of allergen extract products used in subcutaneous immunotherapy programs, reinforcing its position as a leading provider of individualized allergen immunotherapy solutions for allergic rhinitis patients across European specialty allergy centers.
-
Takeda Pharmaceutical (2024): Takeda Pharmaceutical strengthened its position in the Japanese and broader Asian allergic rhinitis drugs market through the launch of an updated formulation of its flagship antihistamine product with improved once-daily dosing convenience, and announced licensing agreements with regional distributors in Southeast Asia to accelerate market penetration in high-growth emerging markets.
Market Trends
The Rise of Combination Drug Therapies, Sublingual Immunotherapy Tablet Adoption, and Biologic Entry Are the Defining Trends Reshaping the Competitive Landscape of the Allergic Rhinitis Drugs Market
One of the most commercially significant trends reshaping the allergic rhinitis drugs market is the rapid growth of fixed-dose combination intranasal sprays that pair corticosteroids with antihistamines in a single device. Products such as Dymista — combining azelastine and fluticasone propionate — have demonstrated superior efficacy compared to either component alone for moderate to severe allergic rhinitis, and their clinical differentiation has supported premium pricing even in a market dominated by generics. As more pharmaceutical companies invest in developing novel combination nasal spray formulations, this trend is expected to become a primary commercial battleground among major allergy drug manufacturers through 2033, driving both revenue growth and prescribing pattern shifts.
The accelerating commercial adoption of sublingual immunotherapy tablets is simultaneously transforming the treatment paradigm for allergic rhinitis toward a disease-modification model. Unlike all symptomatic drug classes — which require continuous use to maintain effect — sublingual immunotherapy tablets can induce lasting allergen tolerance with a defined treatment course, offering patients the prospect of long-term symptom reduction without lifelong pharmacotherapy. This compelling value proposition is gaining recognition among both physicians and health insurers, gradually shifting market dynamics toward disease-modifying treatment and raising the overall clinical and commercial bar for the allergic rhinitis drugs industry through the end of the forecast period.
Segments Covered in the Report
By Drug Class
-
Antihistamines
-
First Generation Antihistamines
-
Second Generation Antihistamines
-
Intranasal Corticosteroids
-
Decongestants
-
Mast Cell Stabilizers
-
Leukotriene Receptor Antagonists
-
Immunotherapy
-
Subcutaneous Immunotherapy
-
Sublingual Immunotherapy
-
Others
By Distribution Channel
-
Hospital Pharmacies
-
Retail Pharmacies
-
Online Pharmacies
By Disease Type
-
Seasonal Allergic Rhinitis
-
Perennial Allergic Rhinitis
By End User
-
Hospitals & Clinics
-
Homecare Settings
-
Specialty Allergy Centers
-
Others
By Region
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
Frequently Asked Questions
Question 1: What is the current size of the global allergic rhinitis drugs market and what growth is expected by 2033?
Answer: The global allergic rhinitis drugs market is valued at USD 12.1 billion in 2025 and is projected to reach USD 18.6 billion by 2033. The market is expected to grow at a CAGR of 5.8% from 2026 to 2033.
Question 2: What are the primary growth drivers in the allergic rhinitis drugs market?
Answer: The key drivers include rising global allergy prevalence driven by climate change and urbanization, and the expanding availability of OTC antihistamines and nasal corticosteroids. Growing adoption of disease-modifying allergen immunotherapy and the entry of biologic therapies are also significantly lifting market value.
Question 3: Which drug class dominates the allergic rhinitis drugs market?
Answer: Intranasal corticosteroids hold the largest revenue share in the allergic rhinitis drugs market at approximately 34%, recognized as the most clinically effective first-line treatment for moderate to severe allergic rhinitis by international guidelines. Second-generation antihistamines are the most widely used drug category overall due to their broad OTC availability and favorable safety profile.
Question 4: Which region leads the allergic rhinitis drugs market and which is the fastest growing?
Answer: North America leads the allergic rhinitis drugs market with approximately 38% of global revenue share, driven by high allergy prevalence and strong pharmaceutical infrastructure. Asia Pacific is the fastest-growing region, projected to grow at a CAGR of approximately 7.1% through 2033 due to worsening urban air quality and a large underserved patient population.
Question 5: What is allergen immunotherapy and why is it important for the future of the allergic rhinitis drugs market?
Answer: Allergen immunotherapy is the only currently available disease-modifying treatment for allergic rhinitis, capable of reducing long-term allergy severity by inducing tolerance rather than simply managing symptoms. Its growing adoption — especially in the form of convenient sublingual immunotherapy tablets — is expected to become one of the most important growth drivers of the allergic rhinitis drugs market through 2033.
